Environmental application of an industrial waste as catalyst for the electro-Fenton-like treatment of organic pollutants
Elena Alfaya,
Olalla Iglesias,
Marta Pazos and
Maria Angeles Sanromán*
Department of Chemical Engineering, University of Vigo, Isaac Newton Building, Campus As Lagoas, Marcosende 36310, Vigo, Spain. Fax: +34 986 812380; Tel: +34 986 812383
First published on 20th January 2015
Abstract
The application of acid mine drainage sludge (AMDS), an industrial waste with high metal content, as catalyst for the electro-Fenton-like technology on the treatment of organic polluted effluents has been investigated. The study has demonstrated the potential use of this waste using dye Lissamine Green B as a model contaminant, which allowed the optimization of sludge dosage and pH. AMDS proved to perform a high decoloration rate at pH 2, however, acidic conditions favored metal leaching. Alginate gel beads were selected to immobilize AMDS and to avoid metal release and the wash out of the reactor operating in continuous mode. This developed catalyst showed high reliability and efficiency in successive batches. The efficacy of the established treatment was also verified on the degradation of pesticide pirimicarb and a degradation pathway was determined by LC-MS studies.
1. Introduction
Contamination of water courses with organic compounds is an increasing concern due to their toxicity. Industrial and agricultural development creates large amounts of recalcitrant organic pollutants whose complexity hinders their treatment with conventional methods. To solve this difficulty the application of advanced oxidation processes (AOPs) is being investigated due to their capability to degrade and mineralize organic pollutants. Traditional AOPs involve the in situ generation of highly reactive radical species, predominantly the hydroxyl radical. This is a very reactive oxidant species, with a high redox potential (2.80 V versus standard hydrogen electrode).1,2 The use of different AOPs such as Fenton processes, photocatalysis, UV-H2O2, ozone, ozone-UV, plasma based processes and sonolysis have been reported in the literature as effective processes for the treatment of diverse wastewaters.3–6 However, the high capital and operating cost requirements due to the elevated use of reagents such us H2O2, ozone or the application of UV radiation in addition to the hazards of their handling hinders the use of such technologies. Therefore, electrochemical advanced oxidation processes (EAOPs) provide an alternative that enables the in situ generation of highly reactive radical species; among them, the electro-Fenton processes have attracted attention in the last years. It permits the Fenton reactions to take place in the reactor without addition of H2O2, with higher degradation ability in comparison to the Fenton process.7 In the electro-Fenton process, Fe2+ catalyzes the hydroxyl radical generation from H2O2 (eqn (1)), however it requires strict acid conditions to prevent iron precipitation. Other transition metals can also catalyze the reaction (eqn (2)) as reported Bokare and Choi:8| H2O2 + Fe2+ → HO˙ + OH− + Fe3+ | (1) |
| H2O2 + M(n)+ → HO˙ + OH− + M(n+1)+ | (2) |
Rapid industrialization and urbanization has resulted in the release of highly metal polluted wastes from various industrial processes.9 Acid mine drainage (AMD) is a persistent environmental problem at many active and abandoned sulphide and coal mine sites.10–12 This drainage is characterized by often containing elevated concentrations of metals and metalloids in addition to its low pH value. The most widespread method used to remediate these effluents is the addition of chemical-neutralizing agents that cause many of the metals to precipitate. The result is the production of an iron-rich sludge such as AMDS, which may also contain various other metals, depending on the chemistry of the mine water treated.11
There are some studies on the reuse of this material for wastewater treatment as low cost adsorbents for the removal of phosphate or dyes.13–15 Flores14 recovered iron oxides from AMD to be used as catalyst of an azo dye by means of Fenton-like treatment, attaining results comparable to those of analytical-grade reagents.
Furthermore, to allow the reuse of catalyst avoiding the generation of metal sludge, heterogeneous Fenton and Fenton-like treatments using several supporting materials to fix the metal have been evaluated. Recently, several catalyst supports have been studied such as clays,15–17 hydrogels,18–20 among others.21–23 Other studies use iron-bearing solid catalysts that are naturally iron rich materials, including iron oxides such as goethite (α-FeOOH),24,25 ferrihydrite (Fe5HO8·4H2O),26 lepidocrocite (γ-FeOOH),27 hematite,28 and magnetite (Fe3O4).28–30 Most of these supports are raw materials; however, few studies deal with the use of wastes as catalyst for AOPs. Martins31 tested iron shavings as low cost materials in the Fenton treatment of actual olive mill wastewaters and Oliveira32 evaluated the use of pipe deposits from water networks for the Fenton treatment of pesticide paraquat polluted wastewaters with successful results even at neutral pH conditions.
