Synthesis, characterization and p–n type gas sensing behaviour of CuFeO2 delafossite type inorganic wires using Fe and Cu complexes as single source molecular precursors†
Julia Patzscha,
Ildiko Baloga,
Peter Kraußa,
Christian W. Lehmannb and
Jörg J. Schneider*a
aFachbereich Chemie, Eduard-Zintl-Institut für Anorganische und Physikalische Chemie, Technische Universität Darmstadt, Alarich-Weiss-Str. 12, 64287 Darmstadt, Germany. E-mail: joerg.schneider@ac.chemie.tu-darmstadt.de; Fax: +49-6151-163470; Tel: +49-6151-16-3225
bMax-Planck-Institut für Kohlenforschung, Chemische Kristallographie, Kaiser-Wilhelm-Platz 1, 45470 Mülheim an der Ruhr, Germany
First published on 17th March 2014
Abstract
CuFeO2 delafossite type nanowires with a diameter of 180 nm were successfully synthesized by electrospinning using an equimolar mixture of molecular copper and iron precursors and polyacrylnitrile as polymer template followed by calcination. This mixture was electrospun under ambient conditions. The copper and iron precursor complexes diaqua-bis[2-(methoxyimino)-propanato] M(II) (M = Cu, Fe) were fully characterized by spectroscopic and electrochemistry methods (NMR, IR, cyclovoltammetry) as well as by single crystal structure and by thermogravimetric analysis (TGA). The pre-ceramic electrospun composite fiber mats were subsequently heat treated in a two step procedure to yield the final CuFeO2 delafossite type nanowires agglomerated as microsized ceramic mats. Intermediate oxide phases as well as the finally obtained CuFeO2 fibrous mats composed of delafossite type nanowires were characterized analytically by temperature dependent powder X-ray crystallography (PXRD). Their topological morphology was studied by scanning electron microscopy (SEM). The obtained CuFeO2 fibrous mats were studied with respect to their O2 gas sensing behaviour and variable p and n-type semiconducting behaviour depending on temperature was observed.
1 Introduction
Delafossites have been studied intensively in the last decade since the discovery that CuAlO2 is a good p-type transparent conducting oxide (TCO).1 Such TCO materials have a wide range of applications in photovoltaics or flat panel displays. In comparison to n-type materials p-type TCOs are rather rare but are interesting for the development of transparent p–n junction-based devices like solar cells or light emitting diodes. Delafossites obey the general chemical composition MIMIIIO2 and can be divided in two groups depending on the monovalent cations MI displaying Ag+ and Cu+ showing p-type behavior or containing Pt+ and Pd+ displaying metallic conductivity.2 MIII is a trivalent cation, e.g. Al3+, Fe3+ or Cr3+. Due to their functional properties copper delafossites of general formula Cu(I)M(III)O2 are most interesting because they show a wide range of electrical conductivities while often obeying optical transparency.3 Indeed CuFeO2 has the highest electrical conductivity of all delafossites when an off-stoichiometric ratio CuFeO2+δ phase is maintained.3e,4 This behavior can even be improved by doping the phase composition with magnesium or chromium.3e,5 To tailor the optoelectronic properties, delafossite films have been prepared by different techniques like pulsed laser deposition on glass6 or radio-frequency (RF) sputtering7 on glass substrates. As alternative to these more expensive processes, different kinds of chemical routes for CuFeO2 synthesis were developed8 which provide advantages like a straightforward synthetic access to delafossites with even higher surface areas.9 Moreover, related studies on delafossites have shown that besides their transparent properties, their direct band gap transition of 1.3 V, is ideal for sunlight conversion as well as for the reduction of Cd2+ over CuFeO2 under visible light in an aqueous systems.10 Due to the high energetic position of the conduction band, generation of photoelectrons with a strong reducing power is possible and their long term chemical stability in neutral solutions thus make them a promising photocatalyst for the reduction of heavy metals. Another potential application is based on the reversible lithium