Switching light with light – advanced functional colloidal monolayers†
K.
Bley
a,
N.
Sinatra
a,
N.
Vogel
ab,
K.
Landfester
a and
C. K.
Weiss
*ac
aMax-Planck Institute for Polymer Research, Ackermannweg 10, 55128 Mainz, Germany
bSchool of Engineering and Applied Sciences, Harvard University, McKay 426, 9 Oxford Street, Cambridge, MA 02138, USA
cUniversity of Applied Sciences Bingen, Berlinstrasse 109, 55411 Bingen, Germany. E-mail: c.weiss@fh-bingen.de
First published on 29th October 2013
Abstract
Colloidal monolayers comprising of highly ordered two dimensional crystals are of high interest to generate surface patterns for a variety of different applications. Mostly, unfunctionalized polymer or silica colloids are assembled into monolayers. However, the incorporation of functional molecules into such colloids offers a convenient possibility of implementing additional properties to the two-dimensional crystal. Here, we present the formation of novel functional colloidal monolayers with photoswitchable fluorescence. The miniemulsion polymerization technique was used to incorporate an appropriate dye system of a perylene-based fluorophore and a bis-arylethene as a photochrome in polymeric colloids in defined ratios. Upon irradiation with UV or visible light the photochrome reversibly isomerizes from the ring-closed form, which is able to absorb light of the emission wavelength of the fluorescent dye and the ring-open form, which is not. The fluorescence emission of the dye can thus be reversibly switched on and off with light even when embedded in colloids. The colloids were self-assembled at the air–water interface to produce hexagonally ordered functional monolayers and more complex binary crystals. We investigate in detail the influence of the polymeric matrix on the switching properties of the fluorophore/photochrome system and find that the rate constants for the photoswitching, which all lie in the same range, are less influenced by the polymeric environment than expected. We demonstrate the reversible switching of the fluorescence emission in self-assembled colloidal monolayers. The arrangement of broadly distributed functional colloids into ordered monolayers with high addressability was obtained by the formation of binary colloidal monolayers.
Introduction
Highly ordered two-dimensional colloidal crystals find many applications in science and technology.1,2 In addition to the investigation of their optical properties,3–5 colloidal monolayers are mainly used in lithographic processes, using colloids as templates for metal evaporation,6–10 as a powerful platform for surface patterning,11 for example to mimic vivid structural color occurring in nature or to control wetting properties and for the creation of plasmonic structures.7–9,12–16 Typically, hexagonally close-packed monolayers are used, which can be prepared in high quality using a variety of methods.17,18 The complexity of the structural design can be increased by the creation of binary monolayers. Here, the two-dimensional (2D) crystal is generated from colloids of two distinct sizes. The small colloids can be located in varied, yet defined amounts in the interstitial sites of a hexagonally close-packed monolayer of large colloids,19–21 or the large colloids are separated by the small colloids randomly22 or in high order.21,23 Kingshott et al.24–26 created binary monolayers using colloids of different materials, i.e. polymer and silica, and different sizes, where one colloid population could be removed selectively by solvents, offering an easy access to sophisticated surface functionalization. Another strategy to implement the functionality to colloidal monolayers and to extend lithographic possibilities is the use of hybrid or functional colloids. This strategy proved very viable, particularly for the preparation of regular arrays of metal nanoparticles. As an extension of micellar lithography,27,28 and an alternative to using hybrid hydrogels,29,30 metal complex loaded colloids were used to prepare hexagonal 2D arrays of metal nanoparticles.31,32 These were subjected to plasma or thermal treatment to eventually generate regular arrays of metal nanoparticles. The metal is determined by the choice of the encapsulated complex,29,33 the size of the metal nanoparticles by the amount of complex,31 and the lattice spacing by the size of the initial colloids.34The defined and controlled encapsulation of metal complexes or other materials in polymeric colloids requires special formulation techniques. Although it is possible to use emulsion polymerization for the encapsulation of metal complexes,33,34 the amount of encapsulated material is hard to control, as the process is controlled by the diffusion of the reactants to the locus of polymerization.35 The encapsulation of multiple compounds in defined ratios, as e.g. required for the formation of alloy particles,29 is virtually impossible. To overcome this issue, the miniemulsion polymerization technique can be used. This heterophase polymerization technique allows using various monomers, and encapsulating compounds in defined amounts35 and multiple compounds in defined ratios.36 This has not only been shown for metal complexes, but also for the co-encapsulation of a photochromic dye and a fluorophore. Furukawa37 has shown that the simultaneous encapsulation of fluorescent and photochromic dyes following the miniemulsion approach can lead to inter-component energy transfer between the dye molecules to create particles with light-switchable fluorescence. The authors used a BODIPY dye (4,4-difluoro-1,3,5,7,8-pentamethyl-4-bora-3a,4a-diaza-s-indacene) as the fluorophore and cis-1,2-dicyano-1,2-bis-(2,4,5-trimethyl-3-thienyl)ethene (CMTE) as the photochromic compound.37
The use of photochromic dyes enables light-switchable emission, as photochromic dyes like CMTE reversibly isomerize upon irradiation with light.38,39 The isomers have different optical absorption properties and, thus, are used in self darkening glasses or color changing commodities. Here, however, one isomer of CMTE can absorb at the emission wavelength of the fluorophore, quenching the emission.
In this contribution, we study in detail the optical switching properties of a fluorophore/photochrome system in polymeric colloids of different materials and assemble such colloids into two-dimensional structures. Thus, the individual colloids become addressable and may be used for optical barcoding, data storage or switching. First, the optical properties of the system CMTE as a photochrome and N-(2,6-diisopropylphenyl)-perylene-3,4-dicarboximide (PMI) acting as a fluorophore are investigated in solutions of the monomers used for colloid preparation. Two polymers are used for the preparation of the colloids: polystyrene (PS), with a glass transition temperature well above room temperature, and poly(butyl acrylate) (PBA) with a glass transition temperature below room temperature. As the photoisomerization of CMTE is accompanied by geometrical rearrangement the rigidity (depending on the Tg) of the polymeric matrix is expected to influence the velocity of the isomerization and the kinetic stability of the thermodynamically unfavored isomer.40 Thus, the optical properties of the photosystem incorporated into colloids are investigated. Although it is possible to create monolayers of PS colloids with some ordering, PBA is not suitable for the formation of colloidal monolayers. It is possible to transform the low-ordered monolayer formed by PS colloids into higher order structures by assembling the colloids into a binary monolayer with larger, unfunctionalized PS colloids. Here, the functional colloids regularly assemble in the interstitial sites of a hexagonally close-packed monolayer of larger colloids. This strategy is also suitable for PBA colloids, after they have been subjected to a seeded emulsion polymerization process with styrene as a monomer in order to prevent film formation of the PBA polymer with low glass transition temperature (Tg). Thus, rigid colloids are generated, which can be deposited on solid substrates. Finally, we provide direct experimental evidence of light-switchable emission in the colloidal monolayers.
