A general route to enhance the fluorescence of graphene quantum dots by Ag nanoparticles†
Chenxin Rana,
Minqiang Wang*a,
Weiyin Gaoa,
Zhi Yanga,
Jinyou Shaob,
Jianping Denga and
Xiaohui Songa
aXi'an Jiaotong University, Electronic Materials Research Laboratory, Key Laboratory of Education Ministry, Xi'an, Shaanxi 710049, P.R. China. E-mail: mqwang@mail.xjtu.edu.cn
bState Key Laboratory of Manufacturing Systems Engineering, Xi'an Jiaotong University, Xi'an 710049, P. R. China
First published on 8th May 2014
Abstract
Graphene quantum dots (GQDs)/Ag nanoparticles with an unexpected quantum yield of 16.3% are synthesized by an efficient simple solvothermal method at atmospheric pressure. Experimental results show that the enhanced fluorescence is caused by the plasmonic effect of the Ag nanoparticles. The possible mechanism of this fluorescence enhancement is also proposed.
In the past few years, fluorescent carbon-based materials, including carbon dots (CDs),1–4 nanodiamonds, carbon nanotubes, fullerene,5,6 and graphene quantum dots (GQDs),7–13 have drawn increasing attention owing to their exceptional advantages such as high optical absorptivity, chemical stability, biocompatibility, and low toxicity. Among them, luminescent GQDs have been extensively investigated due to their strong quantum confinement and edge effects when their sizes are down to 10 nm, which induce new physical properties.14,15 GQDs shows many potential applications such as bioimaging,16,17 sensors,18 optoelectronic devices19 and photocatalysis20 due to its low cytotoxicity, excellent solubility, chemical inertia, stable photoluminescence and better surface grafting.21,22
On the other hand, metal-enhanced fluorescence (MEF) has attracted much attention and been widely reported.23–25 The unique plasmonic behavior of metal surface/nanoparticle is mainly responsible for the observed fluorescence enhancement through modifying the optical properties of the locally situated fluorophore. When the metal nanoparticles are irradiated with electromagnetic radiation of the appropriate wavelength, localized surface plasmon resonance (LSPR) of the conduction band electrons takes place and induces strong electromagnetic fields on and around the particle surface.26 This leads to an enhancement in the radiative and nonradiative electronic properties of the nanoparticles themselves, as well as nearby electronic systems. Therefore, fluorescence of nearby molecules can be enhanced at distances from the surface that are close enough to feel the surface plasmon near-fields of the plasmonic nanoparticles. However, so far, metal nanoparticles (Ag or Pd) decorated GQDs always shows a fluorescence quenching effect.7–13,27
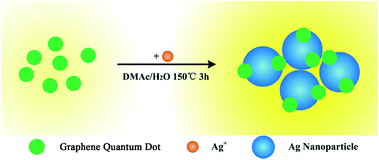
In this communication, we reported a general route to enhance the fluorescence of graphene quantum dots by attaching them on Ag nanoparticles. Here, we synthesized GQDs/Ag composite by using a solvothermal method in a DMF/H2O mixed solvent, which is similar to our previous method with some modification.28,29 The synthesis procedure is shown in Scheme 1, and the detailed experimental process can be seen in ESI.† We, for the first time, observed that the photoluminescence (PL) of GQDs/Ag shows an unexpected enhanced compared with that of initial GQDs, and the mechanism of this enhancement is also discussed.
Fig. 1a shows the transmission electron microscopy (TEM) images of GQDs, which indicates that the products consist of small GQDs particles and are well separated from each other. Inset of Fig. 1a shows the corresponding particle size distribution histograms, and the average diameters of GQDs are 2.90 nm. After the reaction of GQDs with Ag+, Ag nanoparticles are readily formed as shown in Fig. 1b. And the size of GQDs/Ag is measured to be 20–30 nm. Fig. 1c shows the high resolution transmission electron microscopy (HRTEM) image of as-prepared GQDs/Ag composite, it is observed that GQDs (point out by yellow arrows) are attached on the surface of Ag nanoparticles, where the d-spacing plane of Ag can be distinguished and is marked in blue font.
