Palm oil and cardiovascular disease: a randomized trial of the effects of hybrid palm oil supplementation on human plasma lipid patterns†
P.
Lucci
*a,
M.
Borrero
b,
A.
Ruiz
c,
D.
Pacetti
d,
N. G.
Frega
d,
O.
Diez
e,
M.
Ojeda
b,
R.
Gagliardi
 d,
L.
Parra
b and
M.
Angel
b
d,
L.
Parra
b and
M.
Angel
b
aDepartment of Food Science, University of Udine Via Sondrio 2/a, 33100 Udine, Italy. E-mail: paololucci2001@yahoo.it
bDepartment of Nutrition and Biochemistry, Faculty of Sciences, Pontificia Universidad Javeriana, KR 7 # 40-62, Bogota D.C., Colombia
cDepartment of Internal Medicine, School of Medicine, Pontificia Universidad Javeriana, KR 7 # 40-62, Bogota D.C., Colombia
dDepartment of Agricultural, Food, and Environmental Sciences, Università Politecnica delle Marche, Via Brecce Bianche, 60131, Ancona, Italy
eDepartment of Microbiology, Faculty of Sciences, Pontificia Universidad Javeriana, KR 7 # 40-62, Bogota D.C., Colombia
First published on 8th October 2015
Abstract
This study examines, for the first time, the effect of hybrid Elaeis oleifera × E. guineensis palm oil supplementation on human plasma lipids related to CVD risk factors. One hundred sixty eligible participants were randomized and assigned to one of the two treatments: 25 mL hybrid palm oil (HPO group) or 25 mL extra virgin olive oil (EVOO group) daily for 3 months. Fasting venous samples were obtained at baseline and after 1, 2 and 3 months for measurement of plasma lipids (TC, LDL-C, HDL-C and TAGs). Changes in body mass index and waist circumference were also assessed. Although there was an overall reduction in TC (7.4%, p < 0.001) and in LDL-C (15.6%, p < 0.001), no significant differences were found between the treatment groups in a repeated measures analysis of variance for TC (p = 0.0525), LDL-C (p = 0.2356), HDL-C (p = 0.8293) or TAGs (p = 0.3749). Furthermore, HPO consumption had similar effects on plasma lipids to EVOO, thus providing additional support for the concept that hybrid Elaeis oleifera × E. guineensis palm oil can be seen as a “tropical equivalent of olive oil”.
Introduction
Cardiovascular disease is the main cause of death worldwide at the turn of the XXI century. Western countries continue to exhibit high rates of cardiovascular morbidity and mortality, whilst these diseases constitute emerging and neglected epidemics in developing countries.1,2 With regard to the quality of dietary fat in relation to cardiovascular disease (CVD), a large number of studies have demonstrated the beneficial role of diets with a relatively high monounsaturated fatty acid (MUFA) content on CVD risk factors, obesity and diabetes. These beneficial MUFA are mainly provided by the Mediterranean diet and, specifically, by olive oil.3–5 On the other hand, diets rich in saturated fatty acids (SFA) have long been associated with increased plasma cholesterol concentrations and hence with an increased risk of CVD. In 2003, the World Health Organization stated that there is convincing evidence that consumption of palmitic acid-rich oils contributes to an increased risk of developing CVD. Additional evidence suggests that a dietary intervention that reduces SFA intake and increases consumptions of vegetables, fruits and grains may reduce the development of diseases such as cancer and atherosclerosis.6,7 In this context, palm oil (PO), which is mainly extracted from the fruit of the African oil palm (Elaeis guineensis Jacq.), has so far been strongly attacked due to its high content of SFA, specifically palmitic acid (C16:0, ∼44.3%), and its use in food products has been somewhat discouraged. However, in recent years there has been a lot of debate regarding the potential role of SFA in the pathogenesis of atherosclerosis and CVD and a more complex picture concerning the risk factors for CVD has now been developed. In fact, current evidence does not clearly support cardiovascular guidelines that encourage a high consumption of polyunsaturated fatty acids (PUFA) and a low consumption of total saturated fats.8 For instance, Mensink et al.9 have shown that myristic and palmitic acids had little effect on the ratio of the total to HDL cholesterol, and stearic acid reduced the ratio slightly. More recently, a meta-analysis also showed that PO may produce both favorable and unfavorable changes compared with the other primary dietary SFA, MUFA and PUFA, and almost no changes were observed in total cholesterol/HDL cholesterol and LDL cholesterol/HDL cholesterol ratios.10To date in Colombia more than 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hectares of oil palm interspecific hybrid Elaeis oleifera × E. guineensis has been planted. From an agronomic point of view, the good bud rot tolerance of the hybrids, compared to E. guineensis, justifies its use to replant severely decimated zones. However, the oil obtained from hybrid palm fruits may also have unexplored functional properties and health benefits, especially in reducing cardiovascular disease risk. In fact, in our recent study we fully characterized, for the first time, the composition and structure of triacylglycerols (TAGs) and partial glycerides of crude palm oil obtained from interspecific hybrid E. oleifera × E. guineensis grown in Colombia.11 The study revealed interesting nutritional properties of oils obtained from interspecific hybrid palm compared to the oils from African palm oil. Although only four fatty acid types (palmitic, C16:0; stearic C18:0; oleic C18:1; linolenic, C18:2) constituted more than 95% of the total fatty acids of both oils, the hybrid palm oil had a higher percentage of oleic acid (54.6 ± 1.0 vs. 41.4 ± 0.3) together with lower palmitic (28.3 ± 1.0 vs. 40.1 ± 0.1) and stearic (2.8 ± 0.3 vs. 5.0 ± 0.1) acids amounts. The percentage of the essential fatty acid, linoleic acid, does not differ significantly. Furthermore, the