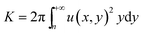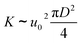Jet flow directed supramolecular self-assembly at aqueous liquid–liquid interface†
Yuefei Wanga,
Wei Qi*acd,
Renliang Huangab,
Rongxin Suacd and
Zhimin Hea
aState Key Laboratory of Chemical Engineering, School of Chemical Engineering and Technology, Tianjin University, Tianjin, 300072, China. E-mail: qiwei@tju.edu.cn
bSchool of Environmental Science and Engineering, Tianjin University, Tianjin, 300072, China
cTianjin Key Laboratory of Membrane Science and Desalination Technology, Tianjin, 300072, China
dThe Co-Innovation Center of Chemistry and Chemical Engineering of Tianjin, 300072, China
First published on 14th March 2014
Abstract
Self-assembly of molecular units at an interface has been an active area of research over the past few decades because of its advantages to prepare highly ordered materials. Here we demonstrated jet flow directed supramolecular self-assembly at the interface between two aqueous solutions, one containing a cationic polyacrylamide (CPAM) and the other, a small self-assembling Fmoc-diphenylalanine (Fmoc-FF) peptide bearing opposite charge. By controlling the jet flow of CPAM solution, we can fabricate macroscopic sac membranes, or microfibers composed of well-aligned Fmoc-FF nanofibers at the interface between two solutions. Moreover, the structure of microfibers may be hollow or solid depending on the intensity of jet flow. The entrainment of the jet flow will enhance the mixing between Fmoc-FF and CPAM solutions, which can be utilized for the fast fabrication of well-defined Fmoc-FF/CPAM microfibers. These microfibers can encapsulate functional components facilely for controlled drug release. Moreover, functional components such as QDs can also be integrated into the self-assembling system to easily achieve additional functionalities. We believe that the organization of components at the aqueous liquid–liquid interface will have great potential for the discovery of highly ordered and functional materials with potential applications in areas such as controlled drug release, wound dressing, enzyme immobilization, biosensors and other non-biological applications.
1 Introduction
Precise control of molecular units to self-assemble into desired nanostructures and then to hierarchically ordered materials is a great challenge in the field of materials science and supramolecular chemistry. Self-assembly of molecular units at an interface has been an active area of research over the past few decades as it can serve as a template to control structures. Up to now, a wide variety of interfacial systems such as Langmuir–Blodgett techniques,1,2 layer-by-layer (LBL) assembly,3 self-assembled monolayers (SAMs) on metals,4,5 physical vapor deposition,6,7 or liquid–liquid interface between two liquids,8–12 have been developed to prepare various supramolecular materials. Among them, aqueous liquid–liquid interface (no organic solvent), a common phenomenon in biology, motivated our great interest in directing molecular self-assembly. As a specific liquid–liquid interface, aqueous liquid–liquid interface is much more biocompatible and biomimetic compared with that of other interfacial systems. Previous studies have demonstrated the electrostatic complexation at aqueous liquid–liquid interface between oppositely charged macromolecules (e.g. chitosan/gellan, chitosan/alginate, etc.) and led to the formation of a dense membrane.13–17 However, the organization of self-assembling biomolecules such as peptides, proteins, DNA, at aqueous liquid–liquid interface, still remains largely unexplored.Recently, Stupp and co-workers demonstrated the dynamic self-assembly of porous membranes with hierarchical structures at the interface between two aqueous solutions, one containing negatively charged polysaccharide (hyaluronic acid) and the other, small amphiphile peptide bearing opposite charge.18 This remarkable self-assembling system, based on aqueous liquid–liquid interface, is highly biomimetic and lead to the formation of materials whose structure can be controlled on several length scales. Moreover, the mechanism of such self-assembling systems allows the use of an electric field to further control the self-assembly of biomolecules into a desired target at the aqueous liquid–liquid interface.19 Self-assembly at an aqueous liquid–liquid interface between two different components may be a feasible approach for us to design self-assembly systems for highly ordered materials.