intercalation ability at a voltage of about 1 V vs. Li/Li+.11 Nanostructured CuFeO2 shows an acceptable performance as anode in lithium ion batteries, however depending on particle size and processing temperature.12 In addition their thermoelectric performance is also of longstanding interest due to its high Seebeck coefficient and low electrical conductivity.13 Finally metal oxide gas sensors with temperature dependent variable sensing behaviour which rely on both, n-type oxide semiconducting and p-type semiconducting behaviour are still widely lacking. Nevertheless their response pattern would be much different to the gas sensing pattern of purely n-type oxide sensors and consequently those materials are thought after e.g. for pattern recognition in different environments.14 In this respect a delafossite type material like CuFeO2 could probably be an interesting candidate. Nevertheless reports towards gas sensoric studies of CuFeO2 are very scarce so far. To the best of our knowledge, besides a recent report on C2H5OH sensing,15 there are no other reports on gas sensing issues for CuFeO2.Using molecular precursor complexes which decompose in a well defined way at low or moderate temperatures giving gaseous or liquid decomposition products allow to deposit metal oxides in pure form and without severe contamination. No organic residues remain in the final ceramic which is on the other hand often the case e.g. when applying sol–gel methods for their synthesis.16 In addition to that, a high stability of these complexes in solution even for longer periods of time allows a homogenous mixing and their subsequent decomposition and calcination giving different homo and heterobimetallic nanostructures. Oximato complexes in which the methoxy-imino-propionato ligand framework is complexed towards different metal cations achieve these requirements thus allowing access to a variety of metal oxides compositions and morphologies like zinc oxide thin films,17 magnesium oxide rods,18 indium oxide nanotubes,19 or decoration of carbon nanotubes or gold nanoparticles with zinc oxide nanoparticles.20
Herein we present studies towards fabrication of hierarchically structured CuFeO2 fibers in nanometer size range and macroscopic length. For that purpose we introduce the usage of molecular precursors in connection with an electrospun polymer template. Electrospinning is a versatile method for producing nanofibers or tubes or wires for different types of applications like gas sensors21 or photoelectrodes for dye-sensitive solar cells.22 The technique has also been employed recently for the synthesis of one dimensional metal oxide nanomaterials.23–25 Microstructured CuAlO2 fibers obtained by electrospinning using PVA and appropriate metal salts have been reported.26 Herein, electrospun composite fibers containing polyacrylnitrile (PAN), and equal amounts of copper and iron oximato complexes as molecular precursors are described. From these, delafossite type CuFeO2 micro-fibrous nanowires organized in microstructured mats have been obtained after the PAN@1/2 composite fibers are further thermally processed first under oxygen followed by processing under an inert argon atmosphere at 750 °C. In addition we report on the gas sensing properties of delafossite type CuFeO2 nanowires towards O2 gas at different temperatures. These studies manifests the new material as variable temperature p/n-type semiconductor.
2 Experimental
2.1 Materials
Ammonium carbonate (NH4CO3, Grünethal), pyruvatic acid (C3H4O3, Merck), methoxylamine hydrochloride (CH3ONH2·HCl > 97%, TCI), iron chloride (FeCl2 > 99%, Riedel de Haën), copper nitrate hydrate (Cu(NO3)2·3H2O, Grünethal).2.2 Synthesis of the iron(II) oximato complex diaquabis[2-(methoxyimino) propanoato] iron(II) 1