sn-2 position of TAGs in hybrid palm oil (HPO) was shown to be predominantly esterified with oleic acid with only 10–15% of the total palmitic acid and 6–20% of stearic acid acylated in the secondary position. These findings are very interesting, especially from a nutritional point of view since fatty acids in the sn-2 position of dietary triacylglycerols are preferentially absorbed through the intestinal wall. Therefore, our results demonstrate that a partial dietary replacement of saturated palmitic acid with monounsaturated oleic acid can be obtained by consuming crude HPO instead of African palm oil, without the need for any fractionation process to separate olein and stearin fractions because of HPO higher degrees of unsaturation. This also means that crude HPO could represent an extremely rich source of health-promoting minor components, since such bioactive compounds are not lost during the refining and fractionation processes required for African PO. Therefore, besides the interesting composition and structure of TAGs, HPO can also provide a substantial amount of antioxidants that might additionally contribute to lower the risk of developing CHD and certain cancers.12 In fact, our latest studies13,14 revealed that HPO unsaponifiable matter is characterized by a high content of tocotrienols (4.5 ± 1.4 mg of α-tocotrienol, 0.4 ± 0.1 β-tocotrienol, 14.8 ± 2.3 mg of γ-tocotrienol, 3.2 ± 0.4 mg of δ-tocotrienol per 100 g oil) with tocopherols, mainly entirely constituted of the α isomer, that accounted for 2.7 mg ± 0.7 per 100 g oil. These results highlight another interesting peculiarity of HPO, since tocotrienols are now well recognized for their superior antioxidant activity compared with tocopherols.
000 hectares of oil palm interspecific hybrid Elaeis oleifera × E. guineensis has been planted. From an agronomic point of view, the good bud rot tolerance of the hybrids, compared to E. guineensis, justifies its use to replant severely decimated zones. However, the oil obtained from hybrid palm fruits may also have unexplored functional properties and health benefits, especially in reducing cardiovascular disease risk. In fact, in our recent study we fully characterized, for the first time, the composition and structure of triacylglycerols (TAGs) and partial glycerides of crude palm oil obtained from interspecific hybrid E. oleifera × E. guineensis grown in Colombia.11 The study revealed interesting nutritional properties of oils obtained from interspecific hybrid palm compared to the oils from African palm oil. Although only four fatty acid types (palmitic, C16:0; stearic C18:0; oleic C18:1; linolenic, C18:2) constituted more than 95% of the total fatty acids of both oils, the hybrid palm oil had a higher percentage of oleic acid (54.6 ± 1.0 vs. 41.4 ± 0.3) together with lower palmitic (28.3 ± 1.0 vs. 40.1 ± 0.1) and stearic (2.8 ± 0.3 vs. 5.0 ± 0.1) acids amounts. The percentage of the essential fatty acid, linoleic acid, does not differ significantly. Furthermore, the sn-2 position of TAGs in hybrid palm oil (HPO) was shown to be predominantly esterified with oleic acid with only 10–15% of the total palmitic acid and 6–20% of stearic acid acylated in the secondary position. These findings are very interesting, especially from a nutritional point of view since fatty acids in the sn-2 position of dietary triacylglycerols are preferentially absorbed through the intestinal wall. Therefore, our results demonstrate that a partial dietary replacement of saturated palmitic acid with monounsaturated oleic acid can be obtained by consuming crude HPO instead of African palm oil, without the need for any fractionation process to separate olein and stearin fractions because of HPO higher degrees of unsaturation. This also means that crude HPO could represent an extremely rich source of health-promoting minor components, since such bioactive compounds are not lost during the refining and fractionation processes required for African PO. Therefore, besides the interesting composition and structure of TAGs, HPO can also provide a substantial amount of antioxidants that might additionally contribute to lower the risk of developing CHD and certain cancers.12 In fact, our latest studies13,14 revealed that HPO unsaponifiable matter is characterized by a high content of tocotrienols (4.5 ± 1.4 mg of α-tocotrienol, 0.4 ± 0.1 β-tocotrienol, 14.8 ± 2.3 mg of γ-tocotrienol, 3.2 ± 0.4 mg of δ-tocotrienol per 100 g oil) with tocopherols, mainly entirely constituted of the α isomer, that accounted for 2.7 mg ± 0.7 per 100 g oil. These results highlight another interesting peculiarity of HPO, since tocotrienols are now well recognized for their superior antioxidant activity compared with tocopherols.
The aim of the present paper was to investigate, for the first time, the health benefits of hybrid palm oil consumption and its potential as a functional food oil for the prevention of CVD. In a randomized trial we compared the effects of supplementation with crude hybrid palm oil (E. oleifera × E. guineensis) and extra-virgin olive oil, which is universally recognized as putative anti-atherogenic and cardioprotective oil, on human plasma lipids related to cardiovascular disease risk.
Materials and methods
Clinical trial design
Laboratory methods
The oil unsaponifiable matter was analysed according to the procedure described by Lucci et al.14
The extraction of the phenolic fraction from the oils was performed as reported by Montedoro et al.17 whereas the total phenolic content of the oils was determined according to the procedure reported by Loizzo et al.18 The results were expressed as gallic acid equivalents (mg per kg oil) based on the calibration curve (R2 = 0.996). The estimation of the total phenolic content was carried out in triplicate and the results were averaged.
