Regio-selective synthesis of polyepichlorohydrin diol using Zn–Co(iii) double metal cyanide complex
Abstract
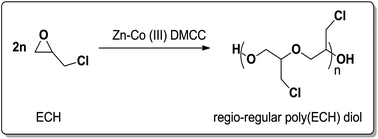
A simple one-pot ring-opening polymerization (ROP) of epichlorohydrin (ECH) was successfully performed by a heterogeneous Zn–Co(III) double metal cyanide complex (Zn–Co(III) DMCC) catalyst, affording regio-regular poly(ECH) diol with a head-to-tail content of >99% in the absence of organic solvent and/or protic compound. No cyclic or oligomer by-product was observed, and the employed Zn–Co(III) DMCC catalyst was recyclable. ROP of (R)- or (S)-ECH led to the generation of semi-crystalline poly(ECH) diol with a melting point of 81 °C or 102 °C, respectively. These regio-regular poly(ECH)s with two end hydroxyl groups are versatile platforms for ECH-based functionalized polymers.

 Please wait while we load your content...
Please wait while we load your content...