In this work, we reported the hierarchical self-assembly of ordered materials at an aqueous liquid–liquid interface via the combination of an Fmoc-diphenyalanine (Fmoc-FF) peptide solution with a solution of high molecular weight cationic polyacrylamide (CPAM), and demonstrated that the materials' morphology could be tailored by control of the jet flow boundary layer localized at the aqueous liquid–liquid interface. Fmoc-FF is an aromatic dipeptide possessing an N-terminal fluorenyl-9-methoxycarbonyl (Fmoc) group (Fig. S1a†), which can self-assemble into high aspect ratio nanofibres in an antiparallel β-sheet arrangement.20,21 The self-assembly of Fmoc-FF molecules can be switched by changing the pH or zeta potential of solution.22,23 At high pH values, Fmoc-FF molecules can be dispersed in water and stay in their monomeric state owing to the strong negative charge.23 CPAM is a linear positively charged macromolecule (Fig. S1b†) and thus be chosen as a stimuli component to trigger the assembly of Fmoc-FF molecules at the aqueous interface between two solutions. When CPAM solution comes into contacting with Fmoc-FF solution, a membrane will be assembled gradually at the aqueous liquid–liquid interface of the two solutions. Based on this point, we investigated the influence of fluid flow on the self-assembly of components at the aqueous liquid–liquid interface. When CPAM solution was injected slowly into Fmoc-FF solution, a well-defined sac membrane will be assembled gradually at the interface between the two solutions. However, by increase of the injection speed, a microfiber composed of aligned Fmoc-FF nanofibres will be formed due to the presence of a remarkable shear flow localized at the aqueous liquid–liquid interface. Moreover, the structure of microfibers may be hollow or solid which can be tailored by control the intensity of CPAM jet flow. The entrainment of jet flow will enhance the mixing between Fmoc-FF and CPAM solutions, which can be utilized for the fast fabrication of well-defined Fmoc-FF/CPAM microfibers. These microfibers can encapsulate functional components facilely for controlled drug release. Based on the aqueous liquid–liquid interface, we further demonstrated that functional components such as quantum dots (QDs) can be also integrated into the self-assembling system to easily achieve additional functionalities.
2 Experimental
2.1 Chemicals and materials
Fmoc-diphenylalanine peptide (Fmoc-FF) was purchased from Bachem; cationic polyacrylamide (CPAM) and sodium hydroxide were purchased form Tianjin Guangfu Fine Chemical Research Institute. All chemicals were used as received without any further purification. Fmoc-FF solution was prepared by dispersing the lyophilized peptide powder in ddH2O through sonication, followed by adding an appropriate amount of 0.5 M NaOH solution to the suspension with subsequent stirring for 30 min.2.2 Zeta potential measurement of CPAM and peptide solutions
Zeta potential measurement of 0.1 wt% CPAM solution was accessed over a wide pH range. The zeta potential of Fmoc-FF solutions were carried out at a constant pH value of 9.75. The ability to form well-defined sac membrane was determined qualitatively at each pH value by injecting an aliquot of 0.75 wt% CPAM solution into a 0.15 wt% Fmoc-FF solution at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio (v/v). pH values were measured by using an microprocessor pH meter (Microbench pH 600, Singapore).
2 ratio (v/v). pH values were measured by using an microprocessor pH meter (Microbench pH 600, Singapore).
