Microstructure and dielectric properties of ion-doped La0.7Sr0.3MnO3 lossy ceramics at radio frequencies
Ke-lan Yana,
Run-hua Fan*a,
Xu-ai Wanga,
Min Chena,
Kai Suna,
Zi-dong Zhanga,
Qing Houa,
Lei Qiana,
Shi-bing Pana and
Ming-xun Yub
aKey Laboratory for Liquid-Solid Structural Evolution and Processing of Materials (Ministry of Education), Shandong University, China. E-mail: fan@sdu.edu.cn; Fax: +86 0531 88392315; Tel: +86 0531 88393396
bShandong Non-metallic Materials, China
First published on 16th May 2014
Abstract
In this paper, the microstructure and dielectric properties of ion-doped La0.7Sr0.3MnO3 are investigated in detail. The polycrystalline ceramics of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0.3 or 0.5) are prepared by the sol–gel/sintering method. The symmetry of the crystal structure is improved and the dielectric properties obtained in the frequency range from 80 MHz to 1 GHz can be tuned by the doping of Fe, Ni, and Cu ions. With regard to the dielectric properties in lossy ceramics, the decrease of real permittivity ε′r for the doping of Fe and Ni will be effective for impedance-matching; the enhanced dielectric losses in the Ni and Cu doping samples will promote stronger absorption. Thus, the improved dielectric properties make ion-doped La0.7Sr0.3MnO3 promising candidates for lossy ceramics in microwave electronics. In addition, the experimental results of permittivity were checked by K–K relations, and the frequency dispersion behaviors of the conductivity within a certain frequency range accord with the Jonscher's power law in the form of σ′ac(f) ∝ (2πf)n, demonstrating that the conductive mechanism is hopping conduction.
Introduction
Lossy materials have been widely used in microwave electronics, e.g., power amplifiers such as traveling wave tubes and gyroklystrons, in which lossy materials are used to suppress unwanted electromagnetic modes, prevent self-oscillations, broaden device bandwidth, and provide matched electromagnetic terminations.1 From the viewpoint of applications, the dielectric loss must be relatively large for microwave attenuating materials used in microwave tubes to dissipate unwanted incident power.2 Moreover, the application for microwave-absorbing coating on the exterior surfaces of military aircrafts and vehicles requires avoiding detection by radars by the zero-reflection determined by wave impedance-matching at the surface of the absorbing layer, which requires proper dielectric permittivity at a given frequency.3 Hence, it is significant to tune the dielectric constant and dielectric loss to obtain both specific frequency-matching and attenuation characteristics. In some cases, the fabrication of some tubes necessitates smaller sized lossy ceramics. Therefore, the properties of lossy ceramics should be homogenous, besides the attenuation and frequency-matching requirements. However, most of the works focus on composites that comprise insulators with conductors or semiconductor fillers dispersed within, while the single-phase materials are more favorable to homogenous performance. However, few works have reported on the single-phase of distorted ABO3-type perovskite ceramics with the form of La1−xSrxMnO3 (LSMO) as well as the ion-doped LSMO, even though this structure exhibits a controllable microstructure and various properties via ion doping.Perovskite La1−xSrxMnO3 (LSMO) ceramics as mixed conducting materials have been extensively investigated as cathode materials for high or intermediate temperature solid oxide fuel cells.4,5 After 1993, LSMO gained renewed attention for the discovery of “colossal” magneto-resistance (CMR).6,7 Recently, LSMO that exhibit double exchange,8 electron–phonon coupling,9 super-exchange function and high chemical stability have attracted considerable attention for their potential applications in magnetic electronics, microwave absorption,10 catalytic materials11 and so on. The microstructure and grain sizes usually play a significant role in the electrical, magnetic, transport and CMR performances of LSMO systems,12–15 and a number of investigations on the ion doping of LSMO have been carried out to explore the CMR phenomenon and thermal and electronic transport properties,16,17 but there are only a few reports on the frequency dispersions of the permittivity and conductivity until now.