Ammonium carbonate (2.88 g, 30 mmol) was added in small portions to a suspension of pyruvatic acid (2.64 g, 30 mmol) and methoxylamine hydrochloride (2.51 g, 30 mmol) in 100 ml water. After stirring for 1 h, iron chloride (1.90 g, 15 mmol) were added. The reaction mixture was stirred over night and the solvent was reduced by rotary evaporation. Yellow crystals were obtained by cooling to 5 °C. Combustion elemental analysis: found C 29.57%, H 4.87%, N 8.44%; calculated for C8H16FeN2O8: C 29.65%, H 4.98%, N 8.64%. 1H-NMR (D2O, 500 MHz, 300 K): δ = 1.6 (s, 3H, CH3), 3.4 (s, 3H, OCH3) ppm. HMBC spectra (D2O, 500 MHz, 300 K): δ = 16.5 (CH3), 61.6 (OCH3) 163.6 (CN) and 175.6 (CO) ppm. IR (ATR): ν = 3187 (s, br), 2965 (w), 2838 (w), 2384 (m), 1987 (w), 1642 (s), 1612 (s), 1437 (m), 1368 (s), 1219 (m), 1180 (m), 1051 (s), 939 (m), 867 (m), 771 (m), 640 (m, br), 559 (s), 450 (s) cm−1.2.3 Synthesis of copper(II) oximato complex diaquabis[2-(methoxyimino) propanoato]copper(II) 2
Ammonium carbonate (2.88 g, 30 mmol) was added in small portions to a suspension of pyruvatic acid (2.64 g, 30 mmol) and methoxylamine hydrochloride (2.51 g, 30 mmol) in 100 ml water. After stirring for 1 h, copper nitrate heptahydrate (3.62 g, 15 mmol) were added. The reaction mixture was stirred over night and the solvent was reduced by rotary evaporation. Blue crystals were obtained by recrystallisation of the obtained powder from ethanol at 5 °C. Combustion elemental analysis: found C 29.41%, H 4.80%, N 8.43%; calculated for C8H16CuN2O8: C 28.69%, H 4.86%, N 8.44%. IR (ATR): ν = 3388 (m), 2988 (w), 2909 (w), 1987 (w), 1635 (s), 1601 (s), 1411 (w), 1364 (w), 1229 (m), 1188 (m), 1053 (s), 945 (s), 884 (m), 762 (m), 735 (m), 558 (m), 458 (s), 417 (s) cm−1.2.4 Electrospinning of PAN/1/2-hybrid fibers and post-treatment to yield CuFeO2 nanowires
An 11 wt% PAN solution was made by dissolving PAN in DMF. These was mixed with a equal molar amount of iron 1 and copper 2 oximato complexes (ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in a 1
1) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 mass ratio of molecular precursors to polymer. After stirring the spinning solution over night, it was loaded into a glass syringe equipped with a stainless steel needle (0.9 × 20 mm). The voltage applied to the needle tip was kept at 30 kV and the distance between the copper counter electrode and tip was 15 cm. The spun fibre mats were peeled of the aluminum foil and dried at room temperature over night. The material was sintered in two steps at 750 °C for 6 h with a slow heating rate of 2 K min−1, first under air and under argon.
10 mass ratio of molecular precursors to polymer. After stirring the spinning solution over night, it was loaded into a glass syringe equipped with a stainless steel needle (0.9 × 20 mm). The voltage applied to the needle tip was kept at 30 kV and the distance between the copper counter electrode and tip was 15 cm. The spun fibre mats were peeled of the aluminum foil and dried at room temperature over night. The material was sintered in two steps at 750 °C for 6 h with a slow heating rate of 2 K min−1, first under air and under argon.
For study of the intermediate phases, the oximato complexes were dissolved in a stoichiometric ratio in ethanol and dried at 80 °C. The remaining green body was cold pressed with ten tons weight for ten minutes. The pellet was sintered at different temperatures for 6 h with a slow heating rate of 2 K min−1.
2.5 Gas sensor measurements
Gas-sensor (DC electrical) measurements were performed to monitor the sensor response of CuFeO2 to O2 in dry nitrogen. A solution of 20 wt% poly(methylmethacrylate) in dimethyl-formamide is used to deposit the sensing layer on alumina substrates homogeneously with Pt electrodes on the front side and a Pt heater on the back side. The samples were heat-treated at 700 °C for 5 h under air atmosphere prior to the measurement. The overall measurement set up is shown in Fig. 1This procedure gives a homogeneous thin film covering the electrodes of the senor device smoothly. Defined concentrations of O2 (1, 2, 5 and 10%) were introduced using a computer-controlled set-up with separate mass-flow controllers for the test gases (MKS, Munich, Germany, see Fig. 1). The gas sensing studies were performed in the temperature range of 200–500 °C. The sensor signal is given as the relative resistance; Rrel = RO2/R0, in which RO2 and R0 denote the sensors' resistances in the presence of O2 and in the absence of O2, respectively.