The total fatty acid profile, the unsaponifiable matter composition and the total phenolic content of the oils are shown in Table 1. It is noteworthy that the amount of antioxidant compounds provided by HPO supplementation was similar to that provided by EVOO supplementation. In fact, the dose of EVOO (25 ml per day) corresponded to a daily intake of tocols and phenols of 4.5 and 38.5 mg per day, respectively. Similarly, the dose of HPO (25 ml per day) corresponded to a daily intake of tocols and phenols of 6.5 and 47.5 mg per day, respectively. Once again, it is important to mention that in HPO tocotrienols constitute ∼90% of the total tocols.
| EVOO | HPO | |
|---|---|---|
| SFA, saturated fatty acids; MUFA, monounsaturated fatty acids; PUFA, polyunsaturated fatty acids; legend for fatty acids – m:n Δx, m = number of carbon atoms, n = number of double bonds, x = position of double bonds.a Tocols = sum of α-tocopherol, α-tocotrienol, β-tocotrienol, γ-tocotrienol and δ-tocotrienol contents. | ||
| Fatty acids (%) | ||
| C16:0 | 12.5 ± 0.8 | 27.9 ± 1.2 |
| C16:1 | 0.9 ± 0.1 | 0.5 ± 0.1 |
| C18:0 | 2.5 ± 0.4 | 2.9 ± 0.3 |
| C18:1Δ9c | 76.0 ± 1.2 | 55.2 ± 1.3 |
| C18:2Δ9c,12c | 5.5 ± 0.8 | 10.8 ± 0.5 |
| C18:3Δ9c,12c,15c | 0.6 ± 0.1 | 0.4 ± 0.0 |
| C20:0 | 0.4 ± 0.2 | 0.3 ± 0.1 |
| C20:1Δ11 | 0.3 ± 0.1 | 0.4 ± 0.2 |
| ∑SFA | 15.4 ± 0.8 | 30.8 ± 1.6 |
| ∑MUFA | 77.2 ± 1.2 | 56.1 ± 1.0 |
| ∑PUFA | 6.1 ± 0.4 | 11.1 ± 0.7 |
| Unsaponifiable components (mg per 100 g oil) | ||
| Squalene | 321± 8.6 | 24.7 ± 0.3 |
| Campesterol | 1.1 ± 0.4 | 9.8 ± 0.3 |
| Stigmasterol | 2.9 ± 0.1 | 8.2 ± 1.0 |
| β-Sitosterol | 127 ± 11.4 | 39.1 ± 3.3 |
| Δ5-Avenasterol | 21.3 ± 2.8 | 1.7 ± 0.2 |
| n-Alkanols | 13.2 ± 1.4 | 6.2 ± 1.7 |
| 4-Methylsterols | 20.7 ± 2.1 | 1.3 ± 0.2 |
| 4,4-Dimethylsterols | 135.6 ± 9.5 | 7.4 ± 1.2 |
| Tocolsa | 18.1 ± 1.5 | 25.9 ± 4.8 |
| Total phenolic content (ppm) | ||
| 154 ± 4.6 | 190 ± 2.5 | |
Statistical analyses
Comparisons between the two groups were evaluated using Student's t-test for paired samples. A p value of less than 0.05 (2-sided) was adopted to determine significant differences. For the differences between treatments, taking into account the visits, repeated measures ANOVA was performed, with the baseline values as covariates. Interactions were considered between treatment and visits, and sex, age and BMI were also explored for effects. A p value <0.05 was considered significant. The statistical analyses were carried out with STATA 13.0 software (StataCorp LP).Results
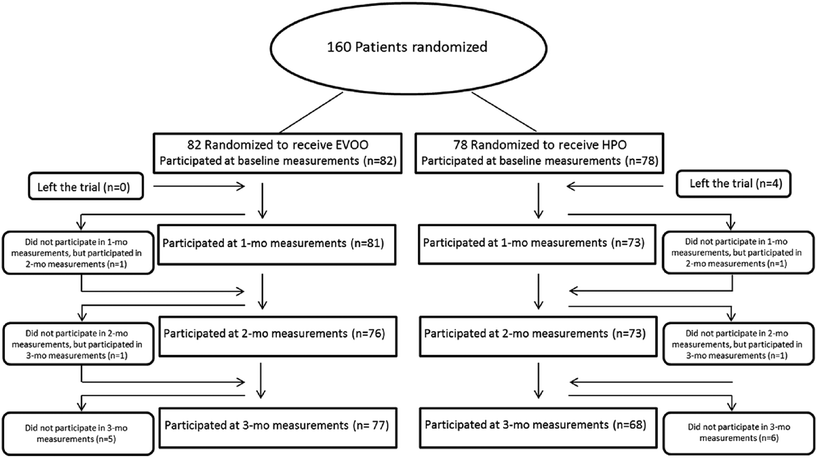
The subjects’ baseline characteristics are presented in Table 2. At baseline, there was no significant difference (p < 0.05) between the group randomized to HPO or EVOO for any of the plasma measurements (TC, HDL-C, HDL-C, TAGs), body mass index (BMI) and waist perimeter. The mean ages of the HPO subjects and EVOO subjects were 64.3 ± 8.5 and 62.8 ± 6.1 years, respectively. The mean BMI in the study population was 28.3 ± 3.8 with a waist circumference of 87.9 ± 9.1. Analysis of the participants’ lipid profile at the start of the trial (baseline) indicated that subjects had normal to high LDL-C, TC and TAG values. The mean TC values of the HPO subjects and EVOO subjects were 206.5 ± 39.1 and 203.8 ± 36.9 mg dL−1, respectively, whereas average population levels of 120.4 ± 38.2 mg dL−1 for LDL-C and of 44.9 ± 12.4 mg dL−1 for HDL-C were found. Overall, more than 35% of the population had hypercholesterolemia (LDL-C >130 mg dL−1) and more than 43% showed high levels of triglycerides (TAGs >200 mg dL−1). Data from 24-hour recall and food frequency questionnaires showed no significant difference in the baseline and follow-up dietary intake of total energy, protein, total fat, carbohydrate, and alcohol between the two study groups (ESI† 1). 11/160 (6.8%) patients did not complete the trial (Fig. 1). Four subjects (2.5%) dropped out during the first month (HPO group) as they were unwilling to continue to participate in the study. In each group, one subject did not attend the visit planned for the first or second assessment (1-month and 2-month measurements). However, these participants were included in the analyses as they continued to participate in the assessments at 2-months and 3-months. The dropout rates observed after three months of oil supplementation (3-mo measurements) were 6.1% (n = 5) and 7.7% (n = 6) for EVOO and HPO groups, respectively. We found no evidence of any adverse effects related to the oils.| Variable | Baseline population | EVOO (n = 82) | HPO (n = 78) | EVOO vs. HPO (p value) |