Results and discussion
Concept for the preparation of functional photoswitchable colloidal monolayers
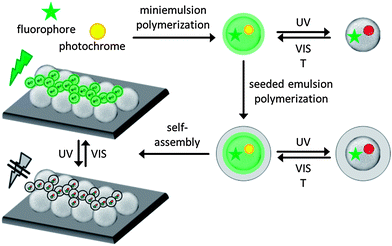
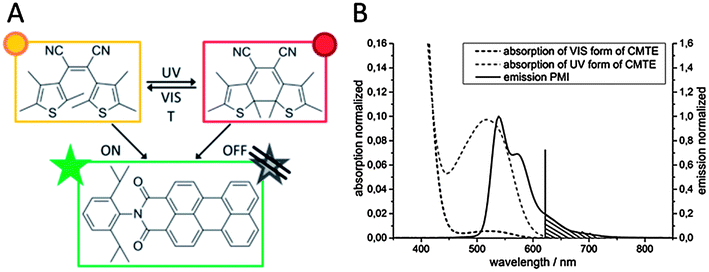
The concept for the creation of photoswitchable polymeric colloidal monolayers as a model for data storage application or light erasable barcoding is shown in Fig. 1. Here, a special dye system consisting of a fluorophore and a photochrome is encapsulated in a polymeric environment in defined ratios for the production of photoswitchable colloids. Thus, miniemulsion polymerization is the appropriate technique for encapsulation of various materials such as dyes in a controllable way. First, we perform a detailed investigation of the optical properties of the photoswitchable dye system containing PMI as the fluorophore and CMTE as the photochrome in solution and in polymeric colloids. The different isomerization states of CMTE were obtained by irradiation with UV and visible light, respectively. The resulting colloids were covered with an additional rigid shell of polystyrene by seeded emulsion polymerization as protection for low glass temperature (Tg) polymers such as PBA and to narrow the size distribution of the colloids.41,42 The seeded colloids can be self-assembled for the creation of functional photoswitchable and highly ordered colloidal monolayers.To produce functional photoswitchable colloidal monolayers a dye system based on a bis-thienyl photochrome (cis-1,2-dicyano-1,2-bis-(2,4,5-trimethyl-3-thienyl)ethene, CMTE) and a perylene based fluorophore (N-(2,6-diisopropylphenyl)-perylene-3,4-dicarboximide, PMI) was used (Fig. 2A).
The dye system allows switching on and off the fluorescence emission of the PMI. By irradiating the photochrome CMTE with UV and VIS-light the CMTE molecule undergoes a ring-closing or ring-opening cyclization (Fig. 2A and B), respectively. In the ring-closed excited state the CMTE is able to absorb the emission of the PMI between 500 nm < λ < 620 nm (Fig. 2B). Therefore, the emission of PMI is switched off. After VIS-light irradiation the emission can be switched on again.
The excited, ring-closed state thermally relaxes into the ring-open state due to the thermodynamically unfavorable strained ring configuration and the loss of aromaticity of the thiophene rings (Fig. 2A).39 In solutions and even in polymer films the molecules can easily diffuse, thus compromising the addressability of a defined spatial storage “pixel” of information or readout. However, when incorporated into spatially confined colloidal particles, which are subsequently assembled into a two-dimensional array, diffusion of the dye is effectively suppressed and individual pixels with resolution defined by the size of the colloids are obtained. Increasing the stability of the dye system to prevent the thermally induced ring-opening cyclization that will compromise the storage of information over longer periods of time is obtained by embedding the dyes into a polymeric matrix. Moreover, addressable units of this dye system are necessary for a defined system of high addressability and easy readout. Therefore, small amounts of the dyes in defined ratios were encapsulated in polymeric colloids using the miniemulsion technique. As the polymeric matrix can affect the switching process of the dye because of geometric restrictions the influence of the polymeric environment was investigated using poly(butyl acrylate) as a soft polymer with low Tg (Tg ≪ room temperature [RT]) and polystyrene as a rigid polymer (Tg ≫ RT). Polymers with low Tg such as poly(butyl acrylate) are not suited for a subsequent assembly of the formed colloids into a colloidal monolayer because they immediately fuse together and form a film. To prevent film formation and allow the application of soft PBA colloids in self-assembly, a seeded emulsion polymerization approach was used to generate a rigid shell around the PBA colloids for protection.42,43 Another advantage of the seeded emulsion polymerization is the adjustment of size and reduction of size distribution,43 which promotes the formation of monolayers of higher order during the self-assembly process. The size distribution plays an important role in the creation of densely packed colloidal monolayers. The highest overall ordering quality can be obtained using particles of uniform size, whereas colloids of broader size distribution reduce the ordering quality at the air–water interface. We assemble the functional colloids into ordered monolayers and more complex binary monolayers and demonstrate optical switching with high resolution. With simple geometric models the particle size range for the small particles which can be co-crystallized can be determined (see ESI 3†).19 The binary monolayers prepared by the co-crystallization method are of high crystallinity and ordering degree because the interstices tolerate a broad range of sizes for the smaller colloids without disturbance of the self-assembly of the larger particles.
Investigation of optical properties of the dyes in solution
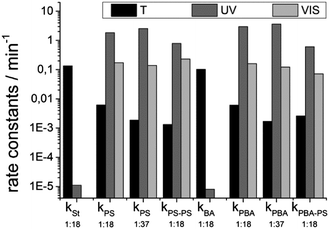
The optical properties of the photochromic dye (CMTE) and the fluorophore (PMI) were investigated in the monomers used for the preparation of colloids. Thus, solutions of CMTE and solutions of PMI in styrene and butyl acrylate were used for absorption (CTME) and fluorescence emission (PMI) spectroscopy, respectively.The thermodynamically stable state of CMTE at room temperature is the ring-open form (Fig. 2A). By absorption of UV-light the molecule undergoes a cyclization reaction to form the ring-closed form (Fig. 2A). In contrast to the open form (black spectrum, Fig. 2B), the ring-closed isomer shows a broad absorption band with a maximum of λ = 520 nm (light grey spectrum, Fig. 2B). Although the isomerization from the ring-closed into the ring-open state is thermodynamically favoured, excellent thermal stability has been reported giving the photochrome a kind of “memory” ability.40 Investigation of the thermal stability of CMTE in the monomers styrene and butyl acrylate showed that the half-life of the ring-closed CMTE is about t1/2 = 5.2 min in solutions of styrene and t1/2 = 6.8 min in butyl acrylate, respectively. The rate constants were calculated from the linear decay fit (eqn (1) and (2)) and summarized in Fig. 3.