Therefore, the reuse of the highly iron loaded industrial waste AMDS as catalyst for hydroxyl radicals production from H2O2 seems to be a new low cost approach for the implementation of highly efficient EAOPs. For this reason, the aim of this study is the application and optimization of AMDS as catalyst for the electro-Fenton-like treatment (EF-AMDS) of organic pollutants. The treatment was firstly optimized by testing the AMDS dosage, pH effect and the immobilization of AMDS in alginate gel beads for the treatment of a model pollutant (Lissamine Green B). After the determination of optimal operational conditions a deeply study of the degradation process on a highly soluble pesticide such as pirimicarb was carried out.
2. Experimental
2.1. Materials
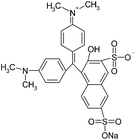
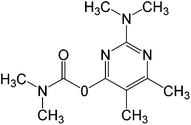
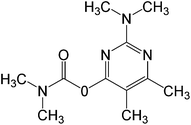
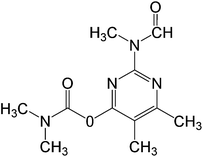
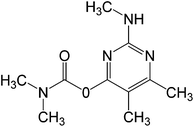
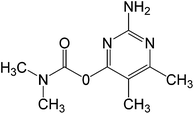
In this work, solution of Lissamine Green B dye and pirimicarb pesticide at Pestanal® analytical standard grade were provided by Sigma-Aldrich (Barcelona, Spain) and used in order to evaluate the electro-Fenton treatments. Their structure and properties are shown in Table 1.AMDS was supplied by a local coal mining industry, AMDS properties are described in Table 2.
| Parameters | Value |
|---|---|
| Electrical conductivity | 2.9 mS cm−1 |
| pH | 7 |
| Metals | Concentration (ppm) |
|---|---|
| Al | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 047 047 |
| Ba | 250 |
| Ca | 9802 |
| Fe | 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 350 350 |
| Mg | 2919 |
| Mn | 746 |
| Na | 1072 |
| S | 4740 |
| Zn | 113 |
Na-alginate and CaCl2 were analytical grade and supplied by Sigma-Aldrich (Barcelona, Spain).
2.2. AMDS characterization
The morphology of AMDS was observed using Scanning Electron Microscopy (SEM). It was performed on a JEOL JSM-6700F equipped with an EDS Oxford Inca Energy 300 SEM using an accelerating voltage of 20 kV (Electron Microscopy Service, C.A.C.T.I., University of Vigo).The protocols used for the chemical extraction and analysis of metals were performed in accordance with EPA Methods 3010 and 3050. Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES) was used to analyze metals. Duplicate measures were carried out with an experimental error lower than 3%.
2.3. Catalyst preparation
The AMDS was heated in a muffle furnace at 500 °C for 24 h. It was cooled down at room temperature and ground into powder to be used directly as catalyst and furthermore, to be tested embedded in alginate gel. The density of this material was around 1.6 g mL−1. For the latter, 0.05 L of a suspension of Na-alginate (3%) purchased from Sigma-Aldrich (Barcelona, Spain) containing 3 g of AMDS was dropped on a solution of 0.2 M CaCl2 (Sigma-Aldrich (Barcelona, Spain)) to create the spherical alginate gel beads with the AMDS entrapped into its structure (AMDS-AB). These particles were cured at 4 °C for 2 h, then filtered and washed repeatedly with distilled water. Finally, they were stored at 4 °C for the EF-AMDS-AB study.2.4. Electro-Fenton-like reactor using AMDS as catalyst (EF-AMDS)
Several batch experiments were done in a glass cylindrical reactor (60 × 120 mm) with a working volume of 0.15 L.33 The reactor was filled with the catalyst and an aqueous solution of Lissamine Green B dye or pirimicarb pesticide at a desired concentration. These solutions were agitated with a magnetic stirred in order to avoid concentration gradients. For all experiments, the electrolyte utilized was 0.01 M Na2SO4. Electric field was applied by two graphite sheets (Carbon Lorraine, France) connected to a direct current (DC) power supply (HP model 3662) at a constant voltage of 5 V (HP model 3662). The electrodes (surface 15 cm2) were placed opposite to each other with an electrode gap of 60 mm.33,34 A continuous bubbling (1 L min−1) of air at atmospheric pressure was located into the reactor on the cathode surroundings, this flow started 10 min before the electro-Fenton process in order to reach continuous saturation of air at atmospheric pressure. This was carried out for the in situ production of H2O2 by the electrochemical reduction of oxygen.2.5. Samples preparation
In all experiments, samples were taken periodically from the electro-Fenton reactor cells to be analyzed for pH, dye concentration and decoloration and pesticide concentration and mineralization; electric parameters were also recorded. Samples were centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min, and the supernatant was separated to be analyzed.