|---|---|---|---|---|
| Sample sizes in parenthesis, S.D. = standard deviation. EVOO = extra-virgin olive oil; HPO = hybrid palm oil; Significance (P < 0.05). | ||||
| Age in years – mean (S.D.) | 63.5 (7.2) | 62.8 (6.1) | 64.3 (8.5) | 0.3591 |
| Gender (% male) | 13 (8.1%) | 6 (7.4%) | 7 (8.9%) | 0.0710 |
| Body mass index – mean (S.D.) | 28.3 (3.8) | 28.1 (3.7) | 28.5 (3.8) | 0.4148 |
| Waist perimeter – mean (S.D.) | 87.9 (9.1) | 87.2 (8.8) | 88.6 (9.4) | 0.3405 |
| Hypercholesterolemia (%) | 56 (35.2%) | 26 (32.1%) | 30 (38.4%) | 0.3738 |
| Hypertriglyceridemia (%) | 70 (43.8%) | 43 (53.0%) | 27 (34.6%) | 0.5802 |
| Cholesterol Total – mean (S.D.) | 205.1 (37.9) | 203.8 (36.9) | 206.5 (39.1) | 0.6429 |
| Cholesterol, HDL – mean (S.D.) | 44.9 (12.4) | 43.5 (10.4) | 46.3 (14.2) | 0.1689 |
| Cholesterol, LDL – mean (S.D.) | 120.4 (38.2) | 116.9 (38.1) | 124.0 (38.1) | 0.2464 |
| Triglycerides – mean (S.D.) | 218.4 (128) | 242.0 (155.6) | 194.9 (85.7) | 0.0698 |
As we can see from Table 3, there were final differences for all fractions, although significant only in the univariate analysis (considering only the treatment effect and not the interactions) for TC and for LDL-C levels (baseline vs. Visit 3).
| Variable | Baselinea | Visit 1 | Visit 2 | Visit 3 | Baseline vs. Visit 3 (p valuec) | p valued | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| EVOO (n = 82) | HPO (n = 78) | EVOO (n = 81) | HPO (n = 73) | EVOO (n = 76) | HPO (n = 73) | EVOO (n = 77) | HPO (n = 68) | EVOO | HPO | ||
| Sample sizes in parenthesis; data are expressed as mean (standard deviation). EVOO = extra-virgin olive oil; HPO = hybrid palm oil; NA = not assessed.a Number of participants who were receiving stable doses of statin across the study: EVOO (n = 37), HPO (n = 39).b Number of participants for whom LDL-C levels were measured by enzymatic colorimetric methods because of TAG levels higher than 400 mg/dL: baseline [EVOO (n = 14), HPO (n = 7)]; Visit 1 [EVOO (n = 15), HPO (n = 7)]; Visit 2 [EVOO (n = 14), HPO (n = 6)]; Visit 3 [EVOO (n = 13), HPO (n = 7)].c p value for univariate analysis [considering only the treatment effect and not interactions; significance (p < 0.05) = ○; significance (p < 0.01) = ●].d p value for multivariate analysis [considering interactions among visits and treatment; significance (p < 0.05)]. | |||||||||||
| Body mass index | 28.1 (3.7) | 28.5 (3.8) | NA | NA | NA | NA | 27.7 (4.1) | 28.3 (3.3) | — | — | 0.2185 |
| Cholesterol Total | 203.8 (36.9) | 206.5 (39.1) | 217.2 (35.4) | 213.2 (33.1) | 201.9 (35.3) | 212.5 (34.2) | 185.8 (28.4) | 193.9 (31.5) | ● | ○ | 0.0525 |
| Cholesterol, HDL | 43.5 (10.4) | 46.3 (14.2) | 50.11 (14.1) | 50.3 (16.6) | 44.3 (17.5) | 45.1 (14.4) | 43.2 (16.6) | 44.3 (17.9) | — | — | 0.8293 |
| Cholesterol, LDLb | 116.9 (38.1) | 124.0 (38.1) | 161.1 (38.2) | 161.2 (34.7) | 119.4 (36.6) | 132.3 (36.1) | 95.7 (33.8) | 107.2 (36.4) | ● | ○ | 0.2356 |
| Triglycerides | 242.0 (155.6) | 194.9 (85.7) | 222.1 (111.4) | 185.5 (83.3) | 224.4 (110.5) | 200.6 (87.8) | 245.4 (122.9) | 210.8 (136.5) | — | — | 0.3749 |
| Total:HDL cholesterol | 4.97 (1.89) | 4.71 (1.20) | 4.61 (1.31) | 4.61 (1.56) | 5.09 (1.75) | 5.22 (1.99) | 5.34 (2.98) | 5.36 (2.48) | — | — | 0.4031 |
| LDL:HDL cholesterol | 2.91 (1.63) | 2.84 (1.06) | 3.48 (1.37) | 3.58 (1.59) | 3.58 (1.59) | 3.11 (1.53) | 2.93 (2.22) | 3.18 (2.87) | — | — | 0.5344 |
In general, there was an initial rise, for both treatments, in TC, LDL-C and HDL-C levels, followed by reduction for both groups (Fig. 2). These results are confirmed by the changes observed throughout the experiment in the overall population (Table 4). A possible explanation for this finding is that in the case of insulin and leptin resistance, monounsaturated oil supplementation increases the liver fat content, which may cause an increased release of lipids from the liver to prevent fatty liver development.19 As a result, a temporary increase in serum lipids could be observed.