| y = mx + n | (1) |
| k = −m | (2) |
Colloids with photoswitchable emission
The miniemulsion polymerization is the appropriate technique to encapsulate multiple compounds in defined ratios in a variety of different polymeric materials. Therefore, a quantitative encapsulation of the dye system for switching fluorescence emission with adjustable ratios in the used polymers, polystyrene and poly(butyl acrylate), is guaranteed. To investigate whether the polymeric matrix restricts the switching process we used styrene and butyl acrylate as monomers for the miniemulsion approach. The glass transition temperature of polystyrene is lowered from that of pure PS (100 °C)44 because of the encapsulation of hexadecane and the dye molecules (Table 1). The rigidity of PS may lead to conformational restrictions for the geometric changes of the CMTE molecules during the irradiation process. Poly(butyl acrylate) on the other hand is a “soft” polymer with Tg ≈ −50 °C (Table 1). We expect the photoswitching to occur faster in a softer and more flexible polymeric environment, which means that the emission intensity of the fluorescent dye should be reduced with higher rate constants in poly(butyl acrylate) than in polystyrene. The irradiation experiments with UV and visible light of the different polymeric colloids with varying amounts of photochromic dye are described in the following.| Polymer | PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE CMTE |
d/nm | σ/nm | Distribution/% | T g/°C |
|---|---|---|---|---|---|
| PS | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
84 | 14 | 17 | 71 |
| PS | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 37 |
94 | 16 | 17 | 72 |
| PBA | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
159 | 20 | 13 | −56 |
| PBA | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 37 |
155 | 30 | 19 | −49 |
| PS–PS | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
191 | 17 | 9 | 88 |
| PBA–PS | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
260 | 31 | 12 | 93 |
Photocyclization by UV-light irradiation
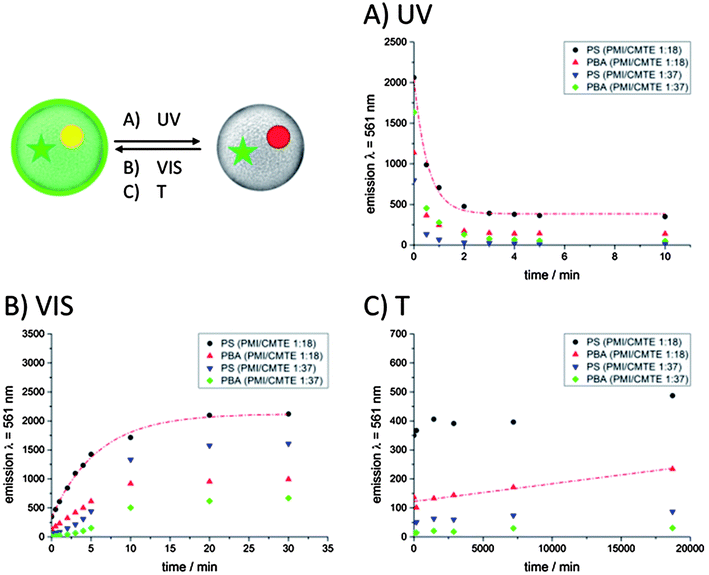
The absorption of PMI's fluorescence emission can be described as intercomponent energy transfer between the fluorophore and the photochrome. Thus, the distance between the CMTE and the PMI molecules, which is determined by the concentration, affects the efficiency of the absorption of PMI's emission.The efficiency of the energy transfer decreases with increasing molecule distance. Thus, we investigated the influence of varying amounts of photochromic dye on the energy transfer or on the switching process, respectively, in different polymeric environments. The development of the fluorescence intensity of the PMI during the irradiation can be followed easily by fluorescence spectroscopy as the emission maximum at a wavelength of λ = 561 nm changes with time due to the formation of the ring-closed or ring-open state of CMTE and, therefore, varying concentrations of molecules absorbing the fluorescence intensity of PMI. Emission spectra were recorded after given intervals of UV-light irradiation until no further increase of the emission signal was visible (ESI 2†). Fig. 4A shows the time dependent decrease of the emission of the fluorescent dye PMI in a colloidal system of polystyrene and poly(butyl acrylate) in the presence of the photochromic dye CMTE (ratios of PMI to CMTE 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 and 1
18 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37). After about 3–4 min of UV-irradiation no further decay of fluorescence is visible, indicating that the entire CMTE has isomerized into the ring-closed form. From the data, the rate constants for the decrease of the fluorescence intensity at the maximum at λ = 561 nm were calculated with an exponential fit (eqn (3) and (4)).
37). After about 3–4 min of UV-irradiation no further decay of fluorescence is visible, indicating that the entire CMTE has isomerized into the ring-closed form. From the data, the rate constants for the decrease of the fluorescence intensity at the maximum at λ = 561 nm were calculated with an exponential fit (eqn (3) and (4)).
y = A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−x/t) + y0 exp(−x/t) + y0 | (3) |
| k = t−1 | (4) |
Moreover, the effect of CMTE concentration on the switching efficiency was investigated. The temporal evolution of the fluorescence emission of the dispersions upon irradiation with UV-light containing a higher amount of CMTE (PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE as 1
CMTE as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37, Fig. 4A, blue triangles pointing down and green rhombi as data points) shows that the fluorescence stays at a constant level after 3 min for PS and PBA, respectively. The emission of the system containing the dyes with ratio 1
37, Fig. 4A, blue triangles pointing down and green rhombi as data points) shows that the fluorescence stays at a constant level after 3 min for PS and PBA, respectively. The emission of the system containing the dyes with ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 in PS as well as in PBA is significantly lower than that with a ratio of PMI
37 in PS as well as in PBA is significantly lower than that with a ratio of PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE of 1
CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 (Fig. 4A, black and red dots). As expected, the switching process with higher concentration of CMTE was faster with increasing amount of activated quenching molecules next to the fluorescent dye (Fig. 3). Compared to the rate constants of the particles with a ratio of PMI
18 (Fig. 4A, black and red dots). As expected, the switching process with higher concentration of CMTE was faster with increasing amount of activated quenching molecules next to the fluorescent dye (Fig. 3). Compared to the rate constants of the particles with a ratio of PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE of 1
CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18, the rate constants of the intercomponent energy transfer process between the ring-closed form of CMTE and the PMI can be increased by 37% for the polystyrene particles and 21% for the poly(butyl acrylate) nanoparticles when using a higher amount of CMTE (ratio PMI
18, the rate constants of the intercomponent energy transfer process between the ring-closed form of CMTE and the PMI can be increased by 37% for the polystyrene particles and 21% for the poly(butyl acrylate) nanoparticles when using a higher amount of CMTE (ratio PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE 1
CMTE 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37).
37).