000 rpm for 5 min, and the supernatant was separated to be analyzed.
2.6. Lissamine Green B analysis
Lissamine Green B concentrations in the samples were measured spectrophotometrically (Unicam Helios β, Thermo Electron Corp.) from 500 to 700 nm using a calibration curve associated with the area under the curve.Dye decoloration, expressed in terms of percentage, was calculated according to the following equation:
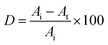 | (3) |
2.7. Pirimicarb analysis
The initial and residual pirimicarb concentrations in the samples were determined by HPLC (Agilent-1100) equipment with an XDF-C8 reverse-phase column (150 × 4.6 mm i.d., 5 μm) using a gradient elution program with acetonitrile and water as the mobile phase. The injection volume was set at 5 μL and the flow rate was 1 mL min−1. Prior to injection, the samples were filtered through a 0.45 μm Teflon filter. Detection was performed with a diode array detector at 246 nm, and the column was maintained at room temperature.In order to identify the transformation products obtained in the pirimicarb degradation several samples were analyzed with a HPLC-MS equipment with a LC column Luna 5u C18 100A. Filtration through a 0.45 μm Teflon filter was done before the injection. In this case the isocratic eluent was 60 (methanol)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 (water, 0.1% of formic acid and 0.1 g L−1 of sodium formate) that was pumped at a rate of 0.4 mL min−1 for 60 min. Detection was carried out with a diode array detector at 220 nm and the column temperature was maintained at 25 °C. The coupled mass spectrometer employed was a Hewlett-Packard 5989B with a detection range from 10 to 2000 Da.
40 (water, 0.1% of formic acid and 0.1 g L−1 of sodium formate) that was pumped at a rate of 0.4 mL min−1 for 60 min. Detection was carried out with a diode array detector at 220 nm and the column temperature was maintained at 25 °C. The coupled mass spectrometer employed was a Hewlett-Packard 5989B with a detection range from 10 to 2000 Da.
2.8. Kinetic studies
Kinetic studies were done in order to model the behavior of the EF-AMDS. The pollutants concentration profiles were fitted by a suitable kinetic equation and the rate constants were calculated by using SigmaPlot 4.00 (1997) software. The SigmaPlot curve fitter uses an iterative procedure, based on the Marquardt–Levenberg algorithm, which seeks the values of the parameters that minimize the sum of the squared differences between the observed and predicted values of the dependent variable.2.9. Measurement of process efficiency
In addition to analyzing pollutants concentration and therefore their removal during the experiments, other specific energetic parameters are useful. In this study, the energy consumption per pollutant (dye and pesticide) mass was evaluated following the eqn (4):
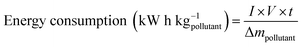 | (4) |
3. Results and discussion
3.1. Characterization of AMDS
The AMDS used in this study was obtained from an active mine drainage facility that neutralizes AMD (containing mainly iron) using carbonates (CaCO3) and hydroxides (Ca(OH)2). Therefore, the main elements of the sludge are iron and calcium as confirms the chemical characterization shown in Table 2. Fig. 1 shows the SEM images of AMDS grains after muffle furnace heating, with a size of 30–100 μm. The metal quantification with ICP-OES indicates the high iron and other transition metals such as manganese and zinc contain which makes this waste a suitable candidate to be used as Fenton-like catalyst.8,14 Thus the efficiency of manganese to degrade different organic pollutants by electro-Fenton process was already determined by Fernández de Dios.203.2. Effect of AMDS dosage
In electro-Fenton processes, the effect of catalyst species has a greater influence on pollutant oxidation because H2O2 is not added to the system but it is in situ generated at the cathode surface and its concentration depends on the dissolved oxygen. Oturan35 established that in the overall reactions 0.5 moles of oxygen with water produces 2 moles of hydroxyl radicals, therefore the required oxygen is very low. H2O2 production rate is constant for each operating conditions, therefore catalyst dosage was optimised as one of the main parameters to increase the production of hydroxyl radicals.Initially, the EF-AMDS was evaluated for the decoloration of dye Lissamine Green B (10 mg L−1). Two AMDS dosages were selected (1 and 3 g) and results are shown in Fig. 2. Usually when the catalyst dosage increases, there is an enhancement in hydroxyl radical production (eqn (2));36,37 however if there is higher catalyst than required secondary reactions can take place and consume H2O2 and hydroxyl radicals.38 The decoloration percentages with 3 g of AMDS are around 10% higher than 1 g of catalyst; for this reason, the bigger amount was selected in order to assure the availability of the low cost catalyst.