2.3 Characterization
For scanning electron microscopic (SEM) analysis, sac membranes or microfibers were rinsed firstly in ddH2O for 5 min. The water was then removed by quick-freezing in liquid nitrogen and subsequent freezing-dry under vacuum. All the samples were sputter-coated with platinum using E1045 Pt-coater (Hitachi High-technologies CO., Japan), and then imaged by an S-4800 field emission scanning electron microscope (SEM, Hitachi High-technologies CO., Japan) at an acceleration voltage of 3 kV.2.4 Drug encapsulation in Fmoc-FF/CPAM microfibers and controlled release
500 μl 1.0 wt% CPAM solution dissolving vitamin B2 or rhodamine B was injected into 0.1 wt% or 0.2 wt% Fmoc-FF peptide solutions at a flow rate of 4.8 cm3 min−1. The microfibers formed instantly within seconds, which were then collected by filtration, washed with ddH2O, and used into the following release experiment. The filtrated solution was collected and measured by UV adsorption spectra to determine the amount of unloaded drugs during the drug encapsulation process, so that we can calculate the encapsulation efficiency of vitamin B2 or rhodamine B in the Fmoc-FF/CPAM microfibers. The Fmoc-FF/CPAM microfiber loaded with vitamin B2 or rhodamine B was then dispersed into 7 ml pH 6.8 PBS solutions in a glass vial, which was then incubated in a rotary shaker at 50 rpm and 37 °C. UV adsorption spectrum was used to determine the amount of vitamin B2 or rhodamine B released from the Fmoc-FF/CPAM microfiber, on a TU-1810 UV/vis spectrophotometer (Shanghai Metash Instruments, China).3 Results and discussion
Scheme 1 illustrated the method we used in our experiment and the jet flow directed self-assembly through the combination of an Fmoc-FF solution with a CPAM solution at the aqueous liquid–liquid interface. The lyophilized peptide was dissolved in water by adding an appropriate amount of 0.5 M NaOH. To initiate the self-assembling process, CPAM solution was then injected into the freshly prepared Fmoc-FF solution. By slow injection, a closed sac membrane composed of entangled Fmoc-FF nanofibres and CPAM chains was formed at the aqueous liquid–liquid interface. In contrast, by quick injection, a microfiber containing highly-ordered and well-aligned Fmoc-FF nanofibres along its axis was formed. For the same molecular units, different materials can be fabricated just through controlling the jet flow boundary layer at the aqueous liquid–liquid interface.3.1 Self-assembly of sac membrane composed of entangled Fmoc-FF nanofibres
Firstly, we investigated the self-assembly of Fmoc-FF and CPAM at slow injection speed. After the slow injection of a CPAM solution into an Fmoc-FF solution, a solid membrane could be formed immediately localized at the interface between two liquids, leading to the formation of a well-defined polymer-filled sac (Fig. 1a). The sac was robust enough to hold its own weight without tearing the membrane when it was pulled up by a tweezers (Fig. 1b), and was stable in water or PBS solution for several months without the use of cross-linking chemistry. The electrostatic interaction occurring at the liquid–liquid interface between small Fmoc-FF molecules and CPAM polymer chains played a key role on the formation of well-defined sac membrane. Based on this point, we investigated the effect of zeta potential in CPAM solution on membrane formation. Robust Sac membrane could be formed only when CPAM solution had strong zeta potential of opposite charge (Fig. 1c). When the pH of CPAM solution approached its isoelectric value of 9.3, a membrane could also be formed but was very weak and unstable due to the lowly positive charge on CPAM polymer chains, which could not provide adequate electrostatic interaction with Fmoc-FF molecules. Further increase of the pH value of CPAM solution could finally lead to the rapid mixing of two solutions and give rise to a weak hydrogel.For fabricating sac membranes with porously hierarchical structures, we combined a 0.75 wt% CPAM solution at pH 4 with a 0.15 wt% Fmoc-FF solution at pH 9.75 with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio (v/v) (Fig. 2). Scanning electron microscopic analysis (Fig. 2a–c) revealed that the sac membranes were composed of well-defined, intertwisting nanofibers with diameter of 30 to 50 nm and length >10 μm. To further understand the self-assembly process, we investigated the microstructure of sac membrane as a function of the growth time. Upon liquid–liquid contact of the two solutions, robust sac membrane consisting of entangled nanofibers could be formed with a thickness of about 150 nm after 10 min (Fig. 2d). We believed that the continuous diffusion of small Fmoc-FF molecules into CPAM solution played a key role in this process. As a result of electrostatic screening by the positively charged CPAM molecules, the Fmoc-FF molecules then self-assembled into well-defined nanofibres localized at the liquid–liquid interface. The negatively charged nanofibres further interacted with flexible CPAM polymer chains through electrostatic complexation, thus leading to the formation of a porous membrane that prevented the rapid mixing of two miscible liquids. Ordered growth of Fmoc-FF nanofibres paralleling to the interface continued to form a membrane with a thickness of 581 nm after 2 h of the initial contact (Fig. 2e). However, with the further growth of membrane, the diffusion of Fmoc-FF molecules through the membrane into CPAM solution side became much slower, making it difficult to form well-defined nanofibres. Therefore, the electrostatic complexation among CPAM and Fmoc-FF molecules or their smaller aggregates led to the formation of a dense and disordered layer with a thickness of 607 nm adjacent to the CPAM solution side after 9 h of the initial contact (Fig. 2f). In previous studies about Fmoc-FF as well as other peptides, the self-assembly of these molecules usually occurred in bulk solution at global thermodynamic equilibrium and usually led to the formation of nanostructures that lacked the ability to further organize themselves into more complex and ordered structures. However, in our experiment, the aqueous liquid–liquid interface could provide a local non-equilibrium environment localized at the interface, thus leading to the organization of molecular components into a macroscopic 2D membrane between two solutions. Moreover, the porosity and thickness of self-assembled sac membrane could be facilely controlled by changing pH (Fig. S2†) and concentration (Fig. S3†) of the two solutions, as well as growth time of the membrane (Fig. 2d–f).