VIS-light induced reverse cyclization isomerization
For the reverse isomerization process the samples were irradiated with visible light of wavelength range 515 nm < λ < 690 nm. The corresponding emission spectra (Fig. 4B) show a time dependent increase of the fluorescence maximum at λ = 561 nm, which correlates with the increasing amount of CMTE molecules isomerized into the ring-open state. These are not able to quench the emission because of the absence of spectral overlap of CMTE absorption and PMI emission.The initial intensity of PMI emission in polystyrene recovered after 20 min of irradiation. The switching rate constants were similar in the different polymeric environments of PS and PBA colloids and generally about an order of magnitude lower than that of the UV-induced isomerization (Fig. 3). For the polystyrene particles with a dye ratio of PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE of 1
CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 a rate constant 20 times slower and for the dye ratio 1
37 a rate constant 20 times slower and for the dye ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 a rate constant 10 times slower compared to the isomerization in solution were calculated for the VIS-light induced ring-opening reaction. For the PBA particles we observed similar behavior but the isomerization is about 20 times slower for a ratio of PMI/CMTE of 1
18 a rate constant 10 times slower compared to the isomerization in solution were calculated for the VIS-light induced ring-opening reaction. For the PBA particles we observed similar behavior but the isomerization is about 20 times slower for a ratio of PMI/CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 and 30 times slower for a ratio of 1
18 and 30 times slower for a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 as the isomerization in solution.
37 as the isomerization in solution.
Reversibility of the light induced cyclization process
We have successfully shown that the fluorescence emission of the colloids with the chosen dye system can be switched by optical stimulation at different wavelengths between UV and VIS-light. For any application a high reversibility and high number of cyclization cycles of the emission are necessary.To perform several switching cycles we used alternating irradiation with visible and UV-light of PS colloids with the ratio of PMI/CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
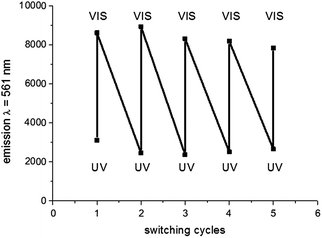
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18. The dispersion was irradiated with UV-light first to ensure all CMTE is present in the excited state, whereas the particles' emission was switched off. Afterwards, the ring-opening reaction was initiated by VIS-light irradiation isomerizing CMTE to the ground state, thus, switching the particles on. After 30 min of irradiation the emission of the sample was investigated by fluorescence spectroscopy. Afterwards, the dispersion was again irradiated with UV-light to initiate the ring-closing reaction. This procedure was repeated several times to obtain a higher number of switching cycles. The resulting emission intensities show the presence of the two states for fluorescence emission on/off in colloids for a well-repeatable isomerization, whereas the emission intensities can be restored completely for every switching cycle, indicating good photoswitchability (Fig. 5).
18. The dispersion was irradiated with UV-light first to ensure all CMTE is present in the excited state, whereas the particles' emission was switched off. Afterwards, the ring-opening reaction was initiated by VIS-light irradiation isomerizing CMTE to the ground state, thus, switching the particles on. After 30 min of irradiation the emission of the sample was investigated by fluorescence spectroscopy. Afterwards, the dispersion was again irradiated with UV-light to initiate the ring-closing reaction. This procedure was repeated several times to obtain a higher number of switching cycles. The resulting emission intensities show the presence of the two states for fluorescence emission on/off in colloids for a well-repeatable isomerization, whereas the emission intensities can be restored completely for every switching cycle, indicating good photoswitchability (Fig. 5).
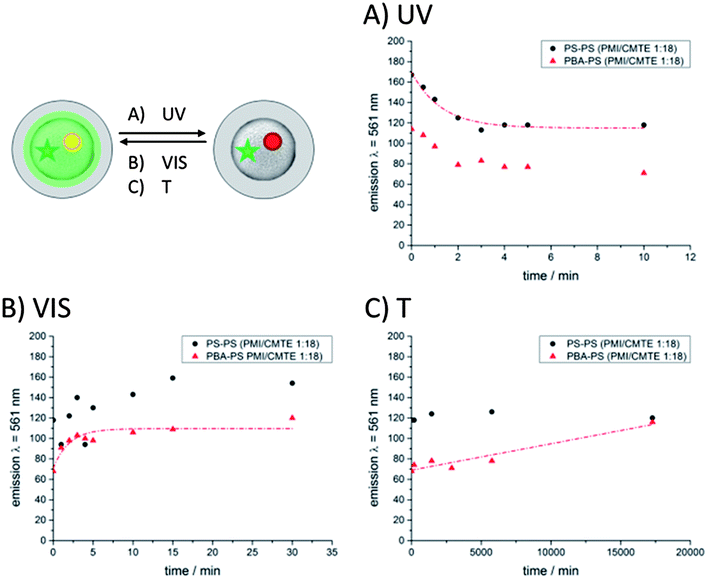
Thermal stability of colloids prepared by miniemulsion polymerization
The stability of the individual on/off states in the absence of a stimulus is of great importance. Storage of information can only be realized with stable and defined states. Although the ring-closed form of CMTE is the thermodynamically unstable state, the thermally activated ring opening isomerization can be decelerated by embedding the dye molecules in a polymeric environment of a colloid. Fig. 4C shows the time evolution of the fluorescence intensity at λ = 561 nm of UV-light irradiated dispersions (room temperature). In both polymeric particles, the fluorescence recovery is very slow. Complete recovery of the emission was not observed even after more than 10 days. The data acquired from the system with a ratio of PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE of 1
CMTE of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 are also shown in Fig. 4C. Compared to the nanoparticles with a lower ratio of PMI
37 are also shown in Fig. 4C. Compared to the nanoparticles with a lower ratio of PMI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CMTE (1
CMTE (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18) the fluorescence intensity decreases with a similar rate constant.
18) the fluorescence intensity decreases with a similar rate constant.
Comparing the rate constants for the thermally induced restoration it is obvious that the reverse cyclization reaction into the ring-open form proceeds with a similar velocity not depending on the nature of the surrounding polymeric matrix. The results underline the excellent thermal stability of the ring-closed state of CMTE and the steric hindrance of the isomerization. The dye system is at least 20 times more stable when being embedded in a polymeric matrix than in solution and light-induced information can easily be stored for more than 10 days.
To summarize, the optical properties and the rate constants confirm that the photoswitching with UV-light proceeds faster in a soft polymeric matrix such as poly(butyl acrylate) (Tg = −50 °C) than in a rigid matrix such as polystyrene (Tg = 71 °C). The VIS-light induced ring opening reaction has a rate constant which is about 10 times smaller than the rate constant for UV-light induced ring closing cyclization. The fluorescence emission can be reversibly recovered by alternating irradiation with visible light within the wavelength range of 515 nm < λ < 690 nm and UV-light for several switching cycles without photo-bleaching effects. The states of the photochromic system shows excellent thermal stability when being embedded in polymeric colloids.