It is therefore an objective of this study to clarify the ion-doping effects on the complex dielectric permittivity and dielectric loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of La0.7Sr0.3MnO3. Generally, LSMO-based perovskite ceramic powders can be prepared by a variety of techniques. As compared to co-precipitation synthesis and solid state reactions, the main advantages of the sol–gel process is that the liquid precursor with respect to the solid ones exhibits a high degree of chemical homogeneity and the atomic-level dispersion of the reagents, obtainable even in very complex compositions to improve the reactivity. This results in high perfection powders and is possible only when the solution turns into a solid without any fractional precipitation or intermediate phase segregation.18,19 Here, we report on the doping effect of Fe, Ni, and Cu on the microstructure and dielectric properties of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0.3 or 0.5). The improved microstructure and electrical properties are obtained by ion doping, which can be used as a candidate for lossy ceramics in microwave electronics.
δ) of La0.7Sr0.3MnO3. Generally, LSMO-based perovskite ceramic powders can be prepared by a variety of techniques. As compared to co-precipitation synthesis and solid state reactions, the main advantages of the sol–gel process is that the liquid precursor with respect to the solid ones exhibits a high degree of chemical homogeneity and the atomic-level dispersion of the reagents, obtainable even in very complex compositions to improve the reactivity. This results in high perfection powders and is possible only when the solution turns into a solid without any fractional precipitation or intermediate phase segregation.18,19 Here, we report on the doping effect of Fe, Ni, and Cu on the microstructure and dielectric properties of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0.3 or 0.5). The improved microstructure and electrical properties are obtained by ion doping, which can be used as a candidate for lossy ceramics in microwave electronics.
Experimental
In this article, lanthanum nitrate La(NO3)3 (purity 99.0%) was dissolved in anhydrous ethanol solvent (purity 99.7%), strontium acetate Sr(OAc)2 (purity 99.0%) and manganese acetate Mn(OAc)2 (purity 99.0%); further, Fe(NO3)3 (purity 98.5%), Cu(NO3)2 (purity 99.0%), and Ni(OAc)2 (purity 98.0%) were fully dissolved in glacial acetic acid (purity 99.5%). Then, both these solutions were mixed and ethanolamine (purity 99.5%) was used as a chelating agent to stabilize the solutions. This mixed solution was stirred to obtain a clear transparent sol. The sol was dried by evaporation in the oven at 80 °C to yield a dry gel; the dry gel was then calcinated at 750 °C to obtain a polycrystalline oxide powder. The powder was pressed into a pellet specimen and sintered at 1200 °C in air for 2 h.The microstructure of the bulks was investigated by X-ray diffraction (XRD) and field emission scanning electron microscopy (SU-70, FESEM). The measurements in the frequency range from 80 MHz (or 10 MHz) to 1 GHz were carried out at room temperature by using an Agilent E4991A RF Impedance/Material Analyzer (Agilent Technologies). In order to determine the permittivity vs. frequency or various kinds of dielectric parameters, the dielectric test fixture of 16453A was used under an AC voltage of 100 mV, and the samples were processed into square discs (16 mm × 16 mm × 2 mm). During the measurement, the real part (ε′r) and imaginative part (ε′′r) of permittivity were then determined from the following formula:
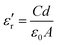 | (1) |
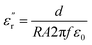 | (2) |
Results and discussion