2.6 Material characterisation
XRD: StadiP (Stoe), Debye–Scherrer geometry using Cu-Kα1 radiation (1.541 Å) for non-iron samples and Mo-radiation (0.70930 Å) for iron samples, with a Ge (111) monochromator and Mythen 1K detector (Fa. Dectris). Single crystal diffraction data for complex 1 were collected on a Bruker Mach3 with Apex-II detector equipped with an Oxford Cryosystems (l)N2 cooling system and a Mo-anode and a focussing mirror (FR591, Helios-Mirror, Incoatec). Data for compound 2 were collected on a Oxford Diffraction Xcalibur single crystal X-ray diffractometer with a sapphire CCD Detector and a large Be window with a Mo-X-ray source. FTIR: Nicolet 6700 with ATR Smart Perfomer (ThermoFisher). TG/MS measurements: TG 209N1 coupled with Aelos QMS 403C (both Netzsch). Combustion analysis (CHN): VarioEL III CHN analyser. Cyclic voltammetry (CV): in H2O/1 M KCl with a calomel and graphitic electrode, Scan rate 100 mV s−1, Amel Mode 552-Potensiostat and 568 Programmable Function generator connected with a Nicolet 3091 and a Omnigraphic 2000 recorder (Houston Instrument). SEM: Philips XL 30 FEG (20 kV). The samples were prior coated with palladium using a Cressington Sputter Coater.3 Results and discussion
3.1 Synthesis and characterization of the molecular precursors 1 and 2
The oximato complexes 1 and 2 (“oximato” refers always to the methoxy-iminopropionato ligand framework herein) were prepared in a one-pot synthesis employing a Schiff-base condensation of pyruvatic acid and methoxylamine hydrochloride followed by the addition of the appropriate metal(II) source in water under basic conditions (Scheme 1). | ||
| Scheme 1 Schiff base condensation reaction forming octahedrally coordinated iron oximato and copper oximato complexes 1 and 2. | ||
Exemplarily, cyclic voltammetry shows that the initial reduction of complex 1 containing divalent iron is not possible (Fig. 2). Electronic However at a potential of +0.8 V the iron(II) complex 1 can be oxidized and at +0.04 V subsequently re-reduced under quasireversible conditions to 1+ containing iron(III). During the electrochemical oxidation half wave a color change of the reaction solution from yellow to orange is observed. The same effect can be achieved by addition of a base to a solution of 1 and reversed by the use of an acid.
The molecular structure of both precursor complexes 1 and 2 was determined by single crystal X-ray crystallography. 1 crystallizes in a monoclinic crystal system directly from water and 2 in a triclinic crystal system from ethanol at 5 °C. Their molecular structures (see Fig. 3 and 4) are closely related to the zinc keto–acidoximato complexes reported.27 The central metal atom in both complexes is located on a crystallographic centre of inversion and only one half of each molecule is crystallographically independent.
Consequently in 1 and 2 the methoxy groups are trans-oriented to each other. Between both complexes 1 and 2, the bond length variations in the ligand framework are minor. The distorted octahedral coordination sphere consists of two oximato ligands and two axially coordinated water molecules. The oxygen and nitrogen atoms of each oximato ligand are nearly square planar coordinated with a very slight deviation of less than 0.1° for 1 and 2 from the ideal square planar geometry. The crystallographic symmetry of both complexes enforces the axis H2O–M–OH2 of the two water molecules bound to Cu or Fe in 1 and 2 to be exactly linear.
The thermal decomposition of the oximato complexes 1 and 2 was examined under oxygen and helium atmosphere by coupled thermogravimetric/mass spectroscopy (TG-MS) studies (Fig. 5 and ESI Fig. S2† for a detailed TG-MS analysis).
 | ||
| Fig. 5 TG plot for the decomposition of the iron oximato complex 1 and of the copper oximato complex 2 in oxygen and helium. | ||
The decomposition of 1 and 2 under oxygen proceeds in three stages. In the first step at about 50 °C, water is already removed corresponding to a mass loss of about 10.4% for the iron complex 1 and 9.5% for the copper complex 2. This loss is due to the removal of the trans coordinated water molecules (calc. 11.1% for 1 and 10.9% for 2, respectively). The second decomposition step at about 190 °C indicates the decomposition of the oximato ligand framework. Subsequently the decomposition products CO2, CO, CHN, CH3CN and water are detected. In agreement with the decomposition of the zinc oximato complex a second order Beckmann decomposition reaction accounts for these products (Scheme 2).15 The final decomposition step occurs with a loss of CO2 at 450 °C to 490 °C, leaving a residual mass of 26.9% for 1 and 26.7% for 2. The theoretical ceramic yield of the expected oxides Fe2O3 (24.7%), Fe3O4 (23.8%) and CuO (23.8%) however is in all cases considerably lower which is typical for a residual minor contamination of the formed oxides with organic compounds from the decomposition process up to this reaction temperature.