Optical properties of colloids prepared by seeded emulsion polymerization
As mentioned earlier, the formation of a PS shell around the functional colloids serves two purposes. First, PBA is covered by a rigid shell so that it can be used for the self-assembly and deposition process, and second, the size distribution of the colloids can be narrowed by the process.44 The optical properties of these colloids were also investigated and they largely resemble that of the primary colloids. The data are summarized in Fig. 6.As the seed particle ideally does not change, the resulting optical properties are expected to be similar to that of the seed particle, with the exception of a lower emission intensity. Although the emission intensity is less intense than that from the seed particles the decrease in the emission maximum at λ = 561 nm with proceeding isomerization is still visible and follows an exponential decay (Fig. 6A). The corresponding rate constants of the seeded particles (Fig. 3) show that the switching process during irradiation with UV-light was decelerated compared to the original seed particles from miniemulsion. The differences may arise from scattering effects due to the enlargement of the particles and the additional shell of polystyrene whereby less light reaches the inner functional core of the hybrid seeded particles.
The fluorescence recovery induced by irradiation with visible light with a wavelength range of 515 nm < λ < 690 nm is shown in Fig. 6B. After 15 min the fluorescence intensity reaches the initial value. The hybrid particles with an additional shell of polystyrene show similar switching behavior to the seed particles. The switching process seems to be slightly decelerated (Fig. 3). This effect might be attributed to scattering effects of the larger particles whereas a low light intensity reaches the dye system. Fig. 6C shows the time dependent development of the thermally induced ring opening reaction of CMTE. The increase of the fluorescence intensity of PMI at λ = 561 nm was plotted vs. time and the rate constants were calculated from the linear fit (eqn (1) and (2)) (Fig. 3). An undesired back-switching to the initial state was not observed for PS–PS colloids in the timeframe of the experiment (several days), thus underlining the excellent stability of the photo-states when incorporated in polymer particles. Moreover, the rate constants for the hybrid particles lie in the same range as the seed colloids without an additional PS shell (Fig. 3). In addition, the excellent thermal stability of the system was also shown in hybrid particle systems.
Direct self-assembly of seeded colloids at the air–water interface
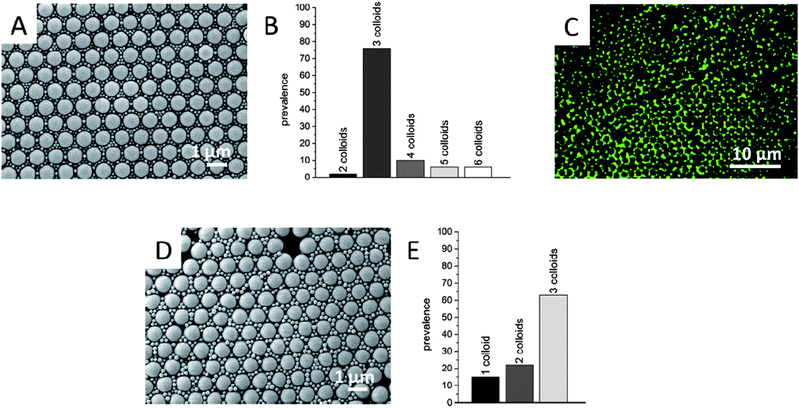
We investigated the optical properties of photoswitchable colloids and showed an excellent thermal stability for the on/off states in all cases. For the creation of a model system for data storage or light erasable barcoding the colloids need to be precisely located in defined positions. This can be realized using the self-assembly approach of colloids at the air–water interface on a Langmuir trough into two dimensional highly ordered colloidal monolayers. The primary colloids with a size distribution of σ > 10% were not suitable for the production of high quality colloidal monolayers. Therefore, we produced larger particles (Table 1) by seeded emulsion polymerization with a narrower size distribution of σ = 9%. If the size distribution is too broad, the particles cannot form a hexagonal lattice and the arrangement at the interface is disturbed. Another possibility to create highly ordered monolayers is the formation of binary colloidal monolayers. Thus, two differently sized particle systems are co-assembled at the interface. We used carboxylated polystyrene particles of about 1150 nm as the template for the particles with photoswitchable emission. The smaller seeded colloids are located in the interstices of the resulting hexagonal lattice of the large particles. Therefore, it was possible to create binary colloidal crystals of high ordering quality, which can be seen in the resulting scanning electron micrographs (Fig. 7A and D). These two dimensional crystals, having domain sizes of several hundreds of μm2, were evaluated graphically by assessing the possible arrangements of the particles at the interstitial sites. Therefore, the size of the interstitial gaps between the particles was calculated (see ESI 3† for a detailed evaluation) and the seeded PS–PS and PBA–PS particles correlated with the size of the gaps. The resulting geometries were distinguished and identified in the scanning electron micrographs (Fig. 7A and D). The number ratio of larger to small particles Nlarge/Nsmall was adjusted to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 at the interface. The statistical evaluation of the binary monolayers shows a predominance of 3 colloids (76%) located in the interstitial positions for the PS–PS particles (Fig. 7B), rather than the maximum arrangement of 6 colloids (up to 6%), which is also in agreement with the adjusted number ratio of Nlarge/Nsmall of 1
6 at the interface. The statistical evaluation of the binary monolayers shows a predominance of 3 colloids (76%) located in the interstitial positions for the PS–PS particles (Fig. 7B), rather than the maximum arrangement of 6 colloids (up to 6%), which is also in agreement with the adjusted number ratio of Nlarge/Nsmall of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 for template to photoswitchable particles.
6 for template to photoswitchable particles.
Fig. 7E shows the statistical evaluation of the binary monolayer of template particles with photoswitchable PBA–PS colloids. As the PBA–PS particles are larger (d = 260 nm) than the PS–PS particles (d = 191 nm), the maximum number of particles for the co-localization at the interstitial site is a geometrical arrangement of 3 colloids (see ESI 3†). The histogram shows again a predominance of 3 colloids (63%) located in the interstitial positions (Fig. 7E).
As can be seen in the scanning electron microscopy (SEM) images and from the statistical evaluation (Fig. 7A, B, D and E) the binary colloidal monolayers tolerate a relatively broad size distribution of the smaller colloids located at the interstitial sites of the carboxylated PS template particles, without disturbing the high order of the template colloids. With the number ratio of large particles to small particles (Nlarge/Nsmall) the configuration can be adjusted reliably, even with broadly distributed colloids.
The binary colloidal monolayer of non-fluorescent template PS particles and photoswitchable PS–PS particles was also investigated by confocal laser scanning microscopy. The corresponding CLSM image (Fig. 7C) shows a very high degree of ordering, where the larger template colloids arranged in a hexagonal lattice (dark spots) and the interstitial sites are filled with the smaller fluorescent seeded particles. Therefore, it would be possible to address one single interstitial site in a very efficient and defined way because of the high crystallinity of the functional colloidal monolayer.
