Designed hierarchical MnO2 microspheres assembled from nanofilms for removal of heavy metal ions
Abstract
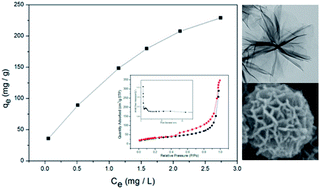
Hierarchical MnO2 microspheres assembled from nanofilms are fabricated through an environmental route and subsequent drying under vacuum. Adsorption kinetics and thermodynamics are investigated in detail. The unique nanofilm assembly structure with high BET surface area of 252.82 m2 g−1 can allow for higher exposure of adsorption sites to adsorbate molecules than a solid one, and thus results in the high removal efficiency of heavy metal ions. The final removal efficiency of Pb(II), Cd(II), Cu(II) and Cr(VI) from acid aqueous solutions can reach 100%, 99.6%, 99.1% and 95.2%, respectively. Moreover, the relatively large submicrometer particles are easy to recover after adsorption. Therefore, it may serve as an ideal candidate for heavy metal ion removal in water treatment.

 Please wait while we load your content...
Please wait while we load your content...