2 ratio (v/v) (Fig. 2). Scanning electron microscopic analysis (Fig. 2a–c) revealed that the sac membranes were composed of well-defined, intertwisting nanofibers with diameter of 30 to 50 nm and length >10 μm. To further understand the self-assembly process, we investigated the microstructure of sac membrane as a function of the growth time. Upon liquid–liquid contact of the two solutions, robust sac membrane consisting of entangled nanofibers could be formed with a thickness of about 150 nm after 10 min (Fig. 2d). We believed that the continuous diffusion of small Fmoc-FF molecules into CPAM solution played a key role in this process. As a result of electrostatic screening by the positively charged CPAM molecules, the Fmoc-FF molecules then self-assembled into well-defined nanofibres localized at the liquid–liquid interface. The negatively charged nanofibres further interacted with flexible CPAM polymer chains through electrostatic complexation, thus leading to the formation of a porous membrane that prevented the rapid mixing of two miscible liquids. Ordered growth of Fmoc-FF nanofibres paralleling to the interface continued to form a membrane with a thickness of 581 nm after 2 h of the initial contact (Fig. 2e). However, with the further growth of membrane, the diffusion of Fmoc-FF molecules through the membrane into CPAM solution side became much slower, making it difficult to form well-defined nanofibres. Therefore, the electrostatic complexation among CPAM and Fmoc-FF molecules or their smaller aggregates led to the formation of a dense and disordered layer with a thickness of 607 nm adjacent to the CPAM solution side after 9 h of the initial contact (Fig. 2f). In previous studies about Fmoc-FF as well as other peptides, the self-assembly of these molecules usually occurred in bulk solution at global thermodynamic equilibrium and usually led to the formation of nanostructures that lacked the ability to further organize themselves into more complex and ordered structures. However, in our experiment, the aqueous liquid–liquid interface could provide a local non-equilibrium environment localized at the interface, thus leading to the organization of molecular components into a macroscopic 2D membrane between two solutions. Moreover, the porosity and thickness of self-assembled sac membrane could be facilely controlled by changing pH (Fig. S2†) and concentration (Fig. S3†) of the two solutions, as well as growth time of the membrane (Fig. 2d–f).
3.2 Self-assembly of microfiber consisting of well-aligned Fmoc-FF nanofibres
The self-assembly of sac membranes with ordered structures at an aqueous liquid–liquid interface opens the way to construct unique self-assembling systems for highly ordered and functional materials. Compared with other interface-assisted self-assembly systems, one outstanding advantage for aqueous liquid–liquid interface is that, the interactions between components at the interface can be further modified by changing the environment of boundary layer between the two solutions, so that we can manipulate the self-assembly of components into a desired target. Previous work by Zhang et al. had demonstrated the formation of aligned peptide amphiphile nanofibers under shear force.24 Inspired by this, we brought a shear flow to the aqueous liquid–liquid interface by quick injection of the CPAM solution into Fmoc-FF solution, and speculated that the shear flow might provide a sustainable driving force to compel the aligned growth of Fmoc-FF nanofibres at the aqueous liquid–liquid interface. Fig. 3a showed the photographic image snapped during the injection of 1.0 wt% CPAM solution into a solution of 0.1 wt% Fmoc-FF at a flow rate of 4.8 cm3 min−1 from a needle capillary (internal diameter of 100 μm). The shear flow of CPAM solution gave rise to a velocity gradient across the liquid–liquid interface. This led to the rapid mixing of two solutions at the early stage of liquid–liquid contact (Fig. 3a, 0.3 and 0.8 s) and then the formation of stable, well-defined Fmoc-FF/CPAM microfibers within seconds (Fig 3a, 1.5 s). The microfiber was stable once it was formed and preserved a structural integrity that was independent with the further incubation of microfiber in peptide solution (Fig. 3b). Polarized optical microscopy (POM) showed uniform birefringence under crossed polarizers (Fig. 3c and d), indicating large-scale alignment along the length of microfiber was achieved.To further investigate the effect of injection speed on the microstructure