Photoswitching in colloidal monolayers
Finally, we demonstrate the successful use of a colloidal monolayer to reversibly store (and erase) information with light. An optical fluorescence microscope was used for the investigation of the photoswitching of the colloidal monolayers. The substrate was first irradiated with visible light to obtain the ring-open state of the CMTE and the highest possible fluorescence intensity of the colloidal particles (Fig. 8A). Afterwards, part of the substrate was exposed to UV-light to induce the photocyclization reaction of the CMTE into the ring-closed state, whereas the fluorescence emission of the fluorophore is absorbed and the fluorescence is switched off in the exposed areas (Fig. 8B).Irradiation with visible light can be used to reversibly erase the stored information: after illumination for 300 ms, a complete recovery of the fluorescence intensity in the monolayer is observed (ESI 4†).
Experimental section
Materials
Sodium dodecyl sulfate (SDS, Sigma Aldrich), 2,2′-azobis(2-methylbutyronitrile) (V59, WAKO-Chemicals), ammonium peroxodisulfate (APS, Sigma Aldrich), hexadecane (99%, Sigma Aldrich), N-(2,6-diisopropylphenyl)-perylene-3,4-dicarboximide (PMI, a donation of BASF), cis-1,2-dicyano-1,2-bis-(2,4,5-trimethyl-3-thienyl)ethene (CMTE, TCI Europe) and ethanol (VWR) were used as received. The monomers styrene (S, Sigma Aldrich) and butyl acrylate (BA, Sigma Aldrich) were purified using flash column chromatography with aluminum oxide to remove any containing inhibitor. Water of Milli-Q quality was used throughout the experiments.Miniemulsion polymerization
100 mg of sodium dodecyl sulfate (SDS) were dissolved in 24 g of water and stirred at 1000 min−1. Another solution was prepared separately from the monomer (styrene or butyl acrylate), 250 mg of the osmotic pressure agent hexadecane, 100 mg of the initiator 2,2′-azobis(2-methylbutyronitrile) (V59) and the desired amounts of fluorescence dye PMI and photochromic dye CMTE. The monomer solution was poured into the aqueous solution and stirred for 1 h. Subsequently, the emulsion was homogenized by ultrasonication with a Branson digital sonifier 450-D (Dietzenbach, Germany) under ice-cooling using 90% amplitude, ½′′ tip for 120 s with a 10 s pulse–10 s pause program. The miniemulsion was transferred into a round bottom flask and polymerized under continuous stirring for 12 h at 72 °C. The resulting dispersion was filtered and dialyzed (membranes with MWCO 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, Carl Roth, Karlsruhe, Germany) for three days in demineralized water, changing the water twice a day. Particle sizes were determined by photon cross-correlation spectroscopy (Nanophox, Sympatec GmbH, Clausthal-Zellerfeld, Germany). The solid content was determined gravimetrically after freeze-drying.
000 g mol−1, Carl Roth, Karlsruhe, Germany) for three days in demineralized water, changing the water twice a day. Particle sizes were determined by photon cross-correlation spectroscopy (Nanophox, Sympatec GmbH, Clausthal-Zellerfeld, Germany). The solid content was determined gravimetrically after freeze-drying.
Seeded emulsion polymerization
A dispersion of 100 mL Milli-Q water containing 0.1 wt% of seed particles and 0.01 wt% of SDS was heated to 75 °C under continuous stirring in a three necked round bottom flask equipped with a condenser and septa. Argon gas was bubbled through the dispersion for 30 min. 500 mg of the initiator ammonium peroxodisulfate (APS) were dissolved in 3 mL water and added using a syringe to the stirred dispersion. 2 g of styrene and 150 mg of acrylic acid were mixed and added with a syringe pump at a flow rate of 1 mL h−1 to the stirred dispersion. Once the addition of monomer was finished, the reaction was left for polymerization at 80 °C for 24 h under an argon atmosphere. The dispersion was filtered and dialyzed (membranes with MWCO 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, Carl Roth, Karlsruhe, Germany) for three days in demineralized water changing the water twice a day.
000 g mol−1, Carl Roth, Karlsruhe, Germany) for three days in demineralized water changing the water twice a day.
Self-assembly of colloidal monolayers on a Langmuir trough
Prior use, the trough (242 cm2) was thoroughly cleaned with ethanol and several times with Milli-Q water. For spreading the colloidal dispersions (1.5 wt%) were purified with an ethanol–water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) by centrifugation and redispersion. Subsequently, the dispersions were diluted with ethanol to a final dispersion of 1.5 wt% containing 50 vol% ethanol. The dispersion was added to the interface via a tilted glass slide reaching into the subphase.45,46 After 15 min, the monolayer was compressed at a speed of 20 mm min−1 and deposited onto a silicon substrate by surface lowering transfer.19 The monolayers were analyzed using scanning electron microscopy (SEM, LEO (Zeiss) 1530 Gemini, Oberkochen, Germany), fluorescence and confocal laser scanning microscopy (CLSM, LSM SP5 STED Leica, Germany).
1 v/v) by centrifugation and redispersion. Subsequently, the dispersions were diluted with ethanol to a final dispersion of 1.5 wt% containing 50 vol% ethanol. The dispersion was added to the interface via a tilted glass slide reaching into the subphase.45,46 After 15 min, the monolayer was compressed at a speed of 20 mm min−1 and deposited onto a silicon substrate by surface lowering transfer.19 The monolayers were analyzed using scanning electron microscopy (SEM, LEO (Zeiss) 1530 Gemini, Oberkochen, Germany), fluorescence and confocal laser scanning microscopy (CLSM, LSM SP5 STED Leica, Germany).
UV-VIS and fluorescence spectroscopy
The absorption spectra of the photochromic dye in solution were obtained using a Perkin Elmer Lambda 25 UV-VIS spectrometer. 3 mL of a solution with a concentration of c = 1 × 10−6 mol L−1 of the photochrome in the corresponding monomer (S or BA) was analyzed. Fluorescence emission spectra were recorded on a TIDAS 3D fluorescence spectrometer with an excitation wavelength of λex = 490 nm. For the measurements in monomer 3 mL of a solution of PMI with a concentration of c = 5.2 × 10−6 mol L−1 were used. The investigations of the colloidal dispersions were made using 3 mL with a solid content of 1.5 wt%.For irradiation a mercury short arc lamp (HBO 100 W/2, 100 W, Osram) was used. The wavelength was adjusted with different optical band filters. For UV-irradiation a dark violet band pass (UG 1, Schott, 270–430 nm) and for VIS-light a combination of a light blue broad band (BG39, Schott) and a yellow optical filter (OG515, Schott) were used transmitting light within a wavelength range of 515 to 690 nm. To investigate the thermal stability of the system the samples irradiated with UV-light were covered with aluminum foil and stored in the dark at room temperature recording emission spectra after given times.