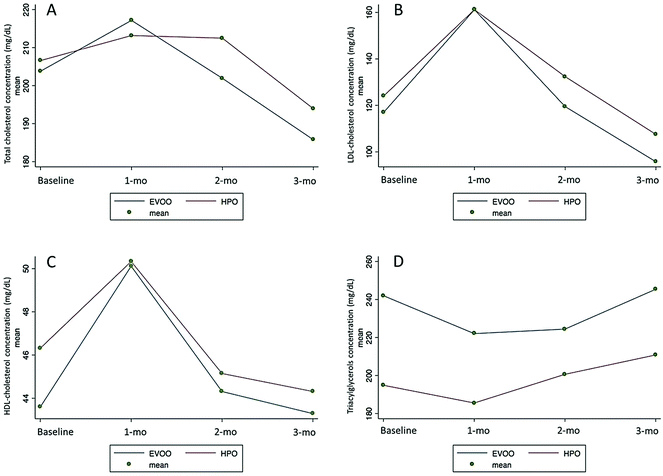 | ||
| Fig. 2 Changes in plasma lipid levels in both groups throughout the experiment. (A) TC levels; (B) LDL-C levels; (C) HDL-C levels; (D) TAG levels. | ||
| TC | LDL-C | HDL-C | TAGs | |||||
|---|---|---|---|---|---|---|---|---|
| Change (mg dL−1) | 95% Conf. Int. | Change (mg dL−1) | 95% Conf. Int. | Change (mg dL−1) | 95% Conf. Int. | Change (mg dL−1) | 95% Conf. Int. | |
| Visit 1 | 9.99 | 3.96 to 16.01 | 40.67 | 33.94 to 47.39 | 5.24 | 2.21 to 8.28 | −14.31 | −29.18 to 0.55 |
| Visit 2 | 1.94 | −4.14 to 8.03 | 5.55 | −1.23 to 12.35 | −0.11 | −3.17 to 2.95 | −7.87 | −22.91 to 7.17 |
| Visit 3 | −15.08 | −21.94 to −8.94 | −18.68 | −25.53 to −11.83 | −1.07 | −4.16 to 2.01 | 9.19 | −5.98 to 24.37 |
In the HPO group, the initial level of TAGs (194.9 ± 85.7 mg dL−1) remained relatively unchanged with a final 3-month concentration of 210.8 ± 136.4 mg dL−1. The same trend has been observed in the EVOO group with TAG values of 242.0 ± 155.6 mg dL−1 and 245.4 ± 122.9 mg dL−1 at baseline and month 3, respectively. In subjects who received HPO, the HDL-C concentration increased by 6.1% at month 1, followed by a decrease gradually leading to the final value (3-month) of 44.3 ± 17.9 mg dL−1 (Table 3; Fig. 2C). A similar pattern was observed in the EVOO group with plasma HDL-C levels of 43.5 ± 10.4 mg dL−1 and 43.2 ± 16.6 mg dL−1, respectively, at the beginning and the end of the study. After 1 month of HPO supplementation, plasma LDL-C concentration was higher than at baseline (124.0 ± 38.1 mg dL−1 compared with 161.2 ± 34.7 mg dL−1). At month 2, however, LDL-C decreased to 132.3 ± 36.1 mg dL−1, and this trend continued over the next month reaching a final 3-month value of 107.2 ± 36.4 mg dL−1 (Fig. 2B). Therefore, during the study period, LDL-C concentration in patients receiving HPO decreased 13.3%. Furthermore, it should be stressed that reduction in LDL-C of HPO subjects was similar to that observed in the patients who received EVOO. In fact, the EVOO group showed a decrease in LDL-C from 116.9 ± 38.1 mg dL−1 at baseline to 95.7 ± 33.8 mg dL−1 at the end of the study. In HPO patients, plasma TC concentrations decreased from baseline (206.5 ± 39.1 mg dL−1) to month 3 (193.9 ± 31.5 mg dL−1), a reduction of 6.3%. Again, as with LDL-C, a small increase of the TC level was observed at both month 1 and month 2: the TC level was 206.5 ± 39.1 mg dL−1 at baseline, increased to 213.2 ± 33.1 mg dL−1 at month 1 and to 212.5 ± 34.2 mg dL−1 at month 2, then decreased to 193.9 ± 31.5 mg dL−1 by month 3 (Fig. 2A). The plasma levels of TC in the EVOO group also showed a reduction over the study period. However, in contrast to the LDL-C results, the reduction in TC was more pronounced in subjects who received EVOO. In fact, plasma levels of TC increased at month 1 (217.2 ± 35.4 mg dL−1) followed by a decline at the second and third assessment times (201.9 ± 35.3 and 185.8 ± 28.4), an overall reduction of approximately 9%. With regard to the TC/HDL-C and LDL-C/HDL-C ratios, used as predictors of ischemic heart disease risk,20 only a small but not significant change has been observed. The TC/HDL-C ratio increased from 4.71 to 5.36 over the study period (from 4.97 to 5.34 in the EVOO group) and a small effect has also been detected for the LDL-C/HDL-C ratio (2.84 baseline vs. 3.18 month 3) (Table 3). In the same way, the triglycerides to HDL-cholesterol ratio (TG/HDL-c), which has been proven to be strongly correlated with the plasma level of small, dense LDL atherogenic particles (phenotype B), showed little increase from 4.20 to 4.75 and from 5.56 to 5.68 for the HPO and EVOO groups, respectively.