 | ||
| Fig. 2 Profiles of decoloration of Lissamine Green B during the EF-AMDS treatments: (●) (1 g of AMDS at pH 2), (■) (3 g of AMDS at pH 2), (♦) (3 g of AMDS at pH 6) and (▲) (3 g of AMDS at pH 8). | ||
The kinetic studies were carried out. In these experiments reaction kinetics were researched and the regression coefficients for zero, first and second-order reactions were calculated. The results indicated that the decoloration of Lissamine Green B under the EF-AMDS could be quantitatively described by a first-order kinetic equation (eqn (5)) with respect to the dye concentration:
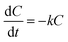 | (5) |
As expected from the degradation profiles, k values show that the percentage of decoloration with time is slightly faster under the electro-Fenton with 3 g of AMDS (k = 0.0922 min−1), compared with 1 g (k = 0.0642 min−1).
3.3. Effect of solution pH
pH is one of the major factors that limits the performance of Fenton-like processes. pH affects the speciation of catalyst and the H2O2 decomposition.8,31,39 The chemical reactivity of iron, the main metal on AMDS, as Fenton catalyst is strictly dependent on the pH-dependent speciation of Fe2+, Fe3+ and ferric hydroxides.8In this study pH 2, an acidic value that commonly attains the highest degradation results, pH 6, the natural pH of solution that does not require the addition of reagents and pH 8 as a basic media were selected to carry out the EF-AMDS by adding H2SO4 or NaOH when necessary. The results for the decoloration of Lissamine Green B are plotted in Fig. 2. As it was expected from previous studies the acid condition highly favours the decoloration of dyes due to the availability of Fe2+ and Fe3+ on the media because the generation of iron hydroxides is avoided.19,33 pH 6 and 8 reduce significantly the decoloration, reaching a 55% after 1 h under the more alkaline conditions. The kinetic coefficient for the first order reaction decreases from 0.0922 min−1 at pH 2 to 0.0222 min−1 at pH 6 and 0.0168 min−1 at pH 8. Therefore, the degradation at neutral and basic pH is possible, however the process requires higher treatment time than pH 2 and this fact increases the energy consumption. Thus, working at pH 2 the energy cost was around 12 kW h kg−1dye and this value increases to 14 kW h kg−1dye when the treatment was carried out at pH 6.
Nevertheless, it is necessary to evaluate the leaching of metals at the different operational conditions tested in this study. It was determined that around a 14.8% of leaching of Al, Ca, Fe, Mg, Mn, Ni and Zn was found after 60 min when EF-AMDS of Lissamine Green B was developed at pH 2, where Fe leaching was 2.7% of initial Fe content on the AMDS on solution. It is known that the acid media favours the release of metals and for this reason the metal content in the liquid phase lowered at pH 6, where just Ca, Mg, Mn and Zn were found, accounting for a 1.8% of initial contain.31 The metal leaching removes catalyst available for the EF-AMDS in continuous mode because of its lost on the outflow; for that reason when the operation was carried out in successive cycles the degradation rate is reduced. This fact was detected when the treatment at pH 6 was tested in two cycles (Fig. 3). In order to solve this problem the AMDS entrapped in hydrogel matrixes such as alginate gel has been proposed.