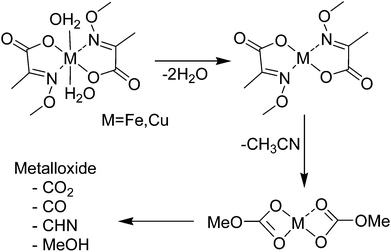 | ||
| Scheme 2 Thermal decomposition pathway and products of the oximato complexes 1 and 2 as found by TG-MS experiments. | ||
The decomposition under helium atmosphere was found to be different from the one under oxygen. The copper complex 2 degrades by showing two sharp decomposition steps while the process is more complex for the iron complex 1. The elimination of CO and methanol starts with an additional decomposition step at 300 °C followed by an additional loss of CO in a final step beginning at 470 °C which seems not even completed at 600 °C. The residual masses are 20.8% for 1 and 25.0% for 2 which are lower than under aerobic conditions. For the 1, the theoretical ceramic yield is lower than for all calculated iron oxides.
We assign this fact to the reduction of Fe(II) to elemental iron during the decomposition under inert atmosphere. For 2, the theoretical ceramic yield is higher than for CuO but formation of Cu2O (42.9%) seems also feasible.
3.2 Synthesis of the CuFe2O2 delafossite type phase from 1 and 2
Mixing equimolar amounts of the precursors 1 and 2 in ethanol, evaporation of the solvent and heating the solid mixture 110 °C gives as brown green solid.The coordinated water was removed and the Fe(II) oxidized to Fe(III) as indicated by the color change to brown. Sintering at different temperatures under oxygen atmosphere gives different metal oxides formed as intermediates (Fig. 6). Variable temperature XRD studies confirm the constituent phases in the green body. At 450 °C CuO and Fe3O4 are formed by decomposition of the green body obtained at around 110 °C. It is assumed that not all iron(II) was oxidized during formation of the green body. By increasing the temperature to 575 °C, the second iron containing phase, Fe2O3, becomes crystalline. At 750 °C the cuprous and ferric oxides reacts to the iron rich phase CuFe2O4 and CuO in a solid state reaction. In the last step under inert atmosphere these two oxides react to form the final delafossite type phase according to 2CuFe2O4 + 2CuO ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) 4CuFeO2 + O2. The resulting delafossite type product was crystalline and chemically pure. No other phases could be detected by XRD. However, this is a reversible reaction, so that CuFe2O4 can be re-formed under oxygen atmosphere from the desired delafossite type product phase. Processing the green body to delafossite under an inert atmosphere was not possible because elementary iron is formed from the oximato complex 1 (see Fig. 5) and elementary copper forms due to a reduction of Cu2O.23 In short, CuFeO2 could be synthesized at lower temperatures compared to molecular based sol–gel techniques so far widely employed.8b,c,12 It is known that by these methods the solute mixing is more homogeneous in the green body which reduces reaction time and temperature in comparison to the use of metal oxides as precursor. The advantage of the molecular precursors is the defined decomposition (see TGA, Fig. 5) in comparison to the undefined condensation steps typically present in the sol–gel type method.
4CuFeO2 + O2. The resulting delafossite type product was crystalline and chemically pure. No other phases could be detected by XRD. However, this is a reversible reaction, so that CuFe2O4 can be re-formed under oxygen atmosphere from the desired delafossite type product phase. Processing the green body to delafossite under an inert atmosphere was not possible because elementary iron is formed from the oximato complex 1 (see Fig. 5) and elementary copper forms due to a reduction of Cu2O.23 In short, CuFeO2 could be synthesized at lower temperatures compared to molecular based sol–gel techniques so far widely employed.8b,c,12 It is known that by these methods the solute mixing is more homogeneous in the green body which reduces reaction time and temperature in comparison to the use of metal oxides as precursor. The advantage of the molecular precursors is the defined decomposition (see TGA, Fig. 5) in comparison to the undefined condensation steps typically present in the sol–gel type method.