The presence of Ag nanoparticles can further be identified from the UV-vis absorption spectrum in Fig. 2. It is observed that typical absorption peak locates at ca. 282 nm, which is speculated to due to the absorption of the graphitic structure. Magnified figure in Fig. 2 shows a visible absorption peak at around 550 nm for GQDs/Ag composite, which is attributed to the LSPR absorption of Ag nanoparticles. The photos in Fig. 2 shows the light yellow color of GQDs solution, which is commonly observed in the literatures.7,12 While GQDs/Ag composite shows a light brownish red color, which is mainly due to the LSPR absorption at 550 nm.
 | ||
| Fig. 2 UV-vis absorption spectra of GQDs and GQDs/Ag solutions. Magnified figure shows the LSPR absorption of GQDs/Ag composite. Inset: photographs taken under visible light. | ||
The optical properties of GQDs and GQDs/Ag were studied using PL spectrum. Under 380 nm irradiation, the GQDs emit strong green fluorescence (Fig. S1, ESI†), which is attributed to surface defects (defect state emission) of GQDs.7 The fluorescence excitation and emission spectra of GQDs shows the maximum excitation and emission wavelength of GQDs are located at 400 and 570 nm, respectively (Fig. S1, ESI†). The full width at half maximum is about 130 nm, which approximates to that of the most reported green emission GQDs.17 Surprisingly, after attaching GQDs onto Ag nanoparticles, unlike almost no fluorescence of GQDs, we observed a significantly enhanced yellow fluorescence of GQDs/Ag under 560 nm excitation (inset of Fig. 3b). From excitation spectrum we can see that GQDs/Ag composite has a strong excitation peak at around 560 nm, which is just the region located at the LSPR absorption peak of Ag nanoparticles. Taking Rh B as a standard (Table S1, ESI†), the PL quantum yield of GQDs/Ag under 560 nm excitation is determined to be 16.3%, which is considerably higher than that of GQDs (0.38%). As a result, we speculate that this enhanced PL originates from the LSPR effect of Ag nanoparticles. Besides, GQDs/Ag composite exhibits long-term stable photoluminescence ability within 2 month (Fig. S2, ESI†).
 | ||
| Fig. 3 Optical excitation and emission PL spectra of (a) GQDs and (b) GQDs/Ag, inset: corresponding photograph taken under 560 nm excitation. | ||
To further explore the optical properties of as-synthesized GQDs and GQDs/Ag composite, a detailed PL study was carried out using different excitation wavelengths. Fig. 4a and b show the PL emission spectrum of GQDs and GQDs/Ag with various excitation wavelengths from 320 to 560 nm, while Fig. 4c shows PL emission spectrum of GQDs physically mixed with citric- capped Ag nanoparticles (GQDs + Ag) for comparison. Generally speaking, the PL spectra of most luminescent GQDs are dependent on excitation wavelength.30 This excitation dependence properties is believed to result from optical selection of differently sized GQDs and/or different emissive sites on GQDs.17,31 However, in our case, the as-prepared GQDs show an excitation-independent PL wavelength: when the excitation wavelength changed from 320 to 560 nm, the maximum emission peak wavelength of GQDs were almost invariable as shown in Fig. 5a (black curve). This could be explained by the high uniformity both in the size and the surface state of those sp2 clusters contained in GQDs.10 It is worth noting that the fluorescence line shape of GQDs is asymmetric, it is because of the presence of multiple PL peaks, which is due to the existence of multiple electron transition pathways within the electronic structure of the GQDs.32 For GQDs/Ag composite and physically mixed GQDs + Ag in Fig. 5a (red and blue curve), it also shows an almost invariable maximum emission peak wavelength, which suggests that the attachment or physical mixture of GQDs on Ag nanoparticles does not affect the structure of GQDs itself. For GQDs/Ag, this is in agreement with first-principles calculation results that adsorption of graphene on Ag (111) surfaces leads to weak bonding, which preserves the typical electronic structure.33 While for GQDs + Ag, the interaction between GQDs and Ag is even weaker due to the presence of stabilizer molecule between them. Fig. 