For the photoswitching of the functional colloidal monolayers an optical wide field fluorescence microscope (Olympus IX81, inverted fluorescence microscope, Hamburg, Germany) with a 2.5× objective (UIS2), a 100 W halogen lamp and different filters, such as a DAPI filter (UMNU 2, λex = 360 nm and λem = 420 nm) for the UV-light irradiation and an eGFP filter (U-MF2, λex = 472 nm and λem = 520 nm) for the VIS-light irradiation were used. The sample was alternately irradiated for 5 min with either UV or VIS-light and images were taken using the eGFP filter for the excitation of PMI.
Physicochemical characterization of nanoparticles
Each dispersion was characterized using photon cross-correlation spectroscopy with auto-NNLS analysis for the determination of the particle size and size distribution with σ as standard deviation. Differential scanning calorimetry was used for the analysis of glass transition temperatures. The corresponding results are summarized in Table 1.Polystyrene colloids produced by miniemulsion polymerization had an average diameter of around 90 nm with a size distribution of 17%, whereas the PBA colloids were about 155 to 160 nm in size with a distribution of 13–19%. The corresponding glass transition temperatures were measured by DSC and result around 71 °C for polystyrene and −56 and −49 °C for the poly(butyl acrylate) particles. After seeded emulsion polymerization the hybrid core–shell PS–PS particles had a size of d = 191 nm with a size distribution of 9%, whereas the PBA–PS particles were larger with d = 260 nm and a size distribution of 12%. The resulting Tg was similar after the seeded emulsion polymerization step with around 90 °C for both batches (PS–PS, PBA–PS). Compared to the colloids from miniemulsion the glass transition temperature is about 20 °C higher. For the miniemulsion technique hexadecane is used as an osmotic agent against Ostwald's ripening, lowering the glass transition temperature of the polystyrene colloids. With an additional rigid shell of polystyrene containing less or even no hexadecane, a higher glass transition temperature is reached. The amount of PBA was too low (23%) to see both Tg steps in the thermogram. SEM images of all particles were obtained (see ESI 1†).
Conclusion
In summary, we present an easy and fast method for the production of functional colloidal monolayers with reversibly photoswitchable colloids. The photoswitchable functional polystyrene and poly(butyl acrylate) colloids were synthesized using the miniemulsion polymerization process to incorporate the appropriate dye system based on a perylene as a fluorophore (N-(2,6-diisopropylphenyl)-perylene-3,4-dicarboximide) and a bis-arylethene as a photochrome (cis-1,2-dicyano-1,2-bis-(2,4,5-trimethyl-3-thienyl)ethene). Thus, addressable photoswitchable entities are generated.We investigate light-induced switching of the fluorescence intensity of the fluorophore via selectively enabling and disabling energy transfer to the photochrome molecule in the confined environment of a colloidal particle. By embedding the dyes in a polymeric matrix of colloids the thermal stability of the photochrome in the excited ring-closed state can be increased by a factor of more than 20 compared to the stability in solution. Information stored in the colloids by the photoswitching process can thus be retained over at least several days without risking of degradation by uncontrolled back reactions. Moreover, the precise incorporation of defined amounts of the dye molecules by miniemulsion polymerization can be used to optimize the quenching of the fluorophore emission. The light-induced switching of fluorescence is completely reversible and cyclable without observation of intensity loss. Functional colloidal monolayers were prepared using the co-assembly method of colloids of two distinct sizes at the air–water interface. The obtained binary monolayers of functional seeded particles and plain larger particles showed high crystallinity of several hundreds of μm2 resulting in addressability of the functional colloids. The possible arrangements for the seeded particles located at the interstices of the hexagonally ordered template particles were assessed from the scanning electron micrographs, resulting in a preferred number of three particles at the interstices for the seeded polystyrene as well as for the seeded poly(butyl acrylate) particles. The preparation method of functional binary monolayers offers the possibility of self-assembly of colloids with broad size distribution into highly ordered hexagonal lattices. Therefore, even single particles or smaller particle arrangements can be addressed. We demonstrate successful reversible optically induced storage and elimination of information in such monolayer structures.
We envision this technology to be of broad interest in information technology as it provides a cheap, simple and efficient way to generate precise substrates for optical manipulation at micro- and nanoscale.
Acknowledgements
Gabriele Schäfer is thanked for providing the 1150 nm carboxylated polystyrene colloids, Petra Räder for DSC measurements, and Dr Sandra Ritz for her help in confocal laser scanning microscopy. NV acknowledges funding from the Leopoldina Fellowship Program.References
- J. Zhang, Y. Li, X. Zhang and B. Yang, Adv. Mater., 2010, 22, 4249–4269 CrossRef CAS PubMed.
- X. Z. Ye and L. M. Qi, Nano Today, 2011, 6, 608–631 CrossRef CAS PubMed.
- S. John, Phys. Rev. Lett., 1987, 58, 2486–2489 CrossRef CAS.
- S. G. Romanov, N. Vogel, K. Bley, K. Landfester, C. K. Weiss, S. Orlov, A. V. Korovin, G. P. Chuiko, A. Regensburger, A. S. Romanova, A. Kriesch and U. Peschel, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 86, 195145 CrossRef.
- C. B. Murray, C. R. Kagan and M. G. Bawendi, Annu. Rev. Mater. Sci., 2000, 30, 545–610 CrossRef CAS.
- G. Zhang and D. Wang, Chem.–Asian J., 2009, 4, 236–245 CrossRef CAS PubMed.
- U. C. Fischer and H. P. Zingsheim, J. Vac. Sci. Technol., 1981, 19, 881–885 CrossRef CAS.
- M. Retsch, M. Tamm, N. Bocchio, N. Horn, R. Förch, U. Jonas and M. Kreiter, Small, 2009, 5, 2105–2110 CrossRef CAS PubMed.
- C. L. Haynes, A. D. McFarland, M. T. Smith, J. C. Hulteen and R. P. Van Duyne, J. Phys. Chem. B, 2002, 106, 1898–1902 CrossRef CAS.
- M. R. Gonçalves, T. Makaryan, F. Enderle, S. Wiedemann, A. Plettl, O. Marti and P. Ziemann, Beilstein J. Nanotechnol., 2011, 2, 448–458 CrossRef PubMed.
- M. Domanski, R. Luttge, E. Lamers, X. F. Walboomers, L. Winnubst, J. A. Jansen and J. G. E. Gardeniers, Nanotechnology, 2012, 23, 065306 CrossRef CAS PubMed.
- N. Vogel, J. Fischer, R. Mohammadi, M. Retsch, H.-J. Butt, K. Landfester, C. K. Weiss and M. Kreiter, Nano Lett., 2011, 11, 446–454 CrossRef CAS PubMed.
- A. J. Haes, S. Zou, G. C. Schatz and R. P. Van Duyne, J. Phys. Chem. B, 2003, 108, 109–116 CrossRef.