No significant differences were observed for BMI between baseline and month 3 in both groups (Table 3). There were significant changes in all fractions when time was considered (across visits) for TC (p < 0.001), LDL-C (p < 0.001), HDL-C (p < 0.0005) and TAGs (p = 0.0191), independent of the treatment received (Table 5).
| TC | LDL-C | HDL-C | TAGs | |
|---|---|---|---|---|
| ANOVA analysis of repeated measures: significance (p < 0.05). | ||||
| Treatment | 0.2898 | 0.0993 | 0.5466 | 0.043 |
| Visit | <0.001 | <0.0001 | <0.0005 | 0.0191 |
| Interaction (between treatment and visit) | 0.0525 | 0.2356 | 0.8293 | 0.3749 |
On the other hand, no statistically significant differences were found between the two groups for TAGs (p = 0.043), TC (p = 0.2898), LDL-C (p = 0.0993) or HDL-C (p = 0.5466). Finally, the repeated measures ANOVA shows that no differences were detected between the two treatment groups for TC (p = 0.0525), LDL-C (p = 0.2356), HDL-C (p = 0.8293) or TAGs (p = 0.3749), once the interaction between treatment and time (visits) was taken into account (Tables 3 and 5).
Discussion
In the present study, we describe for the first time the functional effects of crude hybrid palm oil (E. oleifera × E. guineensis) supplementation on human plasma lipids related to cardiovascular disease risk factors. The results obtained were compared to those achieved from individuals who received an equivalent amount of extra-virgin olive oil. A daily 25 mL dose of hybrid palm oil, which is comparable to the daily consumption of EVOO recommended by the U.S. Food and Drug Administration,21 was shown in our study to have similar effects on plasma lipids to EVOO, which is universally recognized as an (putative) anti-atherogenic and cardioprotective oil.22–24 Several studies have in fact shown an inverse relation between the consumption of oleic acid-rich olive oil and incidence of coronary heart disease. For instance, Nagyova et al.25 published a study focused on the effects of EVOO supplementation on plasma lipids, total antioxidant capacity and indices of serum lipid oxidizability, among others. A total of 26 patients (mean age 69 years) with combined hyperlipidemia consumed approximately 20 g per day of extra virgin olive oil for 6 weeks. Plasma TC and LDL-C decreased significantly after 6 weeks of dietary intervention. These results, together with those from several other studies, are mainly attributed, even not limited, to the ability of oleic acid to increase the resistance of human LDL oxidation which is known to play an important role in the development of atherosclerotic lesions.26–28 In our study, the finding that HPO consumption had a favorable effect on plasma lipids and that this effect was not statistically different from that of EVOO can also be partially attributed to the particular fatty acid composition and triglyceride structure of HPO.11 In fact, we have recently reported that crude hybrid palm oil possesses a high percentage of oleic acid (54.6 ± 1.0) together with a low saturated fatty acid content (33.5 ± 0.5). Another promising outcome was that the sn-2 position of TAGs was predominantly esterified with oleic acid (64.7–66.0 mol%) with only 10–15% of total palmitic acid and 6–20% of stearic acid acylated in the secondary position. These results indicate that the HPO glyceridic composition and structure is quite similar to that of EVOO, even if the latter is characterized by an even higher content of oleic acid as well as by a higher proportion of oleic acid in the position sn-2 of TAGs.29 Nevertheless, our analysis highlighted that HPO should be considered a monounsaturated oleic acid-rich tropical oil rather than a saturated fat, especially from a nutritional point of view. The present study provides further evidence to support this claim. In fact, in our investigation, a daily 25 mL dose of HPO had a partially favorable impact on cardiovascular risk-factor profiles: after three months of HPO consumption, the TC levels decreased by 6.3%, whereas LDL-C concentration decreased by more than 13%.But the observed “positive” effects of HPO consumption may also be associated with the intake of antioxidants contained in crude HPO. As stated before, consistent evidence now suggests that vitamin E, carotenoids and other antioxidant nutrients such as phenolic compounds also offer protection against CVD by decreasing oxidative damage. Thus, nutritional properties of edible oil depend not only on its glyceridic oil composition and structure but also on its potential as a source of health-promoting minor components. In fact, oxidation of LDL-C leads to a change in the lipoprotein conformation by which LDL cholesterol can better enter into the monocyte–macrophage system of the arterial wall and promote the atherosclerotic process. As mentioned previously, no previous studies have been undertaken to determine the health effects of HPO consumption. Studies reported in the literature are almost exclusively focused on refined palm oil fractions and based on the use of animal models. To our knowledge, our study constitutes the first human intervention trial with crude hybrid palm oil. It is certainly true that the effects of refined African palm olein on human plasma lipids and lipoproteins have been relatively well investigated in the past.30–34 However, palm olein, which is the liquid fraction obtained by fractionation of palm oil after crystallization at controlled temperatures, completely loses nutritionally important components such as carotenes, tocopherols and polyphenols. For instance, tocotrienol-enriched fraction from palm oil has been shown to decrease TC and LDL-C plasma levels.35 The favorable effect of crude HPO consumption on plasma lipids can therefore also be attributed to its high content of polyphenols (190 ppm) and tocols (>25 mg per 100 g) (Table 1), as well as by its high carotenoid concentration (>1038 mg per 100 g)13 which may play a crucial role by improving plasma antioxidant defenses and lipid profiles. In fact, because of the greater degree of unsaturation of HPO compared to African palm oil, HPO can be directly consumed without the need for any fractionation or refining process to separate olein and stearin fractions.