3.3 Electrospinning of oximato complexes 1 and 2 and PAN and transformation into 1D delafossite fibers
Fig. 7a shows a SEM image of hybrid polyacrylnitrile (PAN). PAN/1/2 composite fibers were obtained after electrospinning of equal amounts of complexes 1 and 2 together with PAN. The fibers are uniform in size with a smooth surface, their mean diameter is 620 nm. The IR spectrum of the composite material (Fig. 8b) reveals the incorporation of intact precursor complexes 1 and 2 into the hybrid PAN/1 and 2 composite fibers. After the first thermal treatment under oxygen atmosphere at 750 °C the color changed from yellow to brown and the fiber diameter is reduced considerably by about 70% of its original value to 180 nm (Fig. 7b). This shrinkage is due to the complete decomposition of the metal–organic complexes and the PAN polymer which results in a considerable material loss due to the formation of the volatile decomposition products and the formation of the final delafossite type ceramic (see TGA-MS study). Subsequently, the iron rich CuFe2O4 phase is formed as the major constituent of this first thermal treatment congruent with the result which has been observed in the polymer free delafossite synthesis (see XRD). | ||
| Fig. 8 IR spectra of (a) PAN fibers, (b) composite PAN/1 and PAN/2 fibers containing the oximato complexes 1 and 2, (c) iron oximato precursor complexes 1 and 2. | ||
The morphology of the surface of the composite wires remains smooth, displaying no voids or grooves due to disruption of the 1D structure along the nano wire length. After the final thermal processing under inert atmosphere the wire surface however becomes coarse and the wires do show a morphology composed of individual particle aggregates which are intimately interconnected to each other (Fig. 7c). Their diameter of 180 nm remains however nearly constant to the intermediate product obtained under oxygen treatment at 750 °C. The reaction of the mixed oxide composition CuO/Fe2O3/CuFe2O4 to the final delafossite phase effects the overall wire diameter obviously only to a minor extent. The XRD pattern (Fig. 7, upper trace) of the final delafossite type wires shows a similar characteristic as the pure CuFeO2 standard (31918-ICSD) as well to the CuFeO2 material obtained from the pure solid state reaction of the precursors 1 and 2 described before. Only a small impurity of CuO is present in the delafossite type wires. The wires are furthermore still aggregated into larger micrometer sized mats.
3.4 Gas sensing behaviour of the CuFeO2 delafossite fibers
CuFeO2 fibrous mats were deposited onto alumina substrate transducers equipped with Pt electrodes and separate heating wires embedded in the transducers. The gas sensor response was measured towards O2 and is shown for two 200 and 500 °C each for four different O2 concentrations (Fig. 9). At 200 °C the CuFeO2 responded as a p-type semiconductor at concentrations from 1 up to 10% O2. This points to the fact that [h+] are the major charge carriers responsible for that behaviour. In the temperature range between 300° and 350 °C we noticed a change in semiconducting behaviour of the sensor response (not shown) which moves from p to n-type conduction now corresponding to an electron dominated charge carrier behaviour. This observation was substantiated as intrinsic to the new material by measurement of several independent samples from different synthesis batches giving the same results. This change from hole [h+] to electron [e−] conducting behaviour at a temperature above 350 °C can be understood by an increase in the occupancy of electrons in the conduction band beyond that temperature.14 Typically such behaviour was indeed observed for intentionally doped metal oxides14,28–31 but was so far not reported for a stoichiometric oxide. Currently we can not rule out a valency change from copper(I) to copper(II) at higher temperatures under oxidizing conditions giving rise to the observed p to n type switching, however we would not expect a totally reversible effect as it is observed and thus would be solely responsible for the observed behaviour. It is interesting to note that delafossite type CuFeO2 material does show a high oxygen storage capacity under subsequent oxidation/reduction cycles lower than 500 °C.32 This would account for the slow oxygen response observed in our work and can be correlated with a p–n transition in the semiconducting behavior of CuFeO2.4 Conclusions
Molecular iron(II) and copper(II) oximato complexes 1 and 2 were synthesized, their molecular structure and thermal behavior determined. Due to their comparable thermal behavior and decomposition pathway they can be successfully applied to convert them into delafossite type materials of composition CuFeO2. Solutions of a polymer template PAN and 1 and 2 can be electrospun to yield 1D delafossite type wires of composition CuFeO2. The formation pathway yielding this phase is studied by temperature dependent XRD. The CuFeO2 fibers agglomerate during the electrospinning process to yield dense macroscopic mats which can be handled as macroscopic objects and thus are available as porous materials e.g. for gas sensing applications. Gas sensing studies towards O2 reveal a p-type behaviour towards O2 detection at 200 °C which switches to an n-type behaviour at a temperature above 350 °C. This behaviour could be interesting for simultaneous selective gas sensing of reducing and oxidizing gases. We are currently studying this behaviour in more depth.Acknowledgements
We thank Dr K. Hofmann (XRD), Dr M. Nowotny (CV), Dr R. Hoffman (TGA), and S. Foro (all of TU Darmstadt) and H. Schucht (MPI Mülheim) for technical support.Notes and references
- H. Kawazoe, M. Yasukawa, H. Hyodo, M. Kurita, H. Yanagi and H. Hosono, Nature, 1997, 389, 939 CrossRef CAS.