Fig. 1 shows the XRD patterns of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5) sintered at 1200 °C for 2 hours. As shown in Fig. 1a, besides La0.7Sr0.3Mn0.5Ni0.5O3 with a little impurity phase (i.e. NiO), all the samples are polycrystalline perovskite structures without secondary phases, and the diffraction peaks in the patterns can be indexed to the (012), (110), (104), (202), (006), (024), (122), (214), (300), (220), (208), and (134) planes in comparison with the theoretical pattern of the system trigonal (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c H (167)). Therefore, ion doping can be considered as displacement doping. Taking the preferential growth (110) surface as an example (as shown in Fig. 1b), the symmetry of the crystal structure is improved by the doping of Fe, Ni, and Cu ions and the diffraction peaks are gradually transformed from the bimodal of (110) and (104) to the unimodal of (110). This is attributed to the role of the average ionic radius. Further, the stability and symmetry of the perovskite (ABO3) structure can be characterized by the tolerance factor t,20
c H (167)). Therefore, ion doping can be considered as displacement doping. Taking the preferential growth (110) surface as an example (as shown in Fig. 1b), the symmetry of the crystal structure is improved by the doping of Fe, Ni, and Cu ions and the diffraction peaks are gradually transformed from the bimodal of (110) and (104) to the unimodal of (110). This is attributed to the role of the average ionic radius. Further, the stability and symmetry of the perovskite (ABO3) structure can be characterized by the tolerance factor t,20
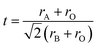 | (3) |
 | ||
| Fig. 1 (a) XRD patterns of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5) after sintering for 2 h at 1200 °C and (b) is the detailed view of the dominant (110) peak of the bulks. | ||
The SEM images taken from the x-section of La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5) show that the bulks are densely sintered (Fig. 2). Further, the average grain sizes of La0.7Sr0.3MnO3, La0.7Sr0.3Mn0.7Fe0.3O3, La0.7Sr0.3Mn0.5Ni0.5O3 and La0.7Sr0.3Mn0.5Cu0.5O3 are about 1 μm, 1.8 μm, 0.2 μm and 0.9 μm, respectively. In addition, it can be seen that the doping with different ions significantly affected the micro-shape. The doping of Fe ion increases the crystal grain size (Fig. 2b), while Ni ion doping has the effect of grain refinement (Fig. 2c). The most obvious influence on the micro-shape is Cu ion doping, as shown in Fig. 2d; the grain shape of La0.7Sr0.3Mn0.5Cu0.5O3 is regularly cubic.
 | ||
| Fig. 2 SEM images of sintered La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5). (a) La0.7Sr0.3MnO3; (b) La0.7Sr0.3Mn0.7Fe0.3O3; (c) La0.7Sr0.3Mn0.5Ni0.5O3; (d) La0.7Sr0.3Mn0.5 Cu0.5O3. | ||
The frequency dependence of permittivity and dielectric loss factor for La0.7Sr0.3Mn1−yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5) is shown in Fig. 3. It can be seen from Fig. 3a that when the doping ions are Fe and Ni, there is an obvious decrease in the real permittivity ε′r when compared with La0.7Sr0.3MnO3, which will be conducive to impedance-matching. However, an abnormal frequency dispersion of ε′r for La0.7Sr0.3Mn0.5Cu0.5O3 was observed, namely, ε′r of La0.7Sr0.3Mn0.5Cu0.5O3 starts to increase above 300 MHz, reaching a peak value of 306 at 800 MHz. There is an absorption peak above 1 GHz for the imaginary permittivity dispersion, implying that the absorption property becomes stronger. The imaginary permittivity measured by the experiment and calculated by the Kramers–Kronig (K–K) relation (marked as the solid line) is shown in Fig. 3b. It is known that K–K relations express relationships between the real and imaginary parts of the complex frequency response of a linear passive causal system and can be conveyed by the following integral transforms for the permittivity:22
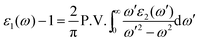 | (4) |
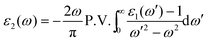 | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of Ni-doped La0.7Sr0.3MnO3 is larger than that of undoped La0.7Sr0.3MnO3, and a dramatic increase of tan
δ) of Ni-doped La0.7Sr0.3MnO3 is larger than that of undoped La0.7Sr0.3MnO3, and a dramatic increase of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ can be seen at about 1 GHz for La0.7Sr0.3Mn0.5Cu0.5O3. Therefore, the enhanced dielectric losses in a wide frequency range can be obtained by appropriate ion doping. Combining Fig. 3a and c, La0.7Sr0.3Mn0.5Ni0.5O3 with large attenuation and improved impedance-matching (relatively small ε′r will be conducive to impedance-matching) will represent perfectly absorbing properties. This improvement in the electrical properties for La0.7Sr0.3Mn0.5Ni0.5O3 would lead to a new category of lossy ceramics.