Our positive findings of crude HPO on plasma lipids are also consistent with previous animal model studies focused on a different kind of mildly refined palm oil called “red palm olein” (RPO) which suggested favorable cardiovascular effects of the oil. Contrary to palm olein, more than 80% of the vitamin E and 75% of carotenoids present in crude palm oil is retained in red palm olein. For instance, Boon et al.36 recently reported changes in serum lipid profiles of red palm olein treated hypertensive rats, with a significant reduction in LDL-cholesterol level and TC/HDL ratio (atherogenic index) compared to the untreated ones. Szucs et al.37 recently investigated the effects of dietary RPO supplementation in a cholesterol-enriched diet-induced hyperlipidemic rat model showing attenuation of the increased susceptibility of the hearts in cholesterol fed rats to ischaemia/reperfusion injury. RPO-supplementation also altered the pre-ischaemic levels of matrix metalloproteinase-2 (MMP2), thus indicating that myocardial MMP2 may be implicated as a possible role player in RPO mediated protection against ischaemia/reperfusion injury in the hearts of cholesterol supplemented rats. Previously, Esterhuyse et al.38 also suggested that dietary RPO-supplementation can improve reperfusion aortic output through mechanisms that may include activation of the NO-cGMP and inhibition of the cAMP pathway.
Conclusions
Our study provides evidence that the consumption of 25 mL per day of crude hybrid Elaeis oleifera × E. guineensis palm oil for a period of 3 months has similar effects on human plasma lipids to EVOO, which is considered the “gold standard” for heart disease prevention amongst all edible oils. The positive effects of HPO can be attributed to its high percentage of oleic acid (esterified mainly at the sn-2 position of TAGs) as well as to its elevated content of health-promoting bioactive minor components, especially tocotrienols, that probably lead to further benefits on LDL-cholesterol levels and oxidative damage in addition to those resulting from its remarkable glyceridic composition and structure.Finally, the results obtained in this study provide additional support for the concept that hybrid Elaeis oleifera × E. guineensis palm oil can be seen as the “tropical equivalent of olive oil”.
Acknowledgements
The authors acknowledge the project “Palm oil and cardiovascular disease: effects of red palm olein-rich diet on plasma lipid pattern in hypercholesterolemic patients” [project ID 4735 – Pontificia Universidad Javeriana (Bogotá – Colombia)] and the Hacienda La Cabaña (Bogotá – Colombia) for their donation of oil samples. The authors would also like to thank Vanessa Olaya for her contribution to blood sample analysis.Notes and references
- K. S. Reddy, N. Engl. J. Med., 2004, 350, 2438–2440 CrossRef CAS PubMed.
- Joint WHO/FAO Expert Consultation on Diet, Nutrition and the Prevention of Chronic Diseases (2002: Geneva, Switzerland). WHO Technical Report Series; 916. WHO, Geneva, 2003.
- E. Ros, Am. J. Clin. Nutr., 2003, 78, 617S–625S CAS.
- J. Sabate, Am. J. Clin. Nutr., 2003, 78, 647S–650S CAS.
- P. Garcia-Lorda, M. I. Rangil and J. Salas-Salvado, Eur. J. Clin. Nutr., 2003, 57, 8S–11S CrossRef PubMed.
- G. Riccioni, T. Bucciarelli, M. Mancini, F. Corradi, C. Di Ilio, P. A. Mattei and N. D'Orazio, Ann. Clin. Lab. Sci., 2007, 37, 89–95 CAS.
- B. V. Howard, L. Van Horn, J. Hsia, J. E. Manson, M. L. Stefanick, S. Wassertheil-Smoller, L. H. Kuller, A. Z. LaCroix, R. D. Langer and N. L. Lasser, et al. , J. Am. Med. Assoc., 2006, 295, 655–666 CrossRef CAS PubMed.
- R. Chowdhury, S. Warnakula, S. Kunutsor, F. Crowe, H. A. Ward and L. Johnson, et al. , Ann. Intern. Med., 2014, 160(6), 398–406 CrossRef PubMed.
- R. P. Mensink, P. L. Zock, A. D. M. Kester and M. B. Katan, Am. J. Clin. Nutr., 2003, 77, 1146–1155 CAS.