- (a) R. D. Shannon, D. B. Rogers and C. T. Prewitt, Inorg. Chem., 1971, 10, 713 CrossRef CAS; (b) C. T. Prewitt, R. D. Shannon and D. B. Rogers, Inorg. Chem., 1971, 10, 719 CrossRef; (c) D. B. Rogers, R. D. Shannon, C. T. Prewitt and J. L. Gilson, Inorg. Chem., 1971, 10, 723 CrossRef.
- (a) H. Gong, Y. Wang and Y. Luo, Appl. Phys. Lett., 2000, 76, 3959 CrossRef CAS; (b) H. Yanagi, S. I. Inoue, K. Ueda and H. Kawazoe, J. Appl. Phys., 2000, 88, 4159 CrossRef CAS; (c) M. K. Jayaraj, A. D. Draeseke, J. Tate and A. W. Sleight, Thin Solid Films, 2001, 397, 244 CrossRef CAS; (d) R. Nagarajan, A. D. Draeseke, A. W. Sleight and J. Tate, J. Appl. Phys., 2001, 89, 8022 CrossRef CAS; (e) R. Nagarajan, N. Duan, M. K. Jayaraj, J. Li, K. A. Vanaja, A. Yokochi, A. Draeseke, J. Tate and A. W. Sleight, Int. J. Inorg. Mater., 2001, 3, 265 CrossRef CAS.
- D. B. Rogers, R. D. Shannon, C. T. Prewitt and J. L. Gillson, Inorg. Chem., 1971, 10, 723 CrossRef.
- (a) T. Nozaki, K. Hayashi and T. Kajitani, Proc. ICT 2007 26th Int. Conf. on Thermoelectr. (Jeju Island, Korea, June 2007), 2008, vol. 168 Search PubMed; (b) M. Lalanne, P. Demont and A. Barnabé, J. Phys. D: Appl. Phys., 2011, 44, 185401 CrossRef.
- D. H. Choi, S. J. Moon, J. S. Hong, S. Y. An, I.-B. Shim and C. S. Kim, Thin Solid Films, 2009, 517, 3987 CrossRef CAS.
- (a) A. Barnabé, E. Mugnier, L. Presmanes and P. Tailhades, Mater. Lett., 2006, 60, 3468 CrossRef; (b) E. Mugnier, A. Barnabé, L. Presmanes and P. Tailhades, Thin Solid Films, 2008, 516, 1453 CrossRef CAS.
- (a) S. Götzendörfer, C. Polenzky, S. Ulrich and P. Löbmann, Thin Solid Films, 2009, 518, 1153 CrossRef; (b) H.-Y. Chen and J.-H. Wu, Thin Solid Films, 2012, 520, 5029 CrossRef CAS; (c) H.-Y. Chen and J.-H. Wu, Appl. Surf. Sci., 2012, 258, 4844 CrossRef CAS; (d) A. Banerjee, K. K. Chattopadhyay and S. W. Joo, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 406, 220 CrossRef CAS; (e) B. Szyszka, P. Loebmann, A. Georg, C. May and C. Elsaesser, Thin Solid Films, 2010, 518, 3109 CrossRef CAS.
- T.-W. Chiun and P.-S. Huang, Ceram. Int., 2013, 39, S575 CrossRef.
- S. Bassaida, M. Chaiba, S. Omeirib, A. Bougueliac and M. Trari, J. Photochem. Photobiol., A, 2009, 20, 62 CrossRef.
- A. M. Sukeshini, H. Kobayashi, M. Tabuchi and H. Kageyama, Solid State Ionics, 2000, 128, 33 CrossRef CAS.
- L. Lua, J.-Z. Wanga, X.-B. Zhua, X.-W. Gaoa and H.-K. Liua, J. Power Sources, 2011, 196, 7025 CrossRef.
- (a) T. Nozaki, K. Hayashi and T. Ajitani, J. Chem. Eng. Jpn., 2007, 40, 1205 CrossRef; (b) F. A. Benko and F. P. Koffyberg, J. Phys. Chem. Solids, 1987, 48, 431 CrossRef CAS.