3.4. Electro-Fenton-like process using AMDS-AB as catalyst (EF-AMDS-AB)
The immobilization of catalyst in different structures is a common strategy to avoid catalyst leaching.40,41 Hydrogels were studied to immobilize Fenton and Fenton-like catalysts in several studies with negligible leaching and high performance.18,20,42In this study, AMDS was immobilized in alginate gel beads due to the feasibility of this hydrogel to contain material with similar physical characteristics without losing its structural properties.16 The EF-AMDS-AB was tested at pH 2 and 6 for the decoloration of Lissamine Green B at the same conditions than free AMDS. Fig. 4 shows the faster and superior decoloration attained when catalyst was immobilized in alginate gel beads. The kinetic constant was much higher than in the EF-AMDS; 0.11225 min−1 at pH 2 and 0.0359 min−1 at pH 6. Acid conditions favored the availability of catalyst species even when they were trapped in the alginate gel beads; furthermore low pH is favorable for the production of H2O2 because the conversion of dissolved oxygen to H2O2 consumes protons from the acidic solution.15 An increase in the decoloration of dyes when catalyst was immobilized in alginate gel beads was also observed by Iglesias19 and Rosales33 due to the improvement on the catalyst efficiency when it is entrapped into the hydrogel structure. They determined that iron can be effectively entrapped in a biopolymer matrix such as alginate without significant reduction in its reactivity. The porosity of alginate beads allows solutes to diffuse into the beads and come in contact with the entrapped material. In addition, another advantage of a supported catalyst system is the easy separation from the reaction solution by filtration, favoring the operation in continuous mode. Furthermore, the leaching of AMDS and metals was negligible at both tested pH values, therefore iron was kept entrapped on the hydrogel structure.
As EF-AMDS-AB at pH 2 reached higher decoloration, this condition was selected to evaluate the reusability of the new heterogeneous catalyst in several cycles. Fig. 5 shows the ability of AMDS-AB to be used in successive batches without losing efficiency nor leaching metals.
3.5. Degradation of pirimicarb
Pesticides are only registered for use if they are verified not to persist in the environment significantly beyond their intentional period of use; however, their degradation products are found ubiquitously in the natural environment in ng L−1 to low μg L−1 concentrations.43 Therefore, pesticide pirimicarb, a highly soluble and representative carbamate was selected in order to evaluate the effectiveness of EF-AMDS (Fig. 6). | ||
| Fig. 6 Electro-Fenton-like degradation profile of pirimicarb with AMDS-AB (■) at pH 2 and AMDS (●) at pH 6. | ||
The treatment of pesticide at high concentration, 100 mg L−1, increases the organic load compared with the dye assays (10 mg L−1) and therefore the treatment time required to attain a high degradation was 180 min; though, after 60 min there was an 80% of reduction. Chen44 reached the total degradation of pirimicarb under UV radiation, nonetheless the initial concentration was 10 times lower.
In addition, the EF-AMDS was also tested at pH 6 in order to avoid leaching (Fig. 6). The kinetic coefficient for the first order reaction was 0.014 min−1 for the EF-AMDS and 0.021 min−1 for the EF-AMDS-AB; this indicates the more appropriate performance of AMDS-AB compared with free AMDS. Fig. 6 shows the two first cycles of EF-AMDS and EF-AMDS-AB of pirimicarb. A good pesticide reduction in both cases is observed, which was repeated for four cycles with a metal leaching lower than 2% in both cases. However AMDS-AB is a more suitable candidate for continuous treatment due to the uncomplicated handling of alginate gel beads as was evidenced by Iglesias34 where the continuous electro-Fenton treatment of pesticide imidacloprid with iron alginate gel beads was efficiently carried out.
AMDS is known for its adsorption capacity of different pollutants such as phosphate13 or arsenic.45,46 To confirm that the electro-Fenton-like with AMDS and AMDS-AB is responsible of pesticide reduction on solution, adsorption assays were done in the same experimental conditions but without electric current. Results after 180 min of AMDS and AMDS-AB contact with pirimicarb showed an adsorption degree lower than 10% of initial pesticide on AMDS and AMDS-AB. These data evidence the catalytic capacity of AMDS and AMDS-AB on the Fenton-like reactions due its high metal content and adsorption process is not responsible of the removal of pesticide in the medium. The energy consumptions were similar with values of 28.2 kW h kg−1pesticide for the EF-AMDS and 29.0 kW h kg−1pesticide for the EF-AMDS-AB. These energy consumptions can be reduced by optimizing parameters such as voltage or testing other electrode materials in order to reduce the economic costs while keeping the efficiency of the process.19
3.6. Pirimicarb degradation pathway
The EF-AMDS-AB of pirimicarb generates several intermediate compounds; their identification was based on their mass fragmentation values (Table 3) and by comparing the mass spectra to a database.The degradation of pirimicarb leads to two main types of compounds: carbamate containing products compounds: compounds (II), (III) and (IV) from Table 3; and carbamate hydrolysis products: compounds (V) and (VI) from Table 3.
