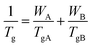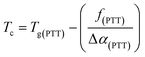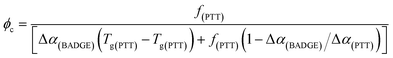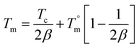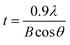DOI:
10.1039/C4RA01429D
(Paper)
RSC Adv., 2014,
4, 25420-25429
Stereochemistry and miscibility of epoxy resin–poly(trimethylene terephthalate) blends
Received
18th February 2014
, Accepted 23rd April 2014
First published on 23rd April 2014
Abstract
Stereochemistry is proposed to contribute to the miscibility of poly(trimethylene terephthalate) (PTT) and bisphenol-A diglycidyl ether (BADGE), since molecular conformation is one of the determinants of the close packing ability and hence the interactions of such a system. Polymer blends were prepared by the conventional melt mixing technique using a HAAKE mixer. The influence of the stereochemistries of the mixing polymers on the miscibility has been poorly investigated in the literature. The amorphous fraction of PTT was found to be miscible with BADGE, while the crystalline fraction was found to be immiscible with BADGE. This is an interesting situation where one fraction of the polymer is miscible while another fraction is immiscible. The stereochemistries of the glycol residues of PTT are different in the crystalline and the amorphous fractions, and therefore the miscibility of the amorphous fraction of PTT with BADGE could be due to the π–π and n–π interactions between the phenyl and carbonyl groups of PTT with the aromatic rings of BADGE. Fourier transform infra-red microscopy (FTIR microscopy), wide angle X-ray scattering (WAXS) and thermogravimetric analyses show that PTT and BADGE retain their identities in the blend, eliminating the possibility that any trans reactions occur between the blend components.
Introduction
The majority of previously investigated polymer pairs were found to be immiscible.1 The development of sophisticated techniques to detect miscibility has resulted in an increase in the known number of miscible polymer blends.1 Ultraki discussed the existence of around 1600 miscibility composition–temperature windows.2 This shows that studies relating to the interactions between blend components are a topic of vital importance because the presence of interactions between blend components has a dominating contribution on the phase morphology and hence towards the final properties of a blend. Here we follow the miscibility of poly(trimethylene terephthalate) (PTT) with a bisphenol-A diglycidyl ether (BADGE) type of epoxy resin. The use of BADGE as a reactive compatibilizer for PTT–poly(butylene terephthalate) (PBT) has been reported previously,3–5 but an in-depth investigation into the nature of the interactions and the exact reasons for the miscibility is missing.
The miscibilities of many polyesters, like poly(ethylene terephthalate) (PET)6 and poly(butylene terephthalate) (PBT)7 with BADGE have been reported. Reactions between the –OH and –COOH end groups of PET/PBT, with the oxirane rings of BADGE were used to explain the miscibility in these systems. The contribution of these reactions towards the miscibility is negligible for two reasons: (1) the number of –OH and –COOH end groups are very low, (2) experimental detection of the reaction is relatively difficult. A detailed investigation shows that the miscibility and the behaviour of PTT towards BADGE is predominantly governed by the π–π and n–π interactions. PTT, a higher homologue of PET and a lower homologue of PBT, has a unique stereochemistry where the phenyl groups on the PTT chain can be considered hard segments, while the ether linked propane chains can be considered soft segments. This gives rise to a helical structure that accounts for the exceptionally high resilience of PTT. Research shows that the tggt conformation of the soft segments corresponds to the crystalline fraction while the gttg and the all trans conformations correspond to the amorphous fraction.8,9 Thus the crystalline and amorphous fractions of PTT have different stereochemistries. The aim of the present research is to investigate the miscibility of PTT with BADGE. The possibility of reactions between PTT and BADGE is considered using differential scanning calorimetry (DSC), Fourier transform infrared (FT-IR) spectroscopy, wide angle X-ray scattering (WAXS), and polarized optical microscopy (POM), while the thermal stabilities of the blends are investigated using thermogravimetry.
Experimental
Materials used
BADGE epoxy resin (Lapox L-12) with an epoxy equivalent between 5.25–5.40 eq. kg−1 and a viscosity between 1.15–1.20 cm2 s−1 was obtained from Atul Industries, Gujarat. Poly(trimethylene terephthalate) (PTT or Sorona 3G) with a density between 1.3–1.5 g cm−3, a number average molecular weight (![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) n) of 22
n) of 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 g mol−1 and a polydispersity index (PDI) of 2.5 was kindly supplied by DuPont Industries, USA. Details of the materials used are given in Table 1, and the chemical structures are shown in Fig. 1.
500 g mol−1 and a polydispersity index (PDI) of 2.5 was kindly supplied by DuPont Industries, USA. Details of the materials used are given in Table 1, and the chemical structures are shown in Fig. 1.