of self-assembled Fmoc-FF/CPAM microfibers, CPAM solution was injected into the Fmoc-FF solution with a moderate speed of 2.4 cm3 min−1 and a relatively higher speed of 4.8 cm3 min−1, respectively. Fig. 4 showed the SEM images of Fmoc-FF/CPAM microfibers formed at a flow rate of 2.4 cm3 min−1, indicating that a polymer filled hollow microfiber was formed (Fig. 4a and c). As the microfiber was squashed during the SEM sample preparation process, the diameter of the microfiber was in fact ∼110 μm. The wall of hollow microfiber was a membrane composed of semi-aligned Fmoc-FF nanofibres (Fig. 4b) and had a thickness of about 350 nm (Fig. 4d). The amorphous flocs within the wall of the microfiber were CPAM polymers (Fig. 4c), and this indicated that the mixing and subsequent electrostatic complexation between Fmoc-FF molecules and CPAM polymer chains only occurred at the exterior surface of the microfiber. However, when CPAM solution was injected at a speed of 4.8 cm3 min−1, SEM analysis revealed that the self-assembled Fmoc-FF/CPAM microfiber exhibited distinct morphologies. Fig. 5a showed the typical morphology of a single microfiber self-assembled at a speed of 4.8 cm3 min−1. The microfiber was much thinner (∼30 μm) compared with that formed at 2.4 cm3 min−1 and had clearly linear striations running along the fiber length. High-magnification SEM of the surface and cross-section (Fig. 5b and d) indicated that the microfiber was solid and composed of well-aligned Fmoc-FF nanofiber bundles (typical diameter of 30–150 nm and length >10 μm). The packing density of Fmoc-FF nanofibres within the microfiber was not very high and this was verified by visualizing the microfiber cross section (Fig. 5d), which revealed a lot of hundred-nanometer-sized voids within interior region of the microfiber. Moreover, Fig. S4 was the POM images of the hollow and solid microfibers.† From the images we can see that, the solid microfibers exhibited higher image contrast than that of the hollow microfibers, due to the higher packing density of Fmoc-FF nanofibres within the solid microfibers. These results indicated that the structure of Fmoc-FF/CPAM microfibers could be fine controlled by changing the injection speed of CPAM solution, from hollow microfibers with a wall composed of semi-aligned Fmoc-FF nanofibres to more compact microfibers with well-aligned Fmoc-FF nanofiber bundles and hundred-nanometer-sized voids.
3.3 Hierarchical self-assembling mechanism
Jet flow is ubiquitous in technology and engineering. Based on the theory of circular jet flow,25 the velocity boundary layer localized at the aqueous liquid–liquid interface will affect greatly on the mixing and organization of components between the two solutions. At very slow injection speed (Reynolds number (Re) < 30), the jet flow exhibits a lamellar boundary layer, in which the mass transfer of components is relatively slow compared with that of electrostatic complexation process. This will lead to the formation of a very thin membrane serving as a diffusion barrier at the initial liquid–liquid contact (Fig. 2d), and the further growth of membrane requires Fmoc-FF molecules to diffuse through the initial formed diffusion barrier into CPAM solution side. This process is very slow and the formation of a well-defined sac membrane with a certain thickness usually takes several hours (Fig. 2e and f). However, at higher injection speed (Re > 30), the jet flow exhibits a turbulent boundary layer, which will give rise to a remarkable entrainment effect (Fig. 6a and b). According to classical circular turbulent jet flow theories by H. Schlichting,25 the quality (Q) of surrounding static fluid entrained into the turbulent jet flow can be described as follows:where x and y are the axial and vertical positions of the circular turbulent jet flow; K is the kinematic momentum; u(x,y) is the local axial velocity of the jet flow. Considering the conservation of the axial momentum of a turbulent jet, the value of K depends on the initial momentum flux of the jet flow which can be described by the expression