Fig. 7 shows a proposed pathway for the degradation of pirimicarb under the EF-AMDS-AB based on the degradation products and bibliography.44,47,48 Compound II is due to the formation of the carbonyl group on the N,N-dimethyl group. Compound III is formed by N-dealkylation of one methyl adjacent to the N atom connected to the pyrimidine ring. Both molecules are two of the main products in photodegradation.44,49 Compound III was also found by Keserü50 on the cytochrome P450 catalysed pesticide metabolism. Compound III can further decompose by N-dealkylation of the methyl group left on the N atom connected to the pyrimidine ring and generate compound IV, also found in photodegradation process on liquid phase.49 The hydrolysis of carbamate group from compound III produces compound VI that is one of the major metabolites in human urine in addition to compound V, which comes from the hydrolylis of carbamate from compound I.51–53 The carbamate hydrolysis products (V and VI) and the carbamate-containing products (II, III, IV) were found by Fenoll47 on the photocatalytic oxidation in aqueous slurries.
 | ||
| Fig. 7 Proposed pathways for the oxidation of pirimicarb by hydroxyl radicals by EF-AMDS-AB. Dotted line indicates hydrolysis after very long time. | ||
All identified compounds were found after 90 min of treatment time; after 5 h of treatment just compound I, II and V were present on the solution. The signals of II and V increase during the pirimicarb decomposition, as was also identified by Pirisi.49 These authors also found that compound III can come from compound II only by hydrolysis after very long times and that compounds II and V are more stable than III; this may explain that compound III and its products were only found at short treatment time. After 15 h pirimicarb disappeared and some unknown compounds were found. Finally, after 24 h of EF-AMDS-AB treatment only some unidentified product was found in very small concentration. Therefore, the EF-AMDS-AB is capable to destruct the pirimicarb molecule and the products that come from its decomposition until a near mineralization.
4. Conclusions
A systematic study on the efficiency of the industrial waste AMDS as catalyst on the electro-Fenton-like treatment of organic pollutants was carried out. Initially, it was evidenced from the composition and properties of the AMDS that this waste was a suitable candidate to be used as catalyst in the heterogeneous electro-Fenton-like for the treatment of several pollutants such as dye Lissamine Green B and pesticide pirimicarb. AMDS dosage did not have a significant effect while acidic pH conditions favoured the removal of dye as expected on the electro-Fenton treatment. Nonetheless, metal leaching at low pH values required the entrapment of AMDS on alginate gel beads, which proved a high performance and stability in several batches. Then, the treatment of pesticide pirimicarb confirmed the successful results attained on the decoloration of the dye. The pesticide adsorption assays on AMDS and AMDS-AB further corroborate that the EF-AMDS-AB is responsible of the depletion of pollutants from solution. In addition the identification of a plausible degradation pathway of pirimicarb confirmed its decomposition.On the other hand, the obtained results pointed out the need of additional evaluations of other important parameters to reduce the energy costs while keeping the efficacy of the process.
Acknowledgements
This research has been financially supported by the Spanish Ministry of Economy and Competitiveness, Xunta de Galicia and by ERDF Funds (Projects CTM2011-26423 and GRC 2013/003). The authors are grateful to the Spanish Ministry of Economy and Competitiveness for financial support of the researcher Marta Pazos under a Ramón y Cajal program.References
- M. Faouzi, P. Cañizares, A. Gadri, J. Lobato, B. Nasr, R. Paz, M. A. Rodrigo and C. Saez, Electrochim. Acta, 2006, 52, 325–331 CrossRef CAS PubMed.
- M. Pera-Titus, V. García-Molina, M. A. Baños, J. Giménez and S. Esplugas, Appl. Catal., B, 2004, 47, 219–256 CrossRef CAS PubMed.
- C. Lopez-Lopez, J. Martín-Pascual, M. V. Martínez-Toledo, J. González-López, E. Hontoria and J. M. Poyatos, Water, Air, Soil Pollut., 2013, 224 Search PubMed.