Table 1 Characteristics of the PTT and the BADGE used for this study
| |
PTT |
BADGE |
| Molar mass as estimated in this work by GPC (Perkin–Elmer). Polystyrene with a low polydispersity was used as the standard. Information provided by the supplier. Glass transition temperature as determined in this work by mT-DSC. Apparent melting temperature for the neat polymer during a first heating scan by DSC. The melting enthalpy of 100% crystalline PTT crystals. |
![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) wa (g mol−1) wa (g mol−1) |
58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
— |
![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) na (g mol−1) na (g mol−1) |
22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
— |
| Viscosityb (cm2 s−1) |
— |
1.15–1.20 |
| Epoxy contentb (equ. kg−1) |
— |
5.2–5.4 |
| Tgc (°C) |
52.5 |
−16 |
| Tmd (°C) |
227 |
— |
| ΔHrefe (J g−1) |
146 |
— |
| Supplier |
DuPont Industries, USA. |
Atul Industries, Gujarat, India. |
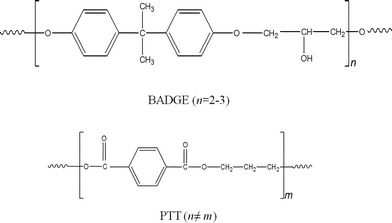 |
| | Fig. 1 Chemical structures of PTT and BADGE (n ≠ m). | |
All of the materials were used as received without further purification. The PTT–BADGE blends with varying amounts of PTT were prepared by a conventional melt mixing technique using a Thermo Fisher Rheomixer. Blends with and without a curing agent were subjected to detailed analyses.
Blend preparation
Neat PTT pellets and PTT–BADGE blends with different amounts of BADGE (5, 10, 20 and 30% w/w) were mixed using a Thermo Fisher Rheomixer at a torque of 10 MPa, and temperature of 245 °C for 15 min. The prepared blends were compression moulded into sheets by pressing at 245 °C, and at 10 kPa, for 15 min. The blends were then subjected to detailed analysis and characterization.
Analysis
Differential scanning calorimetry. Differential scanning calorimetry (DSC) measurements were conducted using a Perkin Elmer Pyris 1 DSC instrument. The prepared blends were subjected to the following studies:(a) glass transition temperature (Tg) measurements.
The Tg studies were performed on the PTT, the PTT–BADGE blends and the BADGE using the Perkin Elmer DSC instrument. About 5 mg of samples were accurately weighed using a Mettler microbalance, heated to 260 °C at a heating rate of 10 K min−1 and annealed for 3 min to remove any thermal history, followed by cooling to 20 °C at a rate of 10 K min−1. The sample was then heated again to 260 °C at 10 K min−1. Endothermic deviations from the baseline observed during the second heating cycle were noted.
(b) Equilibrium melting temperature (Tm°) studies.
The Tm° values for the PTT and the PTT–BADGE blends were calculated by heating the samples to an annealing temperature (Ta) of 260 °C, followed by quenching to selected crystallization temperatures (Tc), and held at those Tc values for 60 s. Each sample was then heated to 260 °C, and the melting temperature (Tm) obtained during the second heating was used for further analysis.
Wide angle X-ray scattering (WAXS) studies. WAXS analysis was carried out on PTT and PTT–BADGE blends using a Bruker AXS D8 X-ray diffractometer equipped with a High Temperature XRD cell (HTK2000), between 2θ ranges of 10–35°.
Fourier transform infrared (FTIR) spectroscopy. FTIR spectroscopy studies on the PTT and the PTT–BADGE blends were carried out using an ATR (Golden Gate Unit) Bruker-FTIR spectrometer, as described by Aravind et al.10
Fourier transform infrared microscopy (FTIR microscopy). FTIR Microscopy analysis was carried out on selected samples (neat PTT and 70% w/w blend) using a Perkin Elmer Spotlight 400 FTIR microscope. Samples were studied using a Germanium ATR attachment, with 64 scans per pixel at a resolution of 16 cm−1. The results obtained were analysed using Perkin Elmer Spectrum Image software. Results obtained for neat PTT and the 70% w/w blend are presented here.
Polarized optical microscopy. The morphologies of the neat PTT and the PTT–BADGE blends (50–50) were imaged with a Nikon Labophot 2 microscope (Yokohama, Japan), equipped with a Linkam heating/cooling unit (Linkam TM 600/s−1) (Surrey, UK), with the images collected using Leica Q Win software. The neat PTT and the 50–50 blends were heated to 250 and 215 °C respectively and then cooled to 198 °C using a liquid nitrogen atmosphere at a cooling rate of 50 K min−1 and then held at that temperature for 1500 s.
Thermogravimetric analysis (TGA). TG analysis was carried out on the PTT and the PTT–BADGE blends using a Perkin Elmer TGA7 instrument calibrated with Curie temperature metal standards. The samples were heated from room temperature to 800 °C at a heating rate of 10 K min−1 in a dry nitrogen atmosphere purge. The results obtained were analysed with Perkin Elmer Pyris software.