5b shows the integrated PL intensity as a function of excitation wavelength for GQDs, GQDs/Ag and physically mixed GQDs + Ag. In the range from 320 nm to 480 nm, the introduction of Ag leads to the quenching effect for both GQDs/Ag and GQDs + Ag, which is in agreement with previous works.18,27 It is previously reported in the literature34 that the PL enhancement and PL quenching effect exist in the system at the same time, and the distance between the fluorescent material and plasmonic nanoparticles is mainly responsible for whether it is PL enhancement or PL quenching. The direct contact of plasmonic nanoparticle with fluorescent materials (in our case, GQDs/Ag) can facilitate the injection of photo-excitated electron on Ag into those fluorescent materials thus enhance their fluorescence, that is, enhancement effect wins over quenching effect. While the quenching effect in the range from 320 nm to 480 nm implies that quenching effect wins over the enhancement effect. It is worth noting that when the excitation wavelength changes from 500 to 560 nm, unlike that of GQDs + Ag, the PL intensity of GQDs/Ag increasingly enhanced rather than decreased, and this range is just the LSPR absorption of Ag nanoparticles (Fig. 2). This result strongly suggests that this enhancement effect is caused by the direct contact of GQDs with Ag in GQDs/Ag composite. Besides, we found no fluorescence emission of Ag nanoparticles itself, which rules out the possibility that the enhancement of fluorescence of GQDs is due to Ag NP emission (Fig. S3, ESI†).
 | ||
| Fig. 4 PL spectra of the (a) GQDs, (b) GQDs/Ag and (c) GQDs + Ag at different excitation wavelength. | ||
 | ||
| Fig. 5 The emission peak wavelength (a) and integrated PL intensity (b) as a function of excitation wavelength for GQDs (black), GQDs/Ag (red) and physically mixed GQDs + Ag (blue). | ||
Recently, Qu's group has reported that the presence of Ag nanoparticle could quench the PL of GQDs, which is total opposite to our result.18 Similar fluorescence quenching effect is also observed in the system of palladium nanoparticles on colloidal GQDs.27 Different from their systems, we didn't introduce any stabilizer in our GQD/Ag system and the direct contact between GQDs and Ag is mainly responsible for the enhanced fluorescence. It is reported in the literature that the LSPR-excited electrons generated in plasmonic metal nanoparticles can efficiently transfer onto semiconductors, which facilitates visible-light-sensitive photocatalysis process.35–37 All composites in these works are stabilizer-free, which further prove the important role of direct contact between Ag and GQDs. In our case, the process mainly involve three steps as shown in Fig. 6: (i) under visible light illumination, LSPR-excited electrons would be generated and enriched on the surface of Ag nanoparticles; (ii) the increased electron density lifts the Fermi energy level of Ag, which makes the transfer of LSPR electrons (hot electrons) from Ag to the conduction band of GQDs energetically favorable; (iii) transition of electrons back to ground state in the form of light emission. Eventually, the large amount of this hot electron transfer leads to the enhanced fluorescence of GQDs/Ag composite.
 | ||
| Fig. 6 The proposed mechanism of the enhanced fluorescence of graphene quantum dots by Ag nanoparticles. | ||
In conclusion, we have demonstrated a general route to prepare GQDs/Ag composite, which shows a significant enhanced fluorescence compared with initial GQDs. Based on the experimental results, we confirm that the enhanced fluorescence of GQDs/Ag is due to the LSPR effect of Ag nanoparticles. Under 560 nm excitation, where the LSPR of Ag was excited, a PL quantum yield as high as 16.3% was obtained, which exceeded by 40 times than that of initial GQDs. Furthermore, we proposed that the hot electron transfer from LSPR state Ag nanoparticle to GQDs is responsible for the enhanced fluorescence. The concept in this work is an important step forward in using plasmonic particles to enhanced fluorescence of GQDs, we expected that it will be beneficial to take advantage of LSPR-enhanced fluorescence for high sensitive probe applications.