- K. Galatsis, L. Cukrov, W. Wlodarski, P. McCormick, K. Kalantar-Zadeh, E. Comini and G. Sberveglieri, Sens. Actuators, B, 2003, 93, 562 CrossRef CAS.
- C. Ruttanapun, Y. Hongaromkid, W. Thowladda and A. Wichainchai, Proc. SPIE, 2013, 8883 Search PubMed : International Conference on Photonics Solutions, 88831L.
- (a) J. Wen and G. Wilkes, Chem. Mater., 1996, 4756, 1667 CrossRef; (b) M. Nentwich and A. Gazsó, in NanoTrust-Dossier 006, Institut Für Technikfolgen-Abschätzung, Wien, 2008, pp. 6–9 Search PubMed; (c) O. Lev, M. Tsionsky and L. Rabinovich, Anal. Chem., 1995, 67, 22 CrossRef.
- (a) J. J. Schneider, R. C. Hoffmann, J. Engstler, S. Dilfer, A. Klyszcz, E. Erdem, P. Jakes and R. A. Eichel, J. Mater. Chem., 2009, 19, 1449 RSC; (b) J. J. Schneider, R. C. Hoffmann, J. Engstler, O. Soffke, A. Issanin and A. Klyszcz, Adv. Mater., 2008, 20, 3383 CrossRef CAS.
- M. Pashchanka, R. C. Hoffmann and J. J. Schneider, J. Mater. Chem., 2010, 20, 957 RSC.
- M. Pashchanka, R. C. Hoffmann, A. Gurlo and J. J. Schneider, J. Mater. Chem., 2010, 20, 8311 RSC.
- J. Khanderi, R. C. Hoffmann, A. Gurlo and J. J. Schneider, J. Mater. Chem., 2009, 19, 5039 RSC.
- W.-S. Kim, B.-S. Lee, D.-H. Kim, H.-C. Kim, W.-R. Yu and S.-H. Hong, Nanotechnology, 2010, 21, 245605 CrossRef PubMed.
- (a) L.-J. Chen, J.-D. Liao, Y.-J. Chuang and Y.-S. Fu, Polymer, 2011, 52, 116 CrossRef CAS; (b) S.-H. Park, H.-J. Choi, S.-B. Lee, S.-M. Lee, S.-E. Cho, K.-H. Kim, Y.-K. Kim, M.-R. Kim and J.-K. Lee, Macromol. Res., 2011, 19, 142 CrossRef CAS.
- S. Agarwal, A. Greiner and J. H. Wendorff, Prog. Polym. Sci., 2013, 38, 963 CrossRef CAS.
- J. M. Li, X. L. Zeng, G. Q. Wu and Z. A. Xu, CrystEngComm, 2012, 14, 525 RSC.
- J. M. Li, X. L. Zeng, Y. H. Dong and Z. A. Xu, CrystEngComm, 2013, 15, 2372 RSC.
- S. Zhao, M. Li, X. Liu and G. Han, Mater. Chem. Phys., 2009, 116, 615 CrossRef CAS.
- M. R. Hill, A. W. Jones, J. J. Russell, N. K. Roberts and R. N. Lamb, Inorg. Chim. Acta, 2005, 358, 201 CrossRef CAS.
- A. Tomescu and R. Roescu, J. Optoelectron. Adv. Mater., 2005, 7, 1529 CAS.
- A. Wisitsoraat, A. Tuantranont, E. Comini, G. Sberveglieri and W. Wlodarski, Thin Solid Films, 2009, 517, 2775 CrossRef CAS.
- N. Savage, B. Chwieroth, A. Ginwalla, B. R. Patton, S. A. Akbar and P. K. Dutta, Sens. Actuators, B, 2001, 79, 17 CrossRef CAS.
- K. Galatsis, L. Cukrov, W. Wlodarski, P. McCormick, K. Kalantar-zadeh, E. Comini and G. Sberveglieri, Sens. Actuators, B, 2003, 93, 562–565 CrossRef CAS.
- S. Kato, R. Fujimaki, M. Ogasawara, T. Wakabayashi, Y. Nakahara and S. Nakata, Appl. Catal., B, 2009, 89, 183 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: TG-coupled MS data, crystal data and structure refinement of complexes 1 and 2. CCDC 970075 and 970076. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c3ra47514j |
| This journal is © The Royal Society of Chemistry 2014 |