- M. Kolle, P. M. Salgard-Cunha, M. R. J. Scherer, F. M. Huang, P. Vukusic, S. Mahajan, J. J. Baumberg and U. Steiner, Nat. Nanotechnol., 2010, 5, 511–515 CrossRef CAS PubMed.
- K. A. Willets and R. P. Van Duyne, Annu. Rev. Phys. Chem., 2007, 58, 267–297 CrossRef CAS PubMed.
- Y. F. Li, J. H. Zhang, S. J. Zhu, H. P. Dong, F. Jia, Z. H. Wang, Z. Q. Sun, L. Zhang, Y. Li, H. B. Li, W. Q. Xu and B. Yang, Adv. Mater., 2009, 21, 4731–4734 CAS.
- N. Vogel, C. K. Weiss and K. Landfester, Soft Matter, 2012, 8, 4044–4061 RSC.
- R. McGorty, J. Fung, D. Kaz and V. N. Manoharan, Mater. Today, 2010, 13, 34–42 CrossRef CAS.
- N. Vogel, L. de Viguerie, U. Jonas, C. K. Weiss and K. Landfester, Adv. Funct. Mater., 2011, 21, 3064–3073 CrossRef CAS.
- K. P. Velikov, C. G. Christova, R. P. A. Dullens and A. van Blaaderen, Science, 2002, 296, 106–109 CrossRef CAS PubMed.
- V. Kitaev and G. A. Ozin, Adv. Mater., 2003, 15, 75–78 CrossRef CAS.
- S. G. Romanov, S. Orlov, A. V. Korovin, O. Zhuromskyy, N. Vogel, K. Landfester, C. K. Weiss and U. Peschel, Interplay of Mie and Bragg resonances in partly ordered monolayers of colloidal spheres, Proc. SPIE, 2012, 8425, 84250V CrossRef PubMed.
- Z. Zhou, Q. Yan, Q. Li and X. S. Zhao, Langmuir, 2006, 23, 1473–1477 CrossRef PubMed.
- G. Singh, H. J. Griesser, K. Bremmell and P. Kingshott, Adv. Funct. Mater., 2011, 21, 540–546 CrossRef CAS.
- G. Singh, S. Pillai, A. Arpanaei and P. Kingshott, Adv. Mater., 2011, 23, 1519–1523 CrossRef CAS PubMed.
- G. Singh, V. Gohri, S. Pillai, A. Arpanaei, M. Foss and P. Kingshott, ACS Nano, 2011, 5, 3542–3551 CrossRef CAS PubMed.
- J. P. Spatz, T. Herzog, S. Mößmer, P. Ziemann and M. Möller, Adv. Mater., 1999, 11, 149–153 CrossRef CAS.
- G. Roman, M. Martin and P. S. Joachim, Nanotechnology, 2003, 14, 1153 CrossRef.
- A. Manzke, A. Plettl, U. Wiedwald, L. Han, P. Ziemann, E. Schreiber, U. Ziener, N. Vogel, C. K. Weiss, K. Landfester, K. Fauth, J. Biskupek and U. Kaiser, Chem. Mater., 2012, 24, 1048–1054 CrossRef CAS.
- N. Vogel, C. Fernández-López, J. Pérez-Juste, L. M. Liz-Marzán, K. Landfester and C. K. Weiss, Langmuir, 2012, 28, 8985–8993 CrossRef CAS PubMed.
- A. Manzke, N. Vogel, C. K. Weiss, U. Ziener, A. Plettl, K. Landfester and P. Ziemann, Nanoscale, 2011, 3, 2523–2528 RSC.
- D. Crespy and K. Landfester, Polymer, 2009, 50, 1616–1620 CrossRef CAS PubMed.
- E. Schreiber, U. Ziener, A. Manzke, A. Plettl, P. Ziemann and K. Landfester, Chem. Mater., 2009, 21, 1750–1760 CrossRef CAS.
- N. Vogel, U. Ziener, A. Manzke, A. Plettl, P. Ziemann, J. Biskupek, C. K. Weiss and K. Landfester, Beilstein J. Nanotechnol., 2011, 2, 459–472 CrossRef CAS PubMed.
- N. Vogel, C. P. Hauser, K. Schuller, K. Landfester and C. K. Weiss, Macromol. Chem. Phys., 2010, 211, 1355–1368 CrossRef CAS.
- C. K. Weiss and K. Landfester, Adv. Polym. Sci., 2012, 233, 185–236 CrossRef.
- H. Furukawa, M. Misu, K. Ando and H. Kawaguchi, Macromol. Rapid Commun., 2008, 29, 547–551 CrossRef CAS.
- M. Irie and M. Morimoto, Pure Appl. Chem., 2009, 81, 1655–1665 CrossRef CAS.
- S. Nakamura, S. Yokojima, K. Uchida, T. Tsujioka, A. Goldberg, A. Murakami, K. Shinoda, M. Mikami, T. Kobayashi, S. Kobatake, K. Matsuda and M. Irie, J. Photochem. Photobiol., A, 2008, 200, 10–18 CrossRef CAS PubMed.
- T. Fukaminato, T. Umemoto, Y. Iwata, S. Yokojima, M. Yoneyama, S. Nakamura and M. Irie, J. Am. Chem. Soc., 2007, 129, 5932–5938 CrossRef CAS PubMed.
- F. K. Hansen and J. Ugelstad, J. Polym. Sci., Part A: Polym. Chem., 1979, 17, 3047–3067 CrossRef CAS.
- X. Z. Kong and E. Ruckenstein, J. Appl. Polym. Sci., 1999, 73, 2235–2245 CrossRef CAS.
- Z. Y. Li, H. Cheng and C. C. Han, Macromolecules, 2012, 45, 3231–3239 CrossRef CAS.
- CRC Chemical Handbook of Chemistry and Physics, ed. W. M. Haynes, T. J. Bruno and D. R. Lide, 94th edn, 2013, online version Search PubMed.
- N. Vogel, S. Goerres, K. Landfester and C. K. Weiss, Macromol. Chem. Phys., 2011, 212, 1719–1734 CrossRef CAS.
- S. M. Weekes, F. Y. Ogrin, W. A. Murray and P.S. Keatley, Langmuir, 2007, 23, 1057–1060 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: S1: scanning electron micrographs of the particles prepared by miniemulsion polymerization and seeded emulsion polymerization, S2: fluorescence emission spectra of the primary colloidal dispersions and the development of PMI's emission during UV-light and VIS-light irradiation, S3: calculations for the size of the interstitial sites of the template colloids and detailed geometric evaluation of the possible assignment of seeded particles, and S4: optical fluorescence micrographs for the photoswitching of a functional colloidal monolayer. See DOI: 10.1039/c3nr04897g |
| This journal is © The Royal Society of Chemistry 2014 |