Acknowledgements
The authors gratefully acknowledge financial support from the NSFC Major Research Plan on Nanomanufacturing (Grant no. 91323303). This work also has been financially supported by Natural Science Foundation of China (Grant nos 91123019 and 61176056) and 111 program (no. B14040).Notes and references
- S. J. Zhu, Q. N. Meng, L. Wang, J. H. Zhang, Y. B. Song, H. Jin, K. Zhang, H. C. Sun, H. Y. Wang and B. Yang, Angew. Chem., Int. Ed., 2013, 52, 3953–3957 CrossRef CAS PubMed.
- W. Kwon, S. Do and S. W. Rhee, RSC Adv., 2012, 2, 11223–11226 RSC.
- M. J. Krysmann, A. Kelarakis, P. Dallas and E. P. Giannelis, J. Am. Chem. Soc., 2012, 134, 747–750 CrossRef CAS PubMed.
- X. Zhai, P. Zhang, C. Liu, T. Bai, W. Li, L. Dai and W. Liu, Chem. Commun., 2012, 48, 7955–7957 RSC.
- A. Krueger, Adv. Mater., 2008, 20, 2445–2449 CrossRef CAS.
- K. Welsher, Z. Liu, S. P. Sherlock, J. T. Robinson, Z. Chen, D. Daranciang and H. Dai, Nat. Nanotechnol., 2009, 4, 773–780 CrossRef CAS PubMed.
- S. J. Zhu, J. H. Zhang, S. J. Tang, C. Y. Qiao, L. Wang, H. Y. Wang, X. Liu, B. Li, Y. F. Li, W. L. Yu, X. F. Wang, H. C. Sun and B. Yang, Adv. Funct. Mater., 2012, 22, 4732–4740 CrossRef CAS.
- D. Y. Pan, J. C. Zhang, Z. Li and M. H. Wu, Adv. Mater., 2010, 22, 734–738 CrossRef CAS PubMed.
- S. Liu, L. Wang, J. Q. Tian, J. F. Zhai, Y. L. Luo, W. B. Lu and X. P. Sun, RSC Adv., 2011, 1, 951–953 RSC.
- L. L. Li, G. H. Wu, G. H. Yang, J. Peng, J. W. Zhao and J. J. Zhu, Nanoscale, 2013, 5, 4015–4039 RSC.
- J. H. Shen, Y. H. Zhu, X. L. Yang, J. hong, J. M. Zhang and C. Z. Li, New J. Chem., 2012, 36, 97–101 RSC.
- S. J. Zhu, J. H. Zhang, L. Wang, Y. B. Song, G. Y. Zhang, H. Y. Wang and B. Yang, Chem. Commun., 2012, 48, 10889–10891 RSC.
- S. J. Zhu, J. H. Zhang, X. Liu, B. Li, X. F. Wang, S. J. Tang, Q. N. Meng, Y. F. Li, C. Shi, R. Hui and B. Yang, RSC Adv., 2012, 2, 2717–2720 RSC.
- L. A. Ponomarenko, F. Schedin, M. I. Katsnelson, R. Yang, E. W. Hill, K. S. Novoselov and A. K. Geim, Science, 2008, 320, 356–358 CrossRef CAS PubMed.
- C. O. Girit, J. C. Meyer, R. Erni, M. D. Rossell, C. Kisielowski, L. Yang, C. H. Park, M. F. Crommie, M. L. Cohen and S. G. Louie, et al., Science, 2009, 323, 1705–1708 CrossRef CAS PubMed.
- S. J. Zhu, J. H. Zhang, C. Y. Qiao, S. J. Tang, Y. F. Li, W. J. Yuan, B. Li, L. Tian, F. Liu, R. Hu, H. N. Gao, H. T. Wei, H. Zhang, H. C. Sun and B. Yang, Chem. Commun., 2011, 47, 6858–6860 RSC.