- E. Fattore, C. Bossetti, F. Brighenti, C. Agostoni and G. Fattore, Am. J. Clin. Nutr., 2014, 99, 1331–1350 CrossRef CAS PubMed.
- M. Mozzon, D. Pacetti, P. Lucci, M. Balzano and N. G. Frega, Food Chem., 2013, 141, 245–252 CrossRef CAS PubMed.
- F. Visioli, P. Bogani, S. Grande and C. Galli, Grasas Aceites, 2004, 55, 66–75 CAS.
- M. Mozzon, D. Pacetti, N. G. Frega and P. Lucci, J. Am. Oil Chem. Soc., 2015, 92, 717–724 CrossRef CAS.
- P. Lucci, D. Pacetti, N. G. Frega and M. Mozzon, Eur. J. Lipid Sci. Technol., 2015, 117, 1027–1036 CrossRef CAS PubMed.
- M. Díaz Perilla and L. Ortiz, Univ. Sci., 2003, 8, 47–55 Search PubMed.
- W. W. Christie, The preparation of derivatives of lipids, in Lipid Analysis, Pergamon Press, Oxford, UK, 2nd edn, 1982, pp. 51–61 Search PubMed.
- G. F. Montedoro, M. Servili, M. Baldioli and E. Miniati, J. Agric. Food Chem., 1992b, 40, 1571–1576 Search PubMed.
- M. R. Loizzo, R. Tundis, M. Bonesi, F. Menichini, V. Mastellone, L. Avallone and F. Menichini, J. Food Compos. Anal., 2012, 25, 179–184 CrossRef CAS PubMed.
- A. L. Guerrerio, R. M. Colvin, A. K. Schwartz, J. P. Molleston, K. F. Murray and A. M. Diehl, et al. , Am. J. Clin. Nutr., 2012, 95, 892–900 CrossRef CAS PubMed.
- L. Lemieux, B. Lamarche, C. Couillard, A. Pascot, B. Cantin, J. Bergeron, G. R. Dagenais and J. P. Després, Arch. Intern. Med., 2001, 161, 2685–2692 CrossRef PubMed.
- FDA allows qualified health claim to decrease risk of coronary heart disease. Press release P04-100. Rockville, MD: U.S. Food and Drug Administration; 1 November 2004. Accessed at http://www.fda.gov/bbs/topics/news/2004/NEW01129.html on 5 December 2007.
- M. A. Carluccio, M. Massaro, E. Scoditti and R. De Caterina, Mol. Nutr. Food Res., 2007, 51, 1225–1234 CAS.
- M. Massaro and R. De Caterina, Nutr., Metab. Cardiovasc. Dis., 2002, 12, 42–51 CAS.
- M. I. Covas, Pharmacol. Res., 2007, 55, 175–186 CrossRef CAS PubMed.
- A. Nagyova, P. Haban, J. Klvanova and J. Kadrabova, Bratisl. Lek. Listy., 2003, 104, 218–221 CAS.
- J. L. Witztum, Lancet, 1994, 344, 793–795 CrossRef CAS.
- M. I. Covas, K. De La Torre, M. Farré-Albaladejo, J. Kaikkonen, M. Fitó, C. López-Sabater, M. A. Pujadas-Bastardes, J. Joglar, T. Weinbrenner, R. M. Lamuela-Raventós and R. de la Torre, Free Radicals Biol. Med., 2006, 40, 608–616 CrossRef CAS PubMed.
- E. Matsuura, G. R. Hughes and M. A. Khamashta, Autoimmun. Rev., 2008, 7, 558–566 CrossRef CAS PubMed.
- S. Vichi, L. Pizzale and S. L. Conte, Eur. J. Lipid Sci. Technol., 2007, 109, 72–78 CrossRef CAS PubMed.
- K. Sundram, Asia Pac. J. Clin. Nutr., 1997, 6, 12–16 CAS.
- V. Bosch, A. Aular, J. Medina, N. Ortiz and R. Apitz, Arch. Latinoam. Nutr., 2002, 52, 145–150 CAS.
- P. J. van Jaarsveld, C. M. Smuts and A. Benadé, Asia. Pac. J. Clin. Nutr., 2002, 11, 424S–432S CrossRef.
- T. Tholstrup, J. Hjerpsted and M. Raff, Am. J. Clin. Nutr., 2011, 94, 1426–1432 CrossRef CAS PubMed.
- M. I. de Z. Giacopini, H. V. Alonso, J. Sánchez, N. García, L. Veliz, I. Golfetto and V. Bosch, Invest. Clin., 2013, 54, 171–179 Search PubMed.
- S. B. Budin, F. Othman, S. R. Louis, M. A. Bakar, S. Das and J. Mohamed, Clinics, 2009, 64, 235–244 CrossRef PubMed.
- C. M. Boon, M. H. Ng, Y. M. Choo and S. L. Mok, PLoS One, 2013, 11, e55908 Search PubMed.
- G. Szucs, D. J. Bester, K. Kupai, T. Csont, C. Csonka, A. J. Esterhuyse, P. Ferdinandy and J. Van Rooyen, Lipids Health Dis., 2011, 10, 103 CrossRef CAS PubMed.
- A. J. Esterhuyse, E. F. du Toit, A. J. Benadè and J. van Rooyen, Prostaglandins Leukot. Essent. Fatty Acids, 2005, 72, 153–161 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5fo01083g |
| This journal is © The Royal Society of Chemistry 2016 |