δ can be seen at about 1 GHz for La0.7Sr0.3Mn0.5Cu0.5O3. Therefore, the enhanced dielectric losses in a wide frequency range can be obtained by appropriate ion doping. Combining Fig. 3a and c, La0.7Sr0.3Mn0.5Ni0.5O3 with large attenuation and improved impedance-matching (relatively small ε′r will be conducive to impedance-matching) will represent perfectly absorbing properties. This improvement in the electrical properties for La0.7Sr0.3Mn0.5Ni0.5O3 would lead to a new category of lossy ceramics.
The frequency dispersions of the real part of ac conductivity in the frequency range from 10 MHz to 1 GHz are shown in Fig. 4. This was determined by the formula of σ′ac = d/RA, where d is the sample thickness, R is the resistance, and A is the electrode plate area. As shown in Fig. 4, the conductivity can be greatly improved by the Cu ion doping. Further, it can be found that the frequency dispersion behavior of the conductivity within a certain range of frequency represents an extension of the universal law, namely, the Jonscher power law23 in the form of σ′ac(f) ∝ (2πf)n with a different power-law index of n. This shows that the conductive mechanism is hopping conduction. For La0.7Sr0.3MnO3, it exhibits a weak frequency-dependent conductivity corresponding to a small value of the exponent n (n ≈ 0.45), yielding a ‘not-quite-dc’ response when the frequency is below 30 MHz. Above this frequency with a higher value of the exponent n (n ≈ 0.87), several papers have described such a type of conduction behavior in a wide range of materials.24 The values of the exponent n for La0.7Sr0.3Mn0.7Fe0.3O3, La0.7Sr0.3Mn0.5Ni0.5O3 and La0.7Sr0.3Mn0.5Cu0.5O3 are 0.62, 0.57 and 0.75, respectively. The physical significance of dispersion at lower frequencies lies in the rise of C′(f) for the formation of interfacial capacitance—the so-called Maxwell–Wagner effect.25
 | ||
| Fig. 4 Frequency dependence of ac conductivity for La0.7Sr0.3Mn1-yMyO3 (M = Fe, Ni, Cu; y = 0, 0.3, 0.5). | ||
Conclusions
In conclusion, the microstructure of La0.7Sr0.3MnO3 can be modified and its dielectric properties and electrical conductivity can be tuned by doping with Fe, Ni, and Cu ions. The XRD results show that ion doping can be considered as displacement doping, and the symmetry of the crystal structure is improved by the doping of Fe, Ni, and Cu ions. According to the SEM images, ion doping significantly influences the size and micro-shape. With regard to Cu ion doping, the grain shape of La0.7Sr0.3Mn0.5Cu0.5O3 is regularly cubic; meanwhile, the real permittivity and conductivity can be greatly improved. With regard to the dielectric properties of lossy ceramics, the tunable dielectric permittivity ε′r and enhanced dielectric losses for La0.7Sr0.3MnO3 can be obtained by appropriate ion doping. Further, La0.7Sr0.3Mn0.5Ni0.5O3 with large attenuation and improved impedance-matching represents perfectly absorbing properties. Therefore, the improved dielectric properties of ion-doped La0.7Sr0.3MnO3 would lead to a new category of lossy ceramics.In addition, the experimental results of permittivity were checked by the K–K relations, and the frequency dispersion behaviors of the conductivity within a certain range of frequency accord with the Jonscher power law in the form of σ′ac(f) ∝ (2πf)n, confirming that the conductive mechanism is hopping conduction.
Acknowledgements
The authors acknowledge the supports of National Natural Science Foundation of China (51172131).Notes and references
- C. Z. He, S. Qiu, X. Z. Wang, J. R. Liu, L. Q. Luan, W. Liu, M. Itoh and K. Machida, J. Mater. Chem., 2012, 22, 22160 RSC.
- Z. J. Wang, L. Wu, J. G. Zhou, B. Z. Shen and Z. H. Jiang, RSC Adv., 2013, 3, 3309 RSC.