- Q. Liu, B. D. Guo, Z. Y. Rao, B. H. Zhang and J. R. Gong, Nano Lett., 2013, 13, 2436–2441 CrossRef CAS PubMed.
- X. Ran, H. J. Sun, F. Pu, J. S. Ren and X. G. Qu, Chem. Commun., 2013, 49, 1079–1081 RSC.
- V. Gupta, N. Chaudhary, R. Srivastava, G. D. Sharma, R. Bhardwaj and S. Chand, J. Am. Chem. Soc., 2011, 133, 9960–9963 CrossRef CAS PubMed.
- S. J. Zhuo, M. W. Shao and S. T. Lee, ACS Nano, 2012, 6, 1059–1064 CrossRef CAS PubMed.
- X. Yan, X. Cui and L. S. Li, J. Am. Chem. Soc., 2010, 132, 5944–5945 CrossRef CAS PubMed.
- P. H. Lou, Z. Ji, C. Li and G. Q. Shi, Nanoscale, 2013, 5, 7361–7367 RSC.
- R. Gill, L. J. Tian, W. R. C. Somerville, E. C. L. Ru, H. V. Amerongen and V. Subramaniam, J. Phys. Chem. C, 2012, 116, 16687–16693 CAS.
- C. A. Tao, W. Zhu, Q. An, H. W. Yang, W. N. Li, C. X. Lin, F. Z. Yang and G. T. Li, J. Phys. Chem. C, 2011, 115, 20053–20060 CAS.
- Y. Chen, K. Munechika and D. S. Ginger, Nano Lett., 2007, 7, 690–696 CrossRef CAS PubMed.
- M. A. Mahmoud, A. J. Poncheri, R. L. Phillips and M. A. El-Sayed, J. Am. Chem. Soc., 2010, 132, 2633–2641 CrossRef CAS PubMed.
- X. Yan, Q. Q. Li and L. S. Li, J. Am. Chem. Soc., 2012, 134, 16095–16098 CrossRef CAS PubMed.
- W. Y. Gao, C. X. Ran, M. Q. Wang, X. Yao, D. L. He and J. B. Bai, J. Nanopart. Res., 2013, 15, 1727 CrossRef.
- C. X. Ran, M. Q. Wang, W. Y. Gao, J. J. Ding, Y. H. Shi, X. H. Song, H. W. Chen and Z. Y. Ren, J. Phys. Chem. C, 2012, 116, 23053–23060 CAS.
- Z. X. Gan, S. J. Xiong, X. L. Wu, C. Y. He, J. C. Shen and P. K. Chu, Nano Lett., 2011, 11, 3951–3956 CrossRef CAS PubMed.
- L. Bao, Z. L. Zhang, Z. Q. Tian, L. Zhang, C. Liu, Y. Lin, B. Qi and D. W. Pang, Adv. Mater., 2011, 23, 5801–5806 CrossRef CAS PubMed.
- X. M. Li, S. P. Lau, L. B. Tang, R. B. Ji and P. Z. Yang, Nanoscale, 2014, 6, 5323–5328 RSC.
- P. A. Khomyakov, G. Giovannetti, P. C. Rusu, G. van den Brocks, J. Brink and P. J. Kelly, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 79, 195425 CrossRef.
- P. Anger, P. Bharadwaj and L. Novotny, Phys. Rev. Lett., 2006, 96, 113002 CrossRef.
- Y. Tian and T. Tatsuma, J. Am. Chem. Soc., 2005, 127, 7632–7637 CrossRef CAS PubMed.
- S. Naya, K. Kimura and H. Tada, ACS Catal., 2013, 3, 10–13 CrossRef CAS.
- Z. J. Zhang, W. Z. Wang, E. P. Gao, S. M. Sun and L. Zhang, J. Phys. Chem. C, 2012, 116, 25898–25903 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Synthesis of GO, GQDs, GQDs/Ag, determination of quantum yield of GQDs/Ag, long-term stability test of GQDs/Ag. See DOI: 10.1039/c4ra03542a |
| This journal is © The Royal Society of Chemistry 2014 |