where u0 and D are the initial velocity and diameter of the jet flow. As a result, with increase of the initial momentum flux, the value of K as well as Q increases. The entrainment effect of the turbulent jet flow will lead to the rapid mixing of Fmoc-FF and CPAM molecules across the boundary layer. The subsequent electrostatic screening and self-assembly of Fmoc-FF molecules in the boundary layer will lead to the instant formation of a well-defined microfiber with aligned Fmoc-FF nanofibres along the fiber axis (Fig. 6c–e). Moreover, the assembly of a hollow (Fig. 4) or solid (Fig. 5) microfiber depends directly on the mass transfer speed of Fmoc-FF molecules across the turbulent jet flow boundary layer into CPAM solution side. We speculate that the interplay of several factors including shear force of the fluid, mass transfer speed of components within the jet flow boundary layer, the quick formation of stable electrostatic complexations, as well as the assembly of Fmoc-FF molecules, are responsible for the formation of various supramolecular materials. For the formation of a hollow microfiber, the entrainment of Fmoc-FF solution into CPAM jet flow is still not strong enough. As a result of the fast electrostatic complexation between Fmoc-FF molecules and CPAM polymer chains, a diffusion barrier at the frontier of the mixing layer will be formed preventing the further mixing of the two solutions. However, with further increase of the injection speed, the entrainment of jet flow dominates, which will entrain adequate amount of Fmoc-FF molecules completely across the whole cross-section of circular CPAM jet flow. As is demonstrated in the work by Zhang et al.,24 the subsequent electrostatic screening and assembly of Fmoc-FF molecules under shear force then lead to the instant formation a solid microfiber composed of well-aligned Fmoc-FF bundles within seconds (Fig. 6c–e).
3.4 Drug encapsulation in Fmoc-FF/CPAM microfibers and controlled release
Up to now, a wide variety of self-assembling systems have been designed to fabricate materials with distinct nano- or microstructures. However, for practical applications, the ability of facile and large-scale fabrication of such self-assembled materials is also very important. As was shown in Scheme 1, the slow injection of CPAM solution into Fmoc-FF solution would lead to the formation of a sac membrane localized at the aqueous liquid–liquid interface. However, the rapid growth of a well-defined sac membrane was limited by the relatively slow diffusion speed. As shown in Fig. 2e and f, the self-assembly of a porous membrane with a thickness of 581 nm required 2 h and further incubation to 9 h just led to the formation of a dense membrane adjacent to the CPAM solution side. However, by quick injection of CPAM solution into Fmoc-FF solution, the entrainment effect of turbulent jet flow accelerated the mixing between Fmoc-FF and CPAM solution at the aqueous liquid–liquid interface (Fig. 6b). This led to the instant formation of a Fmoc-FF/CPAM microfiber within seconds (Fig. 3a and b) with highly ordered nanostructures (Fig. 4 and 5). Based on these points, we believed that the utilization of turbulent jet flow by the quick injection of a polymer solution into a self-assembling peptide solution might be a feasible approach for the fast and massive fabrication of such kind of self-assembled microfibers, which may have potential applications for controlled drug release, wound dressing or serving as scaffold for 3D cell culture with controlled release of functional components from the fiber.As a preliminary study, we incorporated vitamin B2 and rhodamine B into the Fmoc-FF/CPAM microfibers respectively, and investigated their release behaviour. In a typical experiment, 500 μl 1.0 wt% CPAM solution dissolving vitamin B2 or rhodamine B was injected into 0.1 wt% or 0.2 wt% Fmoc-FF peptide solutions at a flow rate of 4.8 cm3 min−1. The microfibers formed instantly within seconds, which were then collected by filtration, washed with ddH2O, and used into the release experiment. As shown in Fig. 7a, the encapsulation efficiency of vitamin B2 depended on the concentration of peptide solution (black and red column). By increase of the concentration of peptide solution, the encapsulation efficiency of vitamin B2 increased from 46.40% to 75.25%. Moreover, if we incorporated a positively charged dye, rhodamine B, into the microfiber, the encapsulation efficiency further increased to 88.84%. Kinetic release curves for vitamin B2 from Fmoc-FF/CPAM microfibers indicated that the controlled release could be achieved by changing the concentration of peptide solution during the microfiber fabrication (Fig. 7b, black and red curve). Moreover, comparing the kinetic release curve of vitamin B2 (Fig. 7b, red curve) with rhodamine B (Fig. 7b, blue curve), the release of positively charged rhodamine B molecules was much slower than that of vitamin B2.
3.5 Integration of CdTe QDs for additional functionality
Up to this point, we have explored how the complexation of small peptide molecules with CPAM at an aqueous liquid–liquid interface can lead to various self-assembled structures by control of the velocity boundary layer of the interface. Another strong advantage of such self-assembling system based on aqueous liquid–liquid interface is that the integration of other components such as negatively charged nanoparticles, quantum dots (QDs) or enzymes, can be easily achieved for additional functionalities. To demonstrate this, we slowly injected an equal volume of CPAM solution (0.75 wt%) into the Fmoc-FF solution (0.15 wt%) dispersed with 10 μM CdTe QDs to fabricate CdTe/Fmoc-FF/CPAM sac membrane. Fig. 8a and b showed the fluorescent and white light images of a vial containing a sac membrane as it grew with the simultaneous integration of CdTe QDs over the period from 5 min to 48 h. The fading yellow-green fluorescent emission of the Fmoc-FF solution was in sharp contrast with that of the sac membrane which intensified with the growth of time (Fig. 8a). In a similar way, the white light images showed that the sac membrane turned yellow gradually, while the Fmoc-FF solution became colorless (Fig. 8b). These suggested that the negatively charged CdTe QDs on the Fmoc-FF solution side diffused gradually toward the interface driving by the electrostatic attraction, and then QDs were incorporated into the membrane as it grew. SEM analysis revealed that the sac membrane was composed of well-defined Fmoc-FF nanofibres, but we still observed some spherical aggregates with diameter of about 50 nm in the 3D network of the porous sac membrane (Fig. 8c). These aggregates were much larger than the CdTe QDs (<10 nm) which might be attributed to the electrostatic complexation between flexible CPAM polymer chains and CdTe QDs. Energy-dispersive X-ray (EDX) analysis of the sac membrane provided further evidence for the existence of CdTe QDs in the sac membrane (Fig. 8d). After the integration of such kind of functional nanoparticles, the hybrid membrane may have potential applications in areas such as biosensor or other non-biological applications.4 Conclusions
In summary, we have demonstrated a jet flow directed self-assembly at the interface between two aqueous solutions, one containing self-assembling Fmoc-diphenylalanine (Fmoc-FF) peptides and the other, linear cationic polyacrylamide (CPAM). By controlling the jet flow of CPAM solution, we can fabricate macroscopic sac membranes, or microfibers composed of well-aligned Fmoc-FF nanofibres at the interface between two solutions. Moreover, the structure of microfibers may be hollow or solid depending on the intensity of jet flow. The entrainment of jet flow will enhance the mixing between Fmoc-FF and CPAM solutions, which can be utilized for the fast fabrication of well-defined Fmoc-FF/CPAM microfibers. These microfibers can encapsulate functional components facilely for controlled drug release. Moreover, functional components such as QDs can also be integrated into the self-assembling system to easily achieve additional functionalities. We believe that the organization of components at the aqueous liquid–liquid interface will have great potential for the discovery of highly ordered and functional materials with potential applications in areas such as controlled drug release, would dressing, enzyme immobilization, biosensor and other non-biological applications.Acknowledgements
This work was supported by the Natural Science Foundation of China (51173128, 20976125, 31071509, 21306134) and Tianjin (no. 10JCYBJC05100), the Program for New Century Excellent Talents in Chinese University (NCET-08-0386), the Key Project of Chinese Ministry of Education (108031), the 863 Program of China (2008AA10Z318), the Program of Introducing Talents of Discipline to Universities of China (no. B06006), and the Ministry of Education (no. 20130032120029).References
- J. Zasadzinski, R. Viswanathan, L. Madsen, J. Garnaes and D. Schwartz, Science, 1994, 263, 1726–1738 CAS.
- A. Ulman, The Handbook of Surface Imaging and Visualization, 1995, p. 277 Search PubMed.
- G. Decher, Science, 1997, 277, 1232–1237 CrossRef CAS.
- C. D. Bain, E. B. Troughton, Y. T. Tao, J. Evall, G. M. Whitesides and R. G. Nuzzo, J. Am. Chem. Soc., 1989, 111, 321–335 CrossRef CAS.