- F. J. Ren, H. J. Yu, L. Wang, M. Saleem, Z. F. Tian and P. F. Ren, RSC Adv., 2014, 4, 14419 RSC.
- E. Lus, R. Küngas, I. Kivi, H. Kurig, P. Möller, E. Anderson, K. Lust, K. Tamm, A. Samussenko and G. Nurk, Electrochim. Acta, 2010, 55, 7669 CrossRef PubMed.
- V. Siruguri, R. Ganguly, P. S. R. Krishna, P. U. Sastry, S. K. Paranjpe, I. K. Gopalakrishnan and J. V. Yakhmi, J. Mater. Chem., 2001, 11, 1158 RSC.
- L. Chen, C. H. Lu, Y. Lu, Z. G. Fang, Y. R. Ni and Z. Z. Xu, RSC Adv., 2013, 3, 3967 RSC.
- E. Dagotto, T. Hotta and A. Moreo, Phys. Rep., 2001, 344(1), 1 CrossRef CAS.
- K. Zhang, J. Sunarso, Z. P. Shao, W. Zhou, C. H. Sun, S. B. Wang and S. M. Liu, RSC Adv., 2011, 1, 1661 RSC.
- Y. Tokura and N. Nagaosa, Science, 2000, 5465(288), 462 CrossRef.
- K. S. Zhou, H. Xia, K. L. Huang, L. W. Deng, D. Wang, Y. P. Zhou and S. H. Gao, Phys. B, 2009, 404, 175 CrossRef CAS PubMed.
- X. D. Wu, L. H. Xu and D. Weng, Catal. Today, 2004, 90, 199–206 CrossRef CAS PubMed.
- Y. L. Cheng, J. M. Dai, X. B. Zhu, D. J. Wu and Y. P. Sun, Mater. Res. Bull., 2010, 664(4), 5663 Search PubMed.
- J. H. Wang, B. X. Gu, H. Sang, G. Ni and Y. W. Du, J. Magn. Magn. Mater., 2001, 223, 50 CrossRef CAS.
- M. Jain, E. Bauer, F. Ronning, M. F. Hundley, L. Civale, H. Y. Wang, B. Maiorov, A. K. Burrell, T. M. McClesky, S. R. Foltyn, R. F. DePaula and Q. X. Jia, J. Am. Ceram. Soc., 2008, 91(6), 1858–1863 CrossRef CAS PubMed.
- Z. H. Li, F. L. Wei, S. Yoshimura, G. Q. Li, H. Asano and H. Saito, Phys. Chem. Chem. Phys., 2013, 15, 628 RSC.
- M. Battabyal and T. K. Dey, Phys. B, 2006, 373, 46–53 CrossRef CAS PubMed.
- R. V. Wandekar, B. N. Wani and S. R. Bharadwaj, Solid State Sci., 2009, 11, 240–250 CrossRef CAS PubMed.
- S. P. Jiang, J. Power Sources, 2003, 124(2), 390–402 CrossRef CAS.
- A. Pohl, G. Westin and K. Jansson, J. Sol-Gel Sci. Technol., 2003, 26(2), 257–260 CrossRef CAS.
- T. Yang, T. Perkisas, J. Hadermann, M. Croft, A. Ignatov, G. V. Tendeloo and M. Greenblatt, J. Mater. Chem., 2011, 21, 199 RSC.
- T. Shibata, B. A. Bunker and J. F. Mitchell, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 68, 024103 CrossRef.
- H. Chen, J. Mater. Chem., 2011, 21(18), 6452 RSC.
- S. K. Prasad, M. V. Kumar, T. Shilpa and C. V. Yelamaggad, RSC Adv., 2014, 4(9), 4453 RSC.
- X. J. Kuang and X. P. Jing, J. Am. Ceram. Soc., 2006, 89(1), 241–246 CrossRef CAS PubMed.
- Q. H. Guo, J. J. Bian and L. Wang, J. Am. Ceram. Soc., 2011, 94(1), 172–178 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |

