Mechanistic insights into operational lithium–sulfur batteries by in situ X-ray diffraction and absorption spectroscopy†
Michael A. Lowe,
Jie Gao and
Héctor D. Abruña*
Department of Chemistry and Chemical Biology, Baker Laboratory, Cornell University, Ithaca, NY 14853, USA. E-mail: hda1@cornell.edu
First published on 7th April 2014
Abstract
The lithium–sulfur battery is an extremely attractive system for electrical energy storage because of its exceptional theoretical capacity and energy density. However, the practical values typically obtained are much lower and inherently determined by the complex chemistry of reduced sulfur species. The lack of methods to probe sulfur species under realistic battery conditions has frustrated chemical understanding and control. We have employed in situ X-ray diffraction (XRD) and sulfur K-edge X-ray absorption near edge spectroscopy (XANES) to probe the sulfur intermediates and products formed in battery electrodes during operation of prototype lithium–sulfur batteries. Correlations between the X-ray and electrochemical data show that the reduction of sulfur to lithium sulfide is mediated through dissociation and disproportionation reactions of a few dominant sulfur species. Deliberate control of these chemical equilibria is essential to approach the theoretical capacity of the lithium–sulfur system.
Introduction
Electrochemical energy storage often involves a tradeoff between chemical complexity and theoretical capacity. Current lithium-ion battery cathodes store charge via ion intercalation into crystalline structures. This results in a theoretical capacity limited by the mass of the crystalline host (e.g. CoO2, FePO4) and, in general, no more than one electron is stored per formula unit. In addition, the practical capacities reached in devices is often well below theoretical values. On the other hand, the most promising chemical systems for future energy storage involve reactions between lithium and molecules of electronegative elements (e.g. S8, O2) that can access multiple electrons per formula unit, promising dramatic gains in capacity (∼10× that of existing lithium-ion batteries) albeit at the cost of increased mechanistic complexity and chemical changes during the reaction. The increased chemical complexity that comes with a shift towards molecular charge storage portends a need for new and chemically-informed approaches to battery design, testing, and characterization.The lithium–sulfur cell system represents an especially attractive and promising case due to the combination of sulfur's reduction potential (>2 V vs. Li/Li+), extraordinary theoretical gravimetric capacity (16 electrons per S8, or 1672 mA h g−1), and condensed phase. However, its full theoretical capacity has not yet been accessed reversibly due to the complex chemistry and mechanistic interplay between lithium and polysulfides. While various mechanisms for sulfur reduction have been proposed based on parameterized models,1 electrochemical probes within batteries,2–4 or electroanalytical studies,5–7 the diversity of possible reactions, combined with the paucity of data on specific intermediates, means that many of the proposed mechanisms are speculative and inconsistent. Additional experimental data, especially under realistic battery operating conditions, about reduced sulfur species are necessary to resolve previous analytical and battery-level studies and provide rational guidelines for approaching the full capacity from the lithium–sulfur system.
The complexity of the sulfur mechanism has been the subject of a number of analytical studies,5–9 although most have pursued the reaction mechanism in solvents that are not appropriate for lithium batteries (e.g. N,N-dimethylformamide; DMF)5,6,10–12 and at much lower, electroanalytical concentrations.6,8,10,13–15 These in situ spectroelectrochemical studies support the following outline of the sulfur reduction mechanism, at least at low sulfur concentrations:7
• Elemental sulfur is initially reduced to form S8− and then S82−.
• S82− rapidly equilibrates to yield several different polysulfides (Sn2−), including S62−.
• S82− and S62− dissociate to form S4− and S3−, respectively, with S3− as the predominant species.
• Only the radical anions S3− and S4− are subsequently reduced in a second electrochemical process. Depending on the solvent, this process is 300–800 mV more negative than the first reduction process.16
The dissociative equilibria between and among polysulfides and the electrochemically active radical anion S8,9 emphasize the need for studies under technologically-relevant conditions of solvent, electrolyte and concentrations. Moreover, the research at electroanalytical concentrations has not probed the appearance of fully-reduced lithium sulfide, which, as the final reduction product, is of utmost importance for lithium–sulfur batteries.
On the other hand, researchers studying lithium–sulfur batteries have proposed various mechanisms which involve the same general outline: sparingly soluble elemental sulfur is first reduced to soluble polysulfides (Sn2−, n > 2) and subsequently to insoluble lower polysulfides (Li2S2 or Li2S).17–22 While the first step is in general agreement with electroanalytical studies, the second reaction is inferred from observed cell capacities,17 electrochemical impedance measurements4 and ex situ diffraction.3,21 Inadequate experimental data on sulfur reduction products are a major obstacle to further understand and control the failure modes of lithium–sulfur cells. For example, insoluble Li2S2 has been proposed to be a major discharge product and the cause of poor cyclability (i.e. rapid capacity fade)17,18,21–23 even though there are no reports of its direct observation. Mechanistically, it is still unclear whether the reduction of polysulfides occurs sequentially1 (i.e. higher polysulfides are reduced before lower polysulfides) or concurrently,24 and the relative importance of mediated reductions25 (e.g. disproportionations) and direct electrochemical reduction.24
The lack of data on reduced sulfur species in lithium–sulfur batteries derives, in large part, from the difficulty of characterizing the air-sensitive ensemble of partially-soluble, partially-reduced, and partially-crystalline sulfur species within the concentration and volumetric constraints of typical prototype cells. While the complexity of the system is imposing, X-ray diffraction and absorption spectroscopy can provide the respective phase and elemental specificity to resolve both crystalline and chemical changes during cell charge/discharge cycles. Moreover, the relatively weak interaction of high energy photons with matter (i.e., high penetration depth) means that these techniques provide a bulk-sensitive response and can probe the changes of sulfur within sealed batteries.
Herein, we present in situ XRD and sulfur K-edge XANES results from the first discharge/charge cycle of operational lithium–sulfur batteries. The electrochemical features are correlated with time-dependent XRD from the crystalline end products, and XANES from all sulfur species present in the battery. Together, the data show that the reduction of sulfur to lithium sulfide proceeds primarily through disproportionations of polysulfides, with specific sulfur species playing a dominant role in the overall mechanism.
Experimental
a. Battery preparation
A sulfur–carbon composite was prepared by physically mixing elemental sulfur and carbon black (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 by mass), transferring the mixture into a Teflon-lined steel autoclave, and heating for 10 h at 155 °C. Elemental sulfur electrodes were prepared by grinding the sulfur-impregnated carbon black composite and polyvinylene difluoride (PVDF) (95
45 by mass), transferring the mixture into a Teflon-lined steel autoclave, and heating for 10 h at 155 °C. Elemental sulfur electrodes were prepared by grinding the sulfur-impregnated carbon black composite and polyvinylene difluoride (PVDF) (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 wt%) with N-methyl-pyrrolidone in a mortar and pestle and spreading the slurry with a doctor's blade. The film was then dried in vacuum for 12 hours, weighed, and stored in an argon-filled glovebox until use. For this work, films were cast on a carbon-coated aluminum mesh current collector to minimize attenuation of the lower-energy X-rays necessary for X-ray absorption spectroscopy at the sulfur K edge. Typical sulfur loadings were 1–2 mg cm−2.
5 wt%) with N-methyl-pyrrolidone in a mortar and pestle and spreading the slurry with a doctor's blade. The film was then dried in vacuum for 12 hours, weighed, and stored in an argon-filled glovebox until use. For this work, films were cast on a carbon-coated aluminum mesh current collector to minimize attenuation of the lower-energy X-rays necessary for X-ray absorption spectroscopy at the sulfur K edge. Typical sulfur loadings were 1–2 mg cm−2.
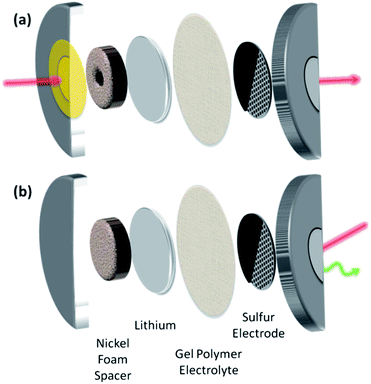
Prototype lithium–sulfur cells were fabricated using modified CR2032 coin cell casings. 3 mm diameter holes were drilled through the casings, and kapton windows were epoxied around the holes (Fig. 1). For XRD measurements, 100 μm thick windows were used on both anode and cathode casings to minimize atmospheric contamination (Fig. 1a), while the XANES study used an unmodified anode casing and a 8 μm thick kapton window on only the cathode casing to minimize photon attenuation at the sulfur K-edge (2472 eV) (Fig. 1b). A 70 nm thick film of aluminum was deposited on both sides of all kapton windows to reduce air and moisture diffusion through the windows while providing good electrical contact.
Cells were prepared using lithium anodes and a film of poly(vinylidene fluoride-co-hexafluoropropene) (PVDF-HFP) saturated with 1 M LiClO4 in tetra(ethylene glycol) dimethyl ether as a gel polymer electrolyte. The PVDF-HFP film was prepared as previously reported by Tarascon et al.26 All cells were stored in hermetically-sealed, argon-filled vials until probed using synchrotron X-ray radiation, and characterized within 4 days of preparation to prevent significant self-discharge.
b. X-ray measurements
Diffraction data were acquired in a transmission geometry with 30 keV incident radiation at beamline A2 of the Cornell High Energy Synchrotron Source (CHESS). Diffraction was collected using a 2D detector (GE), then integrated to obtain 1D diffraction patterns using Fit2D.27XAS spectra were collected at beamline X19A of the National Synchrotron Light Source (NSLS). Batteries were cycled in a He-purged sample chamber, with X-ray fluorescence detected using a large area passivated implanted planar silicon detector (Canberra Industries, Inc.). Datasets were background subtracted and normalized using the Athena software package.28 Care was taken to limit the duration of X-ray exposure to less than 10% of the battery operation, since extended irradiation of the battery at energies above the absorption edge was found to alter the electrochemical and spectroscopic response. This is a consequence of irradiating electronically isolated (solvated) sulfur species in solution, since no spectroscopic evidence for radiation damage was found using solid-phase standards (elemental sulfur or lithium sulfide) or ex situ electrodes.
Results
a. In situ X-ray diffraction
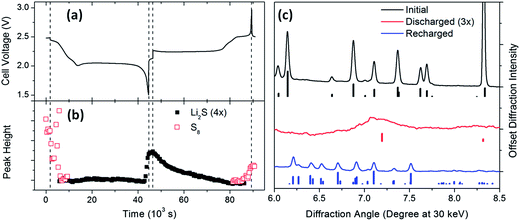
In contrast to other chalcogen-alkali metal phase diagrams, the only crystalline phases in the lithium–sulfur system are the several allotropes of elemental sulfur (S8) and the fully reduced cubic lithium sulfide (Li2S).29,30 However, correlating the electrochemical response with the presence and relative abundance of these phases can have profound implications for the overall reaction mechanism and, consequently, battery performance.Fig. 2 presents typical diffraction data during the first galvanostatic discharge/charge cycle of an operational lithium–sulfur battery prepared with a gel polymer electrolyte at a current rate of 100 mA g−1 (∼C/17). As seen in Fig. 1a and b, the integrated diffraction intensity from orthorhombic elemental sulfur remained essentially constant until the discharge begins, at which point the integrated peak height decreased with discharge time. The scatter in the data is a consequence of dynamic interactions between crystalline and low concentrations of dissolved sulfur, as can be seen in the raw two-dimensional diffraction data that show individual peaks temporarily appearing or increasing in intensity during this time period. (ESI S1†) Midway through the first discharge plateau, all crystallinity disappeared, consistent with the proposed reduction of elemental sulfur to an amorphous polysulfide.
Diffraction from Li2S is not detected until the cell voltage drops below 2 V, at which point the diffraction peak rapidly grows until the end of the discharge. Previous XRD studies of partially-discharged sulfur cathodes reported crystalline Li2S earlier in the discharge,3,31 although the cathodes were probed ex situ and there were significant differences in battery preparation, including the nature and quantity of the electrolyte and the impregnation of the sulfur onto the carbon. Interestingly, a recent report of in situ XRD on operational lithium–sulfur batteries did not observe the formation of any Li2S with several different cathode preparations,32 emphasizing the need for systematic study of how crystallization depends on cell parameters. As seen in Fig. 2c, the average Li2S domain is significantly smaller than the average elemental sulfur domain (7 nm vs. >60 nm, respectively, as determined from the Scherrer equation). The abrupt appearance of crystalline Li2S at the end of the voltage plateau suggests that its formation is due to the reaction mechanism discussed below (vide infra) rather than to a kinetically-limited crystallization of amorphous Li2S.
After maintaining its intensity during open circuit equilibration, the diffraction from crystalline Li2S slowly, but steadily, decreased through the recharging process (Fig. 2b). By the time the recharge reached the upper voltage plateau at 2.5 V, no crystalline lithium sulfide was evident, demonstrating that under these conditions crystalline lithium sulfide does not accumulate. At the end of the final plateau, elemental sulfur is formed, and crystallizing in the monoclinic structure (Fig. 2c). Although orthorhombic sulfur is thermodynamically favored, previous researchers have reported precipitation of monoclinic sulfur nanoparticles at room temperature.33 Upon extended equilibration (>2 h) the crystallinity disappears completely, ostensibly due to self-discharge reactions.
b. In situ X-ray absorption
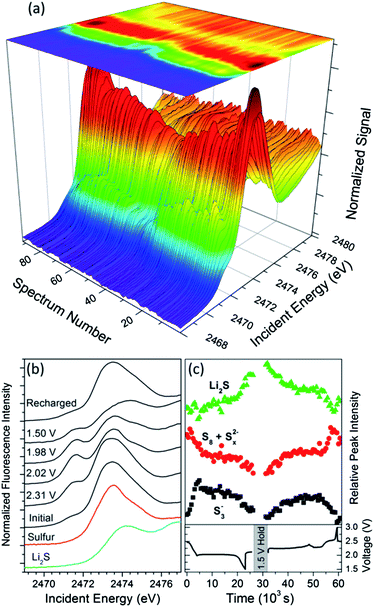
There are limited reports of XAS from polysulfides in solution or in the solid state, due to the competing constraints of the ‘tender’ incident X-ray energy for of the sulfur K edge and the air-sensitivity of polysulfides, especially Li2S. An additional complication is the lack of single-phase polysulfide standards between elemental sulfur and lithium sulfide, since a reasonable solution of any single polysulfide Sn2− will rapidly equilibrate into several adjacent polysulfides Sn−12− and Sn+12−.7,9 Thus, an in situ study of sulfur reduction species is required to obtain truly representative and relevant data on the system.Fig. 3a presents a complete XAS dataset for the discharge and charge of a lithium–sulfur battery with a gel polymer electrolyte. The apparent symmetry of the dataset emphasizes that discharging and charging proceed through a reversible sequence of reactions. Several distinct features evolve through the cycle and are emphasized in the selected spectra shown in Fig. 3b. The relative peak intensities, as indicated by the derivative of the spectra, are plotted in Fig. 2c to correlate XAS and electrochemical features. Below we discuss the trends and propose tentative assignments to various sulfur species.
In the charged state, the spectrum from the sulfur cathode matches that of elemental sulfur (Fig. 3b). At the start of the discharge, there is a distinct increase in the main peak intensity (red data in Fig. 3c) due to the formation of long-chain polysulfides (Sn2−, n > 7).34 Under electroanalytical conditions, cyclic S8 is reduced by two electrons to form an S82− chain at potentials, versus lithium, that are very similar to the voltage of the first discharge plateau.7 It is expected that disproportionation reactions will rapidly give rise to a distribution of long-chain polysulfides35 which have XANES signals that are too similar for spectroscopic speciation/differentiation.
At the end of the first voltage plateau, the intensity of the first peak decreases and a new pre-peak appears (2.31 V spectrum in Fig. 3b and black data in Fig. 3c). A similarly-downshifted peak has been reported from solid-state compounds and assigned to the 1s–3p(π*) transition for S3− radical anions.36 The spectra, in the first part of the transition period, show two clear isosbestic points at 2472.2 and 2475.5 eV, consistent with a simple conversion between two species (ESI S2†). Thus, we propose that the pre-peak is from S3− radical anions formed by dissociation of longer-chain polysulfides. Sulfur electrochemical data have indicated that these equilibria are fast and favor radical anion formation.10 The appearance of S3− is also consistent with several spectroelectrochemical studies at electroanalytical concentrations.10,12,13
As the cell voltage continues to decrease to the second plateau, the main peak also shifts to slightly higher energies (2.02 V spectrum in Fig. 3b). We propose that this shift is related to a decrease in the average polysulfide chain length (i.e. disproportionations to form Sx2−, 4 < x < 8). Charge is more localized for shorter polysulfides, which permits more effective screening by lithium counter-ions and a hypsochromic shift in the position of the main peak. For example, the main peak in the sulfur XANES of lithium sulfide is shifted nearly a full eV to higher energy relative to elemental sulfur (Fig. 3b).
Through the lower voltage plateau, the spectra are dominated by the pre-peak and the shifted main peak (2.02 and 1.98 V spectra in Fig. 3b). The invariance in the peak positions suggests that they are due to a specific set of sulfur species, such as S3− and S62−. Both peak intensities slowly diminish in amplitude during the voltage plateau (Fig. 3c) as S3− and, by equilibration, S62− are electrochemically reduced and consumed. This is consistent with previous electroanalytical reports that have shown that the second (more negative) electrochemical process is due to the S3−/S32− couple.10,16 It is notable that the same two peaks persist while passing more than 400 mA h g−1 in the discharge, even though the theoretical capacity for the one electron reduction of S3− is only 278 mA h g−1. There is also no apparent signal from the reaction product of the second plateau, a consequence of forming multiple or short-lived polysulfide species that do not contribute a single XANES feature. Together, these observations suggest that the second set of reduction processes proceed, not through a sequential reduction of progressively more reduced polysulfides, but rather via dissociation of long-chain polysulfides to form the electroactive S3−, followed by reaction of the reduction product (S32−)5,25 with longer polysulfides. It is likely that the polysulfides formed by conproportionation can subsequently dissociate and partially replenish the oxidized species, S3−, extending the length (duration) of the second discharge plateau. Typical reactions may include:
| S3− + e− → S32− | (1) |
| S32− + Sn2− ↔ S62− + Sn−32− (3 < n < 8) | (2) |
| S62− ↔ 2S3− | (3) |
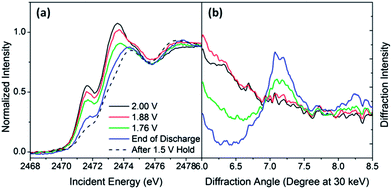
At the end of the lower voltage plateau, there is a concerted decrease in cell voltage and the peak intensities, as seen in the trends of Fig. 3c and the XANES spectra in Fig. 3a. This is consistent with the assignment of the pre-peak to the dominant electroactive species (S3−) during the second voltage plateau. A new peak at higher energies, which matches the first XANES peak of the Li2S standard (Fig. 3b), becomes distinct as abruptly and at the same cell voltages as the diffraction peak from Li2S (Fig. 4b). This observation conclusively confirms that Li2S is formed at the end of discharge for batteries with a gel polymer electrolyte. Since the XANES spectra do not exhibit any clear isosbestic points or intermediate peaks, there must be additional disproportionation steps involved in the formation of lithium sulfide. It is probable that after depletion of the longer polysulfides, an alternate reaction mechanism becomes operative, such as:
| S32− + Li2Sn ↔ Li2S + Sn+22− (n = 2, 3) | (4) |
After the cell voltage is held at 1.5 V for 1 hour, the intensity over the range 2471–2474 eV further decreases relative to the Li2S signal (Fig. 3c), supporting the conclusion that signals over this energy range are due to incompletely reduced polysulfide species.
During recharging, the same spectroscopic signals are observed but in reverse sequence, suggesting that the same key species are involved in the process of sulfide oxidation. This is in contrast to models proposed in the literature,37 again pointing to the need of in situ studies, but emphasizes the chemical reversibility of the reaction pathway. There are a few notable differences in the relative XAS trends during reduction (discharge) and oxidation (charge). In particular, Fig. 3c shows that the signal from Li2S decreases much more slowly during charging than it appeared during discharging, in excellent agreement with the trend in diffraction intensity from Li2S (Fig. 1b).
Discussion
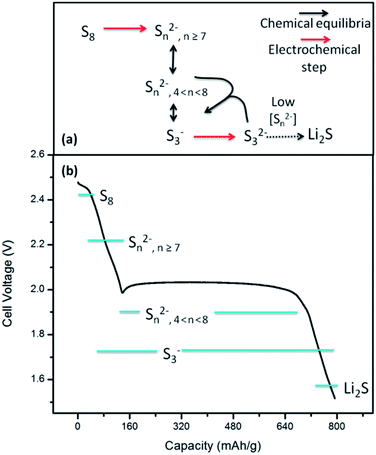
The above spectroscopic observations support a sulfur reduction mechanism primarily defined and controlled by disproportionation reactions. Fig. 5 summarizes the data available through this in situ XRD and XAS study and outlines a proposed mechanism for sulfur reduction in a Li/S battery with a gel polymer electrolyte. In the upper voltage plateau, elemental sulfur (cyclic S8) is reduced and reacts to form longer chain polysulfides. These polysulfides dissociate to form a mix of radical anions and shorter-chain polysulfides during the transition between the voltage plateaus, and are further reduced through the lower voltage plateau by the electrochemical mediation of S3− radical anions and equilibration with more reduced polysulfides (e.g. S32−). Once the concentration of long polysulfides is too low for this reaction to sustain a threshold concentration of radical anions, the cell voltage drops and the lower polysulfides disproportionate to form crystalline lithium sulfide (Fig. 3b).Chemical equilibria between/among polysulfides enable the reaction to approach chemical reversibility under the appropriate conditions, as is apparent in the sulfur XANES of Fig. 3a. However, they also limit the practical discharge capacity of the battery if the chemical equilibria maintain a distribution of incompletely reduced polysulfides. A key point is that chemical equilibria are governed by relative concentrations (mol%), not by the relative mass fraction, and for a given mass fraction a shorter-chain polysulfide will always have a higher molar concentration than a longer-chain polysulfide. This difference in concentrations constitutes a thermodynamic driving force for obtaining a distribution of polysulfide species even at the end of battery discharge. These results identify multiple points in the sulfur reaction mechanism where chemical equilibria play a key role, including the transition from longer-chain polysulfides (e.g. S82−) to shorter-chain polysulfides and radical anions, and in the final formation of lithium sulfide from S32−. The possible effect of the equilibria on the cell capacity is demonstrated by the scenarios outlined in Table 1. In the ideal case, all sulfur is converted to lithium sulfide to obtain the full 1672 mA h g−1 capacity. However, if even 1 mol% of the sulfur species is ‘trapped’ as S82−, the accessible capacity drops by over 100 mA h g−1 (>6% of the full capacity). Based on the XANES spectra of the discharged battery in Fig. 3a, in a more reasonable scenario there are non-negligible concentrations of multiple sulfur species at the end of the first discharge, including at least S62− and S3−. Assuming that reasonable concentrations of these species persist, only 64% of the ideal capacity is possible, even with a high relative concentration of Li2S. These illustrative examples predict achievable capacities that are significantly more than what we observed in this report, but are in line with the observed first discharge capacities of the best reported lithium–sulfur cells.18,38–40 Thus, the presence of sulfur as electrochemically-inactive polysulfides can reasonably account for a significant fraction of the discrepancy from theoretical capacities.
| Scenario | Relative species concentration (% mol/% mass sulfur) | Accessible capacity (mA h g−1) | |||
|---|---|---|---|---|---|
| S82− | S62− | S3− | Li2S | ||
| 1 | 0 | 0 | 0 | 100/100 | 1672 |
| 2 | 1/7.4 | 0 | 0 | 99/92.5 | 1562 |
| 3 | 1/5.3 | 5/19.7 | 10/19.7 | 84/55.3 | 1044 |
Based on the proposed reaction mechanism, sulfur can be most fully and reversibly reduced under conditions in which there is a high concentration of sulfur species within the diffusion layer region of the electrode. Under these conditions, chemical equilibria facilitate formation of electroactive radical anions and the corresponding dianions for the respective reduction and oxidation reactions. This agrees with several reports of promising battery performance using sulfur incorporated into mesoporous carbons,18,38,40 which limit the diffusion of sulfur into solution and maintain high local polysulfide concentrations. Further, the proposed mechanism highlights the possible strategy of altering chemical equilibria to favor formation of the electroactive sulfur species, especially S3−, in order to maximize the available capacity from elemental sulfur.
Conclusions
This work presents the first in situ XRD and XAS study of sulfur in a Li/S battery environment, demonstrating the utility of these techniques to probe the key species involved in lithium–sulfur battery operation and emphasizing the need for an in situ molecular understanding of sulfur reduction pathways. Additional and ongoing efforts are necessary to probe the effects of specific cell parameters (e.g. electrolytes, carbons) on the reaction, as well as to collect and theoretically model absorption spectra for reduced sulfur species in support of the assignments discussed above. However, this research clearly shows that a limited number of sulfur species predominate through the cycle of a sulfur cathode and predicts that altering/controlling chemical equilibria to favor the formation of specific polysulfides can dramatically improve the capacity of lithium–sulfur cells.Acknowledgements
This work was supported by Fuji Heavy Industries, Ltd. MAL is supported by a National Defense Science and Engineering Graduate Fellowship. This work made use of the Cornell Center for Materials Research Facilities, supported by the National Science Foundation under Award Number DMR-0520404. Use of the National Synchrotron Light Source, Brookhaven National Laboratory, was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, under Contract no. DE-AC02-98CH10886. Use of the Cornell High Energy Synchrotron Source (CHESS) was supported by the National Science Foundation and the National Institutes of Health/National Institute of General Medical Sciences under award DMR-0225180. The authors acknowledge useful discussions with CHESS staff scientists Darren Dale and (the late) Alexander Kazimirov.Notes and references
- K. Kumaresan, Y. Mikhaylik and R. E. White, J. Electrochem. Soc., 2008, 155, A576–A582 CrossRef CAS PubMed.
- R. Dominko, R. Demir-Cakan, M. Morcrette and J.-M. Tarascon, Electrochem. Commun., 2011, 13, 117–120 CrossRef CAS PubMed.
- L. Yuan, X. Qiu, L. Chen and W. Zhu, J. Power Sources, 2009, 189, 127–132 CrossRef CAS PubMed.
- V. S. Kolosnitsyn, E. V. Kuzmina, E. V. Karaseva and S. E. Mochalov, J. Power Sources, 2011, 196, 1478–1482 CrossRef CAS PubMed.
- E. Levillain, F. Gaillard and J. P. Lelieur, J. Electroanal. Chem., 1997, 440, 243–250 CrossRef CAS.
- A. Evans, M. I. Montenegro and D. Pletcher, Electrochem. Commun., 2001, 3, 514–518 CrossRef CAS.
- P. Leghie, J.-P. Lelieur and E. Levillain, Electrochem. Commun., 2002, 4, 406–411 CrossRef CAS.
- R. P. Martin, W. H. Doub Jr, J. L. Roberts Jr and D. T. Sawyer, Inorg. Chem., 1973, 12, 1921–1925 CrossRef CAS.
- R. Steudel, Top. Curr. Chem., 2003, 127–152 CrossRef CAS.
- F. Gaillard, E. Levillain and J. P. Lelieur, J. Electroanal. Chem., 1997, 432, 129–138 CrossRef CAS.
- F. Gaillard, E. Levillain, M.-C. Dhamelincourt, P. Dhamelincourt and J. P. Lelieur, J. Raman Spectrosc., 1997, 28, 511–517 CrossRef CAS.
- D.-H. Han, B.-S. Kim, S.-J. Choi, Y. Jung, J. Kwak and S.-M. Park, J. Electrochem. Soc., 2004, 151, E283 CrossRef CAS PubMed.
- B.-S. Kim and S.-M. Park, J. Electrochem. Soc., 1993, 140, 115–122 CrossRef CAS PubMed.
- M. V. Merritt and D. T. Sawyer, Inorg. Chem., 1970, 9, 211–215 CrossRef CAS.
- S. Tobishima, H. Yamamoto and M. Matsuda, Electrochim. Acta, 1997, 42, 1019–1029 CrossRef CAS.
- Y. Jung, S. Kim, B.-S. Kim, D.-H. Han, S.-M. Park and J. Kwak, Int. J. Electrochem. Sci., 2008, 3, 566–577 CAS.