- J. C. Love, L. A. Estroff, J. K. Kriebel, R. G. Nuzzo and G. M. Whitesides, Chem. Rev., 2005, 105, 1103–1170 CrossRef CAS PubMed.
- L. Adler-Abramovich, D. Aronov, P. Beker, M. Yevnin, S. Stempler, L. Buzhansky, G. Rosenman and E. Gazit, Nat. Nanotechnol., 2009, 4, 849–854 CrossRef CAS PubMed.
- J. S. Lee, I. Yoon, J. Kim, H. Ihee, B. Kim and C. B. Park, Angew. Chem., Int. Ed., 2011, 50, 1164–1167 CrossRef CAS PubMed.
- Z. Niu, J. He, T. P. Russell and Q. Wang, Angew. Chem., Int. Ed., 2010, 49, 10052–10066 CrossRef CAS PubMed.
- L. Hu, M. Chen, X. Fang and L. Wu, Chem. Soc. Rev., 2012, 41, 1350–1362 RSC.
- L. Pirondini, F. Bertolini, B. Cantadori, F. Ugozzoli, C. Massera and E. Dalcanale, Proc. Natl. Acad. Sci. U. S. A., 2002, 99, 4911–4915 CrossRef CAS PubMed.
- D. Kohler, M. Schneider, M. Krüger, C. M. Lehr, H. Möhwald and D. Wang, Adv. Mater., 2011, 23, 1376–1379 CrossRef CAS PubMed.
- H. Duan, D. Wang, D. G. Kurth and H. Möhwald, Angew. Chem., Int. Ed., 2004, 43, 5639–5642 CrossRef CAS PubMed.
- S. Wen, Y. Xiaonan and W. T. Stevenson, Biomaterials, 1991, 12, 374–384 CrossRef CAS PubMed.
- O. Gåserød, O. Smidsrød and G. Skjåk-Bræk, Biomaterials, 1998, 19, 1815–1825 CrossRef.
- H. Yamamoto and Y. Senoo, Macromol. Chem. Phys., 2000, 201, 84–92 CrossRef CAS.
- K. Wang and Z. He, Int. J. Pharm., 2002, 244, 117–126 CrossRef CAS PubMed.
- K. Ohkawa, T. Kitagawa and H. Yamamoto, Macromol. Mater. Eng., 2004, 289, 33–40 CrossRef CAS.
- R. M. Capito, H. S. Azevedo, Y. S. Velichko, A. Mata and S. I. Stupp, Science, 2008, 319, 1812–1816 CrossRef CAS PubMed.
- Y. S. Velichko, J. R. Mantei, R. Bitton, D. Carvajal, K. R. Shull and S. I. Stupp, Adv. Funct. Mater., 2012, 22, 369–377 CrossRef CAS PubMed.
- V. Jayawarna, M. Ali, T. A. Jowitt, A. F. Miller, A. Saiani, J. E. Gough and R. V. Ulijn, Adv. Mater., 2006, 18, 611–614 CrossRef CAS.
- A. Mahler, M. Reches, M. Rechter, S. Cohen and E. Gazit, Adv. Mater., 2006, 18, 1365–1370 CrossRef CAS.
- A. M. Smith, R. J. Williams, C. Tang, P. Coppo, R. F. Collins, M. L. Turner, A. Saiani and R. V. Ulign, Adv. Mater., 2008, 20, 37–41 CrossRef CAS.
- C. Tang, A. M. Smith, R. F. Collins, R. V. Ulijn and A. Saiani, Langmuir, 2009, 25, 9447–9453 CrossRef CAS PubMed.
- S. Zhang, M. A. Greenfield, A. Mata, L. C. Palmer, R. Bitton, J. R. Mantei, C. Aparicio, M. O. de la Cruz and S. I. Stupp, Nat. Mater., 2010, 9, 594–601 CrossRef CAS PubMed.
- H. Schlichting and J. Kestin, Boundary-layer theory, McGraw-Hill, New York, 1968 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Molecular structures of the Fmoc-FF and CPAM, SEM images of the sac membranes. See DOI: 10.1039/c3ra47483f |
| This journal is © The Royal Society of Chemistry 2014 |