- A. Arslan, S. Veli and D. Bingöl, Sep. Purif. Technol., 2014, 132, 561–567 CrossRef CAS PubMed.
- S. Sarkar, S. Ali, L. Rehmann, G. Nakhla and M. B. Ray, J. Hazard. Mater., 2014, 278, 16–24 CrossRef CAS PubMed.
- J. P. Guin, D. B. Naik, Y. K. Bhardwaj and L. Varshney, RSC Adv., 2014, 4, 39941–39947 RSC.
- A. R. F. Pipi, A. R. De Andrade, E. Brillas and I. Sirés, Sep. Purif. Technol., 2014, 132, 674–683 CrossRef CAS PubMed.
- A. D. Bokare and W. Choi, J. Hazard. Mater., 2014, 275, 121–135 CrossRef CAS PubMed.
- A. Nair, A. A. Juwarkar and S. Devotta, J. Hazard. Mater., 2008, 152, 545–553 CrossRef CAS PubMed.
- B. Gazea, K. Adam and A. Kontopoulos, Miner. Eng., 1996, 9, 23–42 CrossRef CAS.
- D. B. Johnson and K. B. Hallberg, Sci. Total Environ., 2005, 338, 3–14 CrossRef CAS PubMed.
- A. Akcil and S. Koldas, J. Cleaner Prod., 2006, 14, 1139–1145 CrossRef PubMed.
- Y. R. Wang, D. C. W. Tsang, W. E. Olds and P. A. Weber, Environ. Technol., 2013, 34, 3177–3182 CrossRef CAS PubMed.
- R. G. Flores, S. L. F. Andersen, L. K. K. Maia, H. J. José and R. D. F. P. M. Moreira, J. Environ. Manage., 2012, 111, 53–60 CrossRef CAS PubMed.
- O. Iglesias, M. A. Fernández de Dios, M. Pazos and M. A. Sanromán, Environ. Sci. Pollut. Res., 2013, 20, 5983–5993 CrossRef CAS PubMed.
- M. Á. Fernández de Dios, O. Iglesias, M. Pazos and M. Á. Sanromán, Sci. World J., 2014, 2014, DOI:10.1155/2014/801870.
- L. Y. Taketa, F. Ignachewski, J. C. Villalba, F. J. Anaissi and S. T. Fujiwara, Environ. Sci. Pollut. Res. Int., 2014 DOI:10.1007/s11356-014-3239-3.
- E. Bocos, M. Pazos and M. A. Sanromán, J. Chem. Technol. Biotechnol., 2014, 89, 1235–1242 CrossRef CAS.
- O. Iglesias, M. A. Fernández de Dios, E. Rosales, M. Pazos and M. A. Sanromán, Environ. Sci. Pollut. Res., 2013, 20, 2172–2183 CrossRef CAS PubMed.
- M. A. Fernández de Dios, E. Rosales, M. Fernández-Fernández, M. Pazos and M. Ángeles Sanromán, J. Chem. Technol. Biotechnol., 2014 DOI:10.1002/jctb.4446.
- V. Cleveland, J. Bingham and E. Kan, Sep. Purif. Technol., 2014, 133, 388–395 CrossRef CAS PubMed.
- L. Wang, Y. Yao, Z. Zhang, L. Sun, W. Lu, W. Chen and H. Chen, Chem. Eng. J., 2014, 251, 348–354 CrossRef CAS PubMed.
- Z. Huang, Y. Ye, S. Zhu, Y. Yao, W. Lu and W. Chen, J. Chem. Technol. Biotechnol., 2014 DOI:10.1002/jctb.4504.
- J. Shen, J. Zhu, Y. Kong, T. Li and Z. Chen, Water Sci. Technol., 2013, 68, 1614–1621 CrossRef CAS PubMed.
- H. Wu, X. Dou, D. Deng, Y. Guan, L. Zhang and G. He, Environ. Technol., 2012, 33, 1545–1552 CrossRef CAS PubMed.
- X. Zhang, Y. Chen, N. Zhao, H. Liu and Y. Wei, RSC Adv., 2014, 4, 21575–21583 RSC.
- G. Zhang, S. Wang and F. Yang, J. Phys. Chem. C, 2012, 116, 3623–3634 CAS.