Results and discussion
Glass transition temperature
The glass transition, related to cooperative segmental motion involving about 50–100 backbone carbon atoms, is an Ehrenfest type second order transition.11 Glass transitions exist between the glassy region, and the liquid super-cooled region to below its melting temperature. A single glass transition temperature for polymer blends indicates that the domain size (dd) is between 2–15 nm.11,12 Thus a single Tg for polymer blends does not imply miscibility and in turn gives information about the state of dispersion.11 Tg values can be correlated with miscibility by using equations like the Fox equation,13 the Couchman equation,14 and the Gordon–Taylor equation.15 The DSC heating curves of the PTT–BADGE blends are shown in Fig. 2.
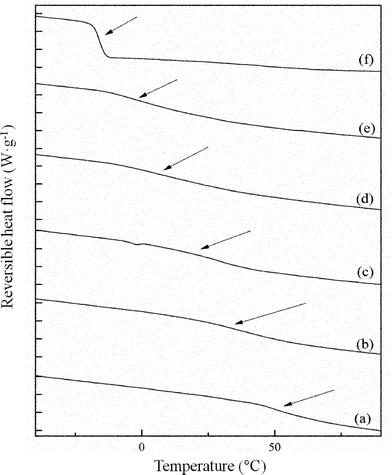 |
| | Fig. 2 mT-DSC curves for the PTT–BADGE blends. (a) Neat BADGE, (b) 50% w/w of BADGE, (c) 30% w/w of BADGE, (d) 20% w/w of BADGE, (e) 10% w/w of BADGE and (f) neat PTT. | |
All of the blends showed a single Tg value and the experimentally observed Tg values were fitted with the Fox model (1).
| |
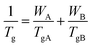 | (1) |
where
WA and
WB are the weight fractions of the neat polymers while
TgA and
TgB are the corresponding glass transition temperatures. The experimentally observed
Tg values showed a large deviation from the Fox model as shown in
Fig. 3.
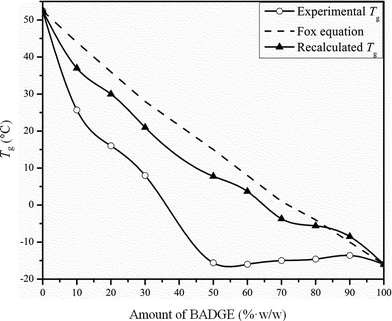 |
| | Fig. 3 Tg values for the PTT–BADGE blends as a function of the BADGE composition. | |
Such a deviation from the Fox model has been reported previously for BADGE–PEO (polyethylene oxide) blends.16 Therefore the contribution from the crystalline fraction of the PTT was reduced, and the recalculated Tg values showed a better fit with the Fox equation. The contribution from the crystalline fraction was reduced using eqn (2)
| | |
W(PTT)′ = (W(PTT) − Xc)/(1 − Xc)
| (2) |
where
W(PTT)′ is the weight fraction of amorphous PTT in the blend,
W(PTT) is the actual weight fraction of PTT in the blend, and
Xc is the crystallinity calculated from the DSC measurements. This result confirms that the amorphous fraction of PTT is miscible with BADGE. Similarly an increase in Δ
Tg and Δ
Cp values indicated an increased segmental mobility in the blends as the amount of BADGE increased. The experimentally observed
Tg values shown in
Fig. 3 follow a negative deviation with a pronounced “cusp” or discontinuity. The deviation of the
Tg values from the linear behaviour is considered as a measure of the strength of the interactions between the blend components.
17 A “cusp” or discontinuity in
Tg values was first observed by Kovacs
et al.17 and was explained using the free volume theory. Accordingly if the difference in
Tg values between the blend components is more than 50 degrees, then the free volume of the polymer with the higher
Tg value will approach zero at a certain critical temperature (
Tc). The
Tc and the corresponding critical composition (
ϕc) can be calculated using
eqn (3) and
(4), as proposed by Kovacs.
17| |
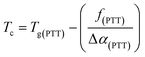 | (3) |
| |
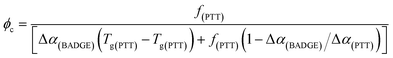 | (4) |
where
f(PTT) is the free volume of PTT at
Tg(PTT), and Δ
α(PTT) is the difference between the volume expansion coefficients in the glassy and liquid states. It is significant to note that the observed “cusp” can be masked if the contribution of the crystallinity is not taken into consideration, which matches with the results shown in
Fig. 3.
Equilibrium melting temperature
The equilibrium melting temperature (Tm°) is one of the basic physical quantities of polymers, and is defined as the melting temperature (Tm) of ideal crystals with an infinite crystal size.18 The DSC exotherm was used to calculate the equilibrium melting temperature (Tm°) using the Hoffmann–Weeks eqn (5).19| |
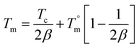 | (5) |
Here Tm and Tc are the observed melting and crystallization temperatures respectively while β is the lamellar thickness. The Tm values obtained at different Tc values are plotted against each other and from this the Tm° values are calculated by extrapolation to where Tm = Tc. Usually the addition of an amorphous component into a semi-crystalline polymer should result in a decrease in the Tm° value, and this is usually observed.19 Thus a plot of (1/Tm°–1/Tm°′) against ϕ2 should give a straight line with a negative slope (where Tm°′ represents the equilibrium melting temperature of the blend), which can be used to calculate the interaction parameter between the blend components by the Nishi–Wang equation.20,21 The experimentally observed Tm values for the PTT–BADGE blends were plotted against the Tc values as shown in Fig. 4, and the Tm° values were calculated. The calculated values of Tm° are shown in Table 2. The Tm° values calculated using the Hoffmann–Weeks method (see Fig. 4) were plotted against the volume fraction of BADGE (ϕ), as shown in Fig. 5. This led to the fact that a plot of (1/Tm° – 1/Tm°′) against ϕ2 shows a positive deviation rather than the expected negative deviation (for low amounts of BADGE). This means that the crystalline fraction of PTT is incompatible with BADGE. On increasing the amount of BADGE above 20% w/w, BADGE enters into the PTT spherulites, resulting in a change in the morphology of the PTT, as observed in the decrease in lamellar thicknesses shown in Table 3 using WAXS studies. This can be due to the entropy contribution associated with the low molecular weight of BADGE. This conclusion of a change in morphology was confirmed by the POM images (see Fig. 14).
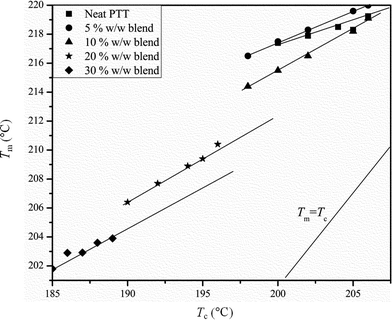 |
| | Fig. 4 Tm against Tc curves for the PTT–BADGE blends. | |
Table 2 Tm° values along with the slope coefficients (α) and the correlation coefficients (r) for the PTT–BADGE blends
| % w/w of BADGE |
Tm°/°C |
α |
r |
| 0 |
231.8 |
0.490 |
0.853 |
| 5 |
232.1 |
0.476 |
0.990 |
| 10 |
232.4 |
0.573 |
0.995 |
| 20 |
232.6 |
0.660 |
0.998 |
| 30 |
218.6 |
0.490 |
0.915 |
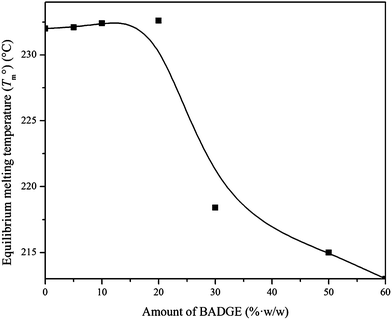 |
| | Fig. 5 Plot of Tm° values against the amount of BADGE in the PTT–BADGE blends. | |
Table 3 Variation of W1/2 values and lamellar thicknesses as a function of the amount of BADGE
| Composition (% w/w) |
Peak width at half maximum (W1/2) (°) |
Lamellar thickness t (nm) |
| 0 |
0.2814 |
2846.9 |
| 5 |
0.2926 |
2737.9 |
| 10 |
0.3729 |
2148.3 |
| 20 |
0.3202 |
2501.9 |
| 30 |
0.3206 |
2498.8 |
| 50 |
0.4396 |
1822.1 |
Thus in these PTT–BADGE blends we have two situations; an amorphous PTT fraction which is miscible with BADGE and a crystalline PTT fraction that is immiscible with BADGE. This situation could arise from the fact that the crystallization of PTT results in an expulsion of BADGE from the intra-spherulitic region into the inter-spherulitic region. The optical micrograph for the 50% w/w blend, after 3600 s of crystallization at 186 °C, as shown in Fig. 6, confirms this argument. The inter-spherulitic regions, where the amorphous fractions of PTT and BADGE localize, are shown in black circles. The reason behind the selection of the 50% w/w blend, was that it showed more visible regions when compared to others with lower amounts of BADGE. Wide angle X-ray scattering (WAXS) studies on the PTT–BADGE blends show that the PTT retains its unit cell dimensions, which rules out the possibility of any trans reactions between the PTT and the BADGE. Fig. 7 shows the diffraction patterns observed for PTT with varying amounts of BADGE. WAXS plots show that the intensity of the diffraction peaks (010–012) increase as the amount of BADGE increases, indicating slight changes in the unit cell dimensions that can be neglected. The peak width at half maximum height (W1/2) gives indirect information about the lamellar thickness. The diffraction peak at (010) was taken for this calculation. The W1/2 values for neat PTT and for PTT–BADGE blends show an increase, while the lamellar thicknesses decrease. The lamellar thicknesses were calculated using the Scherrer eqn (6).
| |
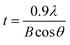 | (6) |
 |
| | Fig. 6 POM image for the 50% w/w blend after 3600 s of crystallization at a temperature of 186 °C. | |
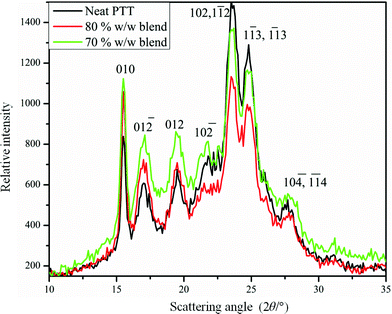 |
| | Fig. 7 WAXS profiles for the PTT–BADGE blends. | |
Here t is the lamellar thickness, λ = 0.154 nm is the wavelength of the X-ray, B is the full width at half maximum (FWHM), and θ is the scattering angle corresponding to the peak under selection. The lamellar thickness values shown in Table 3 decrease as the amount of BADGE in the blends increases, which can be due to two reasons: (1) the higher the loadings of BADGE, the more the morphology of the PTT spherulites changes to a sheaf-like or dendrimeric structure; (2) increasing the amount of BADGE reduces the crystallization of PTT, which in turn increases the amount of the amorphous fraction of PTT. FTIR spectra of PTT have been studied in detail by different groups,9,22 and it has been concluded that the peaks at 933, 947, 1037, 1358 and 1465 cm−1 correspond to the crystalline fraction of PTT, while the peaks at 811, 976, 1173, 1328, 1385, 1452, and 1577 cm−1 correspond to the amorphous fraction. It is assumed that in the crystalline fraction the stereochemistry of the glycol residues (soft segments) are predominantly tggt, whereas in the amorphous fraction the stereochemistry of the soft segments are gttg or all trans. These results were further confirmed by photoluminescence studies at different temperatures.23 The above studies show that the tggt conformations result in the arrangement of the phenyl rings at a distance of 0.36 nm and thereby the π–π interactions between the phenyl rings assist crystallization. The FTIR spectra of neat PTT and PTT–BADGE blends are shown in Fig. 8. The results obtained were used to calculate the blend mixing ratios, and the amount of gauche conformations in the PTT and the PTT–BADGE blends. In the calculation of blend composition using FTIR spectroscopy, the peaks at 826 cm−1 (the C–H out-of plane aromatic ring (p-phenyl), 819 cm−1 in the blend), and 730 cm−1 (rocking due to the aromatic ring), were used as the internal reference,10,24 and can be considered as constant internal standards for the calculation of the blend mixing ratio. The calculated blend mixing ratios are presented in Fig. 9, and this shows a linear relationship (within the error range), confirming the blend compositions. The amount of gauche confirmations in the amorphous and crystalline fractions of PTT can be obtained from the A1358/A1504 ratio. A plot of A1358/A1504 against the amount of BADGE shows a decrease in the A1358/A1504 ratio as the amount of BADGE increases, as shown in Fig. 10. This confirms the presence of weak interactions between the PTT and the BADGE. The possibility of reactions between the hydroxyl (–OH) and carboxylic acid (–COOH) end groups of PTT with the epoxy groups of BADGE was studied in detail. For this the peaks at 826 cm−1, 913 cm−1 and 1507 cm−1 were considered. An almost linear fit was observed within the error range for the A913/A1507, and the A913/A826 ratios plotted against the amount of BADGE (see Fig. 11), which confirms that the contribution of these reactions towards the miscibility is negligible. Thus the contribution of chemical reactions, between the end groups of PTT and BADGE, towards miscibility can be neglected. The FTIR microscopy images for the neat PTT show the presence of the same spectral fingerprint, as expected. A chemimap image, along with the spectra of different regions of the specimen, is shown in Fig. 12. The FTIR microscopy image of the 70% w/w blend given in Fig. 13 shows the presence of two regions: (1) the PTT matrix and (2) dispersed BADGE droplets. This confirms our statement that the crystallization of PTT results in an expulsion of BADGE from the intra-spherulitic region into the inter-spherulite region. POM images of the neat PTT and 50–50 blend are shown in Fig. 14. The neat PTT shows a banded spherulite structure with well-defined maltese crosses, whereas in the case of the 50–50 blend, the bands are not present. The 50–50 blend shows well-defined maltese crosses and dendrimeric spherulites. This can be due to the fact that the excess BADGE causes a swelling of the PTT lamellae. TGA of the PTT–BADGE blends shows that the addition of BADGE has little effect on the degradation temperature of PTT, as shown in Fig. 15. The interesting conclusion here is that as the amount of BADGE increases gradually, the degradation of BADGE starts to appear as shown in Fig. 15(b). Thus, until the 90% w/w blend, there is a single stage degradation, after which a two stage degradation is observed, with the first degradation occurring at a temperature close to that of the degradation temperature of BADGE. The above discussion leads to the conclusion that the amorphous fraction of PTT is miscible with BADGE, while the crystalline fraction is immiscible with BADGE. This can be explained using the schematic given in Fig. 16. In the homogenous melt there is a uniform distribution of BADGE as shown in Fig. 16(a). On cooling the tggt confirmations of the crystalline fraction result in effective packing, that leads to the expulsion of BADGE from the intra-spherulitic region into the inter-spherulitic space, resulting in a decrease in the interactions between PTT and BADGE, as shown in Fig. 16(c). Fig. 16 represents the situations that can be expected during the mixing of PTT with BADGE. In the molten state the BADGE monomers are expected to be distributed uniformly throughout the amorphous PTT matrix, this is shown in Fig. 16(a) with the blue droplets representing the BADGE monomers. On crystallization, the packing of the PTT lamellae causes an expulsion of the BADGE monomers, and the amorphous fractions of PTT, which are miscible with BADGE, are shown in blue in Fig. 16(b) and (c). The expected reason for the miscibility of the amorphous fraction of PTT with the BADGE monomers can be envisaged as shown in Fig. 16(d). The more open structure of amorphous PTT (the gttg and the all trans conformations) allows extensive interactions with BADGE resulting in miscibility, while the opposite can be expected for the crystalline fractions of PTT, as shown in Fig. 16(e).
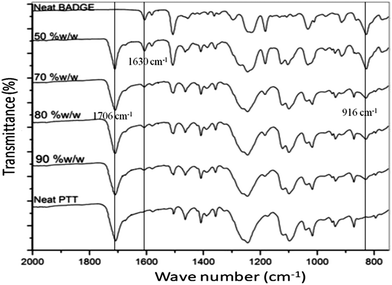 |
| | Fig. 8 FTIR spectra of PTT and PTT–BADGE blends. | |
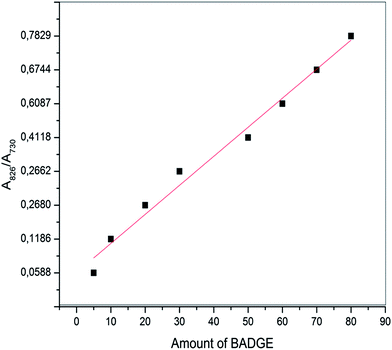 |
| | Fig. 9 Ratios of the absorbance peaks (A826/A730) against the amount of BADGE in the PTT–BADGE blends. | |
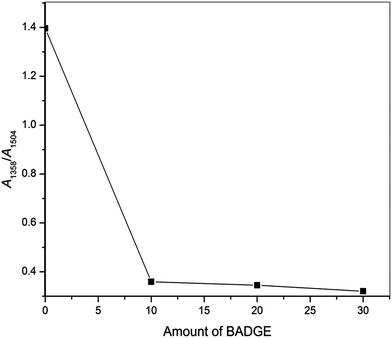 |
| | Fig. 10 Ratios of the absorbance peaks at 1358 to 1504 cm−1 (A1358/A1054) for the neat PTT and the PTT–BADGE blends. | |
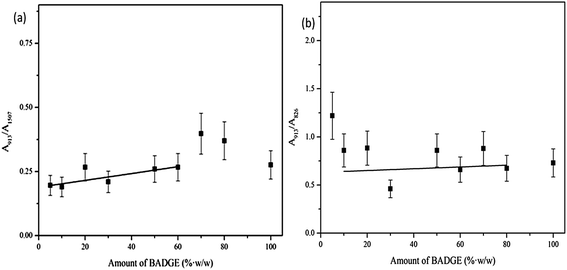 |
| | Fig. 11 FTIR results for the epoxy indices calculated using the two peaks corresponding to the oxirane rings of BADGE. (a) Epoxy index 1 (ep 1) (A913/A1057) against the amount of BADGE. (b) Epoxy index 2 (ep 2) (A913/A826) against the amount of BADGE. | |
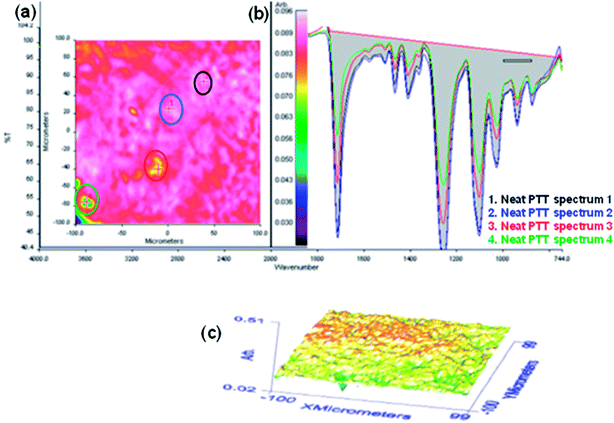 |
| | Fig. 12 FTIR microscopy images for neat PTT. (a) The relative intensity of the distribution of the region between 1740–750 cm−1. (b) The relative intensity of the spectra. (c) The 3D projection image of the surface. | |
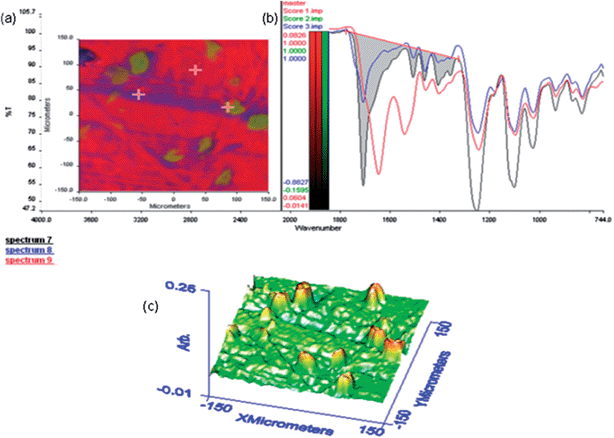 |
| | Fig. 13 FTIR microscopy images for the 70% w/w blend. (a) A chemimap image of the region under investigation. (b) Spectra from the three different regions indicated in the chemimap image. (c) The 3D projection image of the region of interest. | |
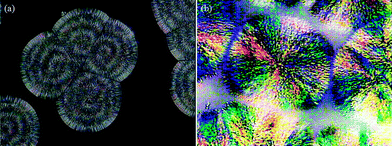 |
| | Fig. 14 POM images of (a) neat PTT and (b) the 50–50 blend at 198 °C after 1300 s at a scale bar of 25 μm. | |
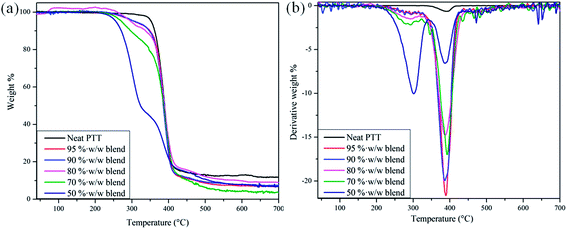 |
| | Fig. 15 Plots of mass against temperature for the PTT–BADGE blends. (a) A plot of the mass% as a function of temperature. (b) The derivative weight% as a function of temperature. | |
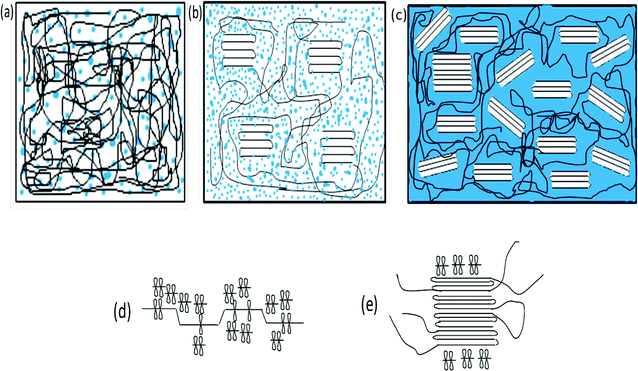 |
| | Fig. 16 Schematic representation of the interactions between PPT and BADGE in the crystalline and amorphous regions of PTT. (a) The situation expected in the molten state. (b) The situation after the onset of crystallization (c) The situation close to the endset of crystallization. (d) The reason expected for the interactions between PTT and the amorphous BADGE monomers. (e) The reason for the immiscibility of the crystalline fraction of PTT with BADGE. | |
Conclusions
The influence of the stereochemistry of the thermoplastic polyester PTT on its miscibility with BADGE has been investigated. Glass transition temperature (Tg) studies show that the blend’s behaviours are described by the Fox equation when crystallinity is excluded from the blend weight fractions. This indicates that the amorphous fraction of PTT is miscible with BADGE. Apparent melting temperature (Tm°) values show a slight increase that points towards incompatibility, that is the crystalline fraction is immiscible with BADGE. This apparent change in miscibility can be explained by the fact that the tggt conformations of the soft segments of PTT result in effective packing that reduces the possibility of interactions between the PTT and BADGE. Whereas the gttg and the all trans conformations of the amorphous fraction of PTT, result in more open structures that induce greater π–π and n–π interactions between PTT and BADGE. WAXS studies show that the lamellar thicknesses gradually decrease as the amount of BADGE in the blends increases. Thus at higher loadings of BADGE, a change in the morphology of the PTT crystals is expected and this is in exact correlation with the POM results. TGA shows the presence of multiple decomposition peaks, the lower decomposition peak corresponds to that of BADGE, while the decomposition at higher temperature corresponds to that of PTT. The intensity of the lower decomposition peak increases as the amount of BADGE in the blends increases. The FTIR microscopy results show the presence of two different regions that indicate two results: (1) no possibility of any reaction between PTT and BADGE at higher compositions, (2) crystallization of PTT results in an expulsion of BADGE from the intra-spherulite region into the inter-spherulite region. Thus the stereochemistries of the polymers play a major role in the miscibilities of the blends. The results obtained are extremely important in the fabrication of novel materials with smart applications, and further studies into this relationship are in progress.
References
- L. M. Robeson in Polymer blends: a comprehensive review, ed. L. M. Robeson, Hanser Verlag, Cincinnati, 2007, 2, pp. 11–54 Search PubMed.
- G. Groueninckx, M. Vanneste, V. Evaraert in Polymer blends handbook, ed. L. A. Utracki, Kluwer Academic Pub, The Netherlands, 2nd edn, 2002, 2, ch. 3, pp. 203–289 Search PubMed.
- M. L. Xue, Y. L. Yu, H. H. Chuah and G. X. Qiu, J. Macromol. Sci., Part B: Phys., 2007, 46, 387–401 CrossRef CAS.
- M. L. Xue, Y. L. Yu, J. Sheng, H. H. Chuah and C. H. Geng, J. Macromol. Sci., Part B: Phys., 2005, 44, 317–329 CrossRef CAS PubMed.
- M. L. Xue, Y. L. Yu and H. H. Chuah, J. Macromol. Sci., Part B: Phys., 2007, 46, 603–615 CrossRef CAS.
- P. Huang, Z. Zhong, S. Zheng, W. Zhu and Q. Guo, J. Appl. Polym. Sci., 1999, 73, 639–647 CrossRef CAS.
- H. Zhang, M. Ren, Q. Chen, S. Sun, H. Zhang and Z. Mo, J. Polym. Sci., Part B: Polym. Phys., 2006, 44, 1320–1330 CrossRef CAS.
- H. H. Chuah, Macromolecules, 2001, 34, 6985–6993 CrossRef CAS.
- M. Yamen, S. Ozkaya and N. Vasanthan, J. Polym. Sci., Part B: Polym. Phys., 2008, 46, 1497–1504 CrossRef CAS.
- I. Aravind, K. J. Eichhorn, H. Komber, D. Jehnichen, N. Zafeiropoulos, K. H. Ahn, Y. Grohens, M. Stamm and S. Thomas, J. Phys. Chem. B, 2009, 113, 1569–1578 CrossRef CAS PubMed.
- L. Utracki, Adv. Polym. Technol., 1985, 5, 33–39 CrossRef.
- K. Frisch, D. Klempner and H. Frisch, Polym. Eng. Sci., 1982, 22, 1143–1152 CAS.
- T. Fox, Bull. Am. Phys. Soc., 1956, 1, 123–123 CAS.
- P. Couchman, Macromolecules, 1980, 13, 1272–1276 CrossRef CAS.
- M. Gordon and J. S. Taylor, J. Appl. Chem., 1952, 2, 493–500 CrossRef CAS.
- Q. Guo, C. Harrats, G. Groeninckx and M. Koch, Polymer, 2001, 42, 4127–4140 CrossRef CAS.
- M. Aubin and R. E. Prud'Homme, Macromolecules, 1988, 21, 2945–2949 CrossRef CAS.
- K. Yamada, M. Hikosaka, A. Toda, S. Yamazaki and K. Tagashira, Macromolecules, 2003, 36, 4790–4801 CrossRef CAS.
- H. Marand, J. Xu and S. Srinivas, Macromolecules, 1998, 31, 8219–8229 CrossRef CAS.
- T. Nishi and T. Wang, Macromolecules, 1975, 8, 909–915 CrossRef CAS.
- T. Nishi, T. Wang and T. Kwei, Macromolecules, 1975, 8, 227–234 CrossRef CAS.
- H. H. Chuah, Macromolecules, 2001, 34, 6985–6993 CrossRef CAS.
- W.-a. Luo, Y. Chen, X. Chen, Z. Liao, K. Mai and M. Zhang, Macromolecules, 2008, 41, 3912–3918 CrossRef CAS.
- A. Synytska, S. Michel, D. Pleul, C. Bellmann, R. Schinner, K. J. Eichhorn, K. Grundke, A. Neumann and M. Stamm, J. Adhes., 2004, 80, 667–683 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) n) of 22
n) of 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 g mol−1 and a polydispersity index (PDI) of 2.5 was kindly supplied by DuPont Industries, USA. Details of the materials used are given in Table 1, and the chemical structures are shown in Fig. 1.
500 g mol−1 and a polydispersity index (PDI) of 2.5 was kindly supplied by DuPont Industries, USA. Details of the materials used are given in Table 1, and the chemical structures are shown in Fig. 1.
![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) wa (g mol−1)
wa (g mol−1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400
400![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) na (g mol−1)
na (g mol−1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500
500