- E. Peled, Y. Sternberg, A. Gorenshtein and Y. Lavi, J. Electrochem. Soc., 1989, 136, 1621–1625 CrossRef CAS PubMed.
- X. Ji, K. T. Lee and L. F. Nazar, Nat. Mater., 2009, 8, 500–506 CrossRef CAS PubMed.
- V. S. Kolosnitsyn, E. V. Karaseva, D. Y. Seung and M. D. Cho, Russ. J. Electrochem., 2002, 38, 1314–1318 CrossRef CAS.
- J. Shim, K. A. Striebel and E. J. Cairns, J. Electrochem. Soc., 2002, 149, A1321–A1325 CrossRef CAS PubMed.
- S.-E. Cheon, K.-S. Ko, J.-H. Cho, S.-W. Kim, E.-Y. Chin and H.-T. Kim, J. Electrochem. Soc., 2003, 150, A800–A805 CrossRef CAS PubMed.
- H. Yamin, A. Gorenshtein, J. Penciner, Y. Sternberg and E. Peled, J. Electrochem. Soc., 1988, 135, 1045–1048 CrossRef CAS PubMed.
- B. H. Jeon, J. H. Yeon, K. M. Kim and I. J. Chung, J. Power Sources, 2002, 109, 89–97 CrossRef CAS.
- V. S. Kolosnitsyn and E. V. Karaseva, Russ. J. Electrochem., 2008, 44, 506–509 CrossRef CAS.
- Y. Jung and S. Kim, Electrochem. Commun., 2007, 9, 249–254 CrossRef CAS PubMed.
- A. Du Pasquier, P. C. Warren, D. Culver, A. S. Gozdz, G. G. Amatucci and J. M. Tarascon, Solid State Ionics, 2000, 135, 249–257 CrossRef CAS.
- A. P. Hammersley, S. O. Svensson, M. Hanfland, A. N. Fitch and D. Häusermann, High Pressure Res., 1996, 14, 235–248 CrossRef.
- B. Ravel and M. Newville, J. Synchrotron Radiat., 2005, 12, 537–541 CrossRef CAS PubMed.
- R. A. Sharma, J. Electrochem. Soc., 1972, 119, 1439–1443 CrossRef CAS PubMed.
- H. Okamoto, J. Phase Equilib., 1995, 16, 94–97 CrossRef CAS.
- H. S. Ryu, H. J. Ahn, K. W. Kim, J. H. Ahn and J. Y. Lee, J. Power Sources, 2006, 153, 360–364 CrossRef CAS PubMed.
- J. Nelson, S. Misra, Y. Yang, A. Jackson, Y. Liu, H. Wang, H. Dai, J. C. Andrews, Y. Cui and M. F. Toney, J. Am. Chem. Soc., 2012, 134, 6337–6343 CrossRef CAS PubMed.
- Y. Guo, J. Zhao, S. Yang, K. Yu, Z. Wang and H. Zhang, Powder Technol., 2006, 162, 83–86 CrossRef CAS PubMed.
- B. Eckert and R. Steudel, Top. Curr. Chem., 2003, 181–191 Search PubMed.
- R. D. Rauh, F. S. Shuker, J. M. Marston and S. B. Brummer, J. Inorg. Nucl. Chem., 1977, 39, 1761–1766 CrossRef CAS.
- M. E. Fleet and X. Liu, Spectrochim. Acta, Part B, 2010, 65, 75–79 CrossRef PubMed.
- X. Ji and L. F. Nazar, J. Mater. Chem., 2010, 20, 9821–9826 RSC.
- C. Liang, N. J. Dudney and J. Y. Howe, Chem. Mater., 2009, 21, 4724–4730 CrossRef CAS.
- X. Liang, Z. Wen, Y. Liu, H. Zhang, L. Huang and J. Jin, J. Power Sources, 2011, 196, 3655–3658 CrossRef CAS PubMed.
- N. Jayaprakash, J. Shen, S. S. Moganty, A. Corona and L. A. Archer, Angew. Chem., Int. Ed., 2011, 50, 1–6 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: A video of 2D diffraction images from lithium sulfur batteries and a plot of spectra during transition between first and second discharge plateaus. See DOI: 10.1039/c4ra01388c |
| This journal is © The Royal Society of Chemistry 2014 |