- C. C. Amorim, M. M. D. Leão, R. F. P. M. Moreira, J. D. Fabris and A. B. Henriques, Chem. Eng. J., 2013, 224, 59–66 CrossRef CAS PubMed.
- Y. Zhong, X. Liang, Z. He, W. Tan, H. He, R. Zhu, Y. Zhong, J. Zhu, P. Yuan and Z. Jiang, J. Nanosci. Nanotechnol., 2014, 14, 7307–7314 CrossRef CAS PubMed.
- P. V. Nidheesh, R. Gandhimathi, S. Velmathi and N. S. Sanjini, RSC Adv., 2014, 4, 5698–5708 RSC.
- R. C. Martins, L. R. Henriques and R. M. Quinta-Ferreira, Chem. Eng. Sci., 2013, 100, 225–233 CrossRef CAS PubMed.
- C. Oliveira, M. S. F. Santos, F. J. Maldonado-Hódar, G. Schaule, A. Alves and L. M. Madeira, Chem. Eng. J., 2012, 210, 339–349 CrossRef CAS PubMed.
- E. Rosales, O. Iglesias, M. Pazos and M. A. Sanromán, J. Hazard. Mater., 2012, 213–214, 369–377 CrossRef CAS PubMed.
- O. Iglesias, J. Gómez, M. Pazos and M. A. Sanromán, Appl. Catal., B, 2013, 144, 416–424 CrossRef PubMed.
- M. A. Oturan, J. Appl. Electrochem., 2000, 30, 475–482 CrossRef CAS.
- B. Li and J. Zhu, Chem. Eng. J., 2014, 255, 225–232 CrossRef CAS PubMed.
- H. Cheng, S. Chou, S. Chen and C. Yu, J. Environ. Sci., 2014, 26, 1307–1312 CrossRef CAS.
- M. Umar, H. A. Aziz and M. S. Yusoff, Waste Manage., 2010, 30, 2113–2121 CrossRef CAS PubMed.
- A. Y. Mounia and Z. Djilali, Desalin. Water Treat., 2014 DOI:10.1080/19443994.2014.954144.
- M. Hartmann, S. Kullmann and H. Keller, J. Mater. Chem., 2010, 20, 9002–9017 RSC.
- X. Liu, R. Tang, Q. He, X. Liao and B. Shi, J. Hazard. Mater., 2010, 174, 687–693 CrossRef CAS PubMed.
- W. Wang, Y. Liu, T. Li and M. Zhou, Chem. Eng. J., 2014, 242, 1–9 CrossRef CAS PubMed.
- K. Fenner, S. Canonica, L. P. Wackett and M. Elsner, Science, 2013, 341, 752–758 CrossRef CAS PubMed.
- T. Chen, F. Fu, Z. Chen, D. Li, L. Zhang and G. Chen, J. Chromatogr. A, 2009, 1216, 3217–3222 CrossRef CAS PubMed.
- J. Yang, Y. Kim, S. Park and K. Baek, Environ. Sci. Pollut. Res., 2014, 21, 10878–10889 CrossRef CAS PubMed.
- M. Ko, J. Kim, J. Lee, J. Ko and K. Kim, Appl. Geochem., 2013, 35, 1–6 CrossRef CAS PubMed.
- J. Fenoll, I. Garrido, P. Hellín, P. Flores, N. Vela and S. Navarro, J. Photochem. Photobiol., A, 2015, 298, 24–32 CrossRef CAS PubMed.
- C. Coscollà, E. Hart, A. Pastor and V. Yusà, Atmos. Environ., 2013, 77, 394–403 CrossRef PubMed.
- F. M. Pirisi, P. Cabras, V. L. Garau, M. Melis and E. Secchi, J. Agric. Food Chem., 1996, 44, 2417–2422 CrossRef CAS.
- G. M. Keserü, G. Balogh, I. Czudor, T. Karancsi, A. Fehér and B. Bertók, J. Agric. Food Chem., 1999, 47, 762–769 CrossRef PubMed.
- C. Sams, K. Patel and K. Jones, Toxicol. Lett., 2010, 192, 56–60 CrossRef CAS PubMed.
- C. Sams and K. Jones, Toxicol. Lett., 2011, 200, 41–45 CrossRef CAS PubMed.
- K. Jones, M. Everard and A. Harding, Int. J. Hyg. Environ. Health, 2014, 217, 392–398 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |