Bright-field electron tomography of individual inorganic fullerene-like structures†
Maya
Bar Sadan
*a,
Sharon G.
Wolf
b and
Lothar
Houben
a
aInstitute of Solid State Research, Ernst Ruska-Centre for Microscopy and Spectroscopy with Electrons, Research Centre Jülich GmbH, 52425 Jülich, Germany. E-mail: m.bar-sadan@fz-juelich.de
bElectron Microscopy Unit, Weizmann Institute of Science, Rehovot 76100, Israel
First published on 8th December 2009
Abstract
Nanotubes and fullerene-like nanoparticles of various inorganic layered compounds have been studied extensively in recent years. Their characterisation on the atomic scale has proven essential for progress in synthesis as well as for the theoretical modelling of their physical properties. We show that with electron tomography it is possible to achieve a reliable reconstruction of the 3D structure of nested WS2 or MoS2 fullerene-like and nanotube structures with sub-nanometre resolution using electron microscopes that are not aberration-corrected. Model-based simulations were used to identify imaging parameters, under which structural features such as the shell structure can be retained in the tomogram reconstructed from bright-field micrographs. The isolation of a particle out of an agglomerate for the analysis of a single structure and its interconnection with other particles is facilitated through the tomograms. The internal structure of the layers within the particle alongside the shape and content of its internal void are reconstructed. The tomographic reconstruction yields insights regarding the growth process as well as structural defects, such as non-continuous layers, which relate to the lubrication properties.
Introduction
The application of functional materials on the nanoscale requires syntheses that controls shape, size and atomic architecture. The knowledge of the three-dimensional structure and composition on the lattice length-scale holds the information about the unique physical properties of nanomaterials compared with their bulk ancestors. The structural resolution required is usually in the range of a few nm, the typical size of the active nanoparticles, while crucial information on the crystalline structure is achieved only at higher resolution. A rapidly progressing technique for the study of materials on the nanoscale in 3D is tomography in the transmission electron microscope (TEM).1 In particular catalysts are extensively investigated by electron tomography2 because of the close relationship between 3D structure and functionality.The present manuscript focuses on high-resolution electron tomographic reconstruction of inorganic nanostructures from bright-field images recorded in the TEM. It is well established that a rich variety of inorganic materials can be used for the fabrication of closed-cage nanostructures.3,4 It has been proposed5,6 that the formation of fullerenes is a genuine property of two-dimensional (layered) compounds, such as WS2. The WS2 molecular sheet is composed of a layer of tungsten atoms sandwiched between two outer sulfur layers, each of them covalently bonded and the sheets are held together by van der Waals interactions, just like graphite. A rich variety of materials can be used for the fabrication of inorganic nanotubes (INT) and inorganic fullerene-like nanoparticles (IF) by diverse chemical routes.3,4 The experimental data shows that this hollow phase of layered compounds is the thermodynamically stable form, given the constraint that the particles can be arranged in a concentric seamless form in an onion-like morphology. Lately even more complex structures have been produced, for example core–shell nanotubes where PbI2 crystallizes within the WS2 template.7
The burgeoning large-scale applications of IF as superior solid lubricants8–10 and as reinforcing materials for ultra-high-strength nanocomposites make the detailed evaluation of their structure and growth mechanism highly warranted. If it is possible to decipher the exact atomic arrangement of these structures down to the atomic level, such knowledge will provide insight into their growth mechanism11 and may serve as a basis for theoretical calculations, predicting their electronic properties.12,13 Layer defects, surface reconstructions and capping defects associated with the closure of the nanostructures are of particular interest since these determine their physical properties.
We present experimental high-resolution electron tomograms of nested tungsten disulfide and molybdenum disulfide structures, both onion-like fullerenes and nanotubes, which allow the material to be studied in three dimensions down to the sub-nm level. The 3D reconstruction is used for these structures since the important information resides at the cross section of the tubes or onions and is not accessible via regular 2D images. The interference phenomena associated with bright-field imaging hinder accurate tomographic reconstruction. However, by using image simulations of model structures, proper setting of experimental conditions can be established and high-resolution tomograms can be achieved.
Results and discussion
Projection requirement
While bright-field electron tomography is widespread in biological sciences for the examination of biological macromolecules, cell sections, tissues and entire cells,14–16 there has been a reluctance to use bright-field tomography as a basic tool for the analysis of solid-state structures in the physical sciences. The perceived major barrier involves inherent problems associated with bright-field contrast. In order to obtain a meaningful 3D reconstruction, the 2D images are required to be true projections of the structure. For conventional reconstruction algorithms the intensity of the images is required to represent a monotonic function of the amount of material projected parallel to the electron beam.17 This requirement has been termed ‘projection requirement’.18 For crystalline materials whose major components have high atomic Z-numbers, the contrast in the bright-field imaging mode is dominated by diffraction and multiple beam interferences. Therefore, the bright-field image of a crystal might be the result of a diversion from the monotonic dependence of the intensity on the thickness of the sample and contradict the projection requirement.Why bright-field tomography?
Despite the complication of non-linear diffraction and optical imaging, bright-field images can have invaluable advantages. A good signal-to-noise ratio at low dose can be achieved in bright-field. Images are free from unsystematic distortions for a complete tilt series, contrary to scanning imaging techniques where high-resolution information down to the atomic-scale is subject to random fluctuations in magnification and image distortion. In addition, the sensitivity for single-atom imaging even for light elements makes bright-field tomography in general a good candidate for high-resolution tomography.One example in which bright-field images were successfully applied in catalyst research using electron tomography was presented by Koster and co-workers.19 Tomography was used for the first time to detect the 3D structure of catalysts (composites of a matrix and active nanoparticles), using amorphous materials where the mass and thickness of the sample dominates the contrast. The resolution required is usually in the range of a few nm, the typical size of the active nanoparticles, while crucial information on the crystalline structure demands achieving even higher resolution. Indeed, sub-nanometre information was achieved using bright-field tomography in another paper by de Jong et al.20 who for the first time resolved 0.6 nm spaced MoS2 Bragg fringes inside a commercial Ni–Mo/γ–Al2O3 catalyst.
Pre-conditions for the reconstruction from bright-field images
Bright-field tomography does challenge the ‘projection requirement’ at higher resolution better than one nm and care has to be taken in order to maintain linearity between image intensity and atom density. For example, when Bragg diffraction effects are present, fringe maxima and minima may not be identified with atomic layers. We therefore demonstrate first that reliable 3D reconstructions with true structure resolution can be achieved for the wide range of closed-cage nanomaterials. In these structures, the complicated 3D structure prevents the occurrence of atomic columns in the transmission direction of the electron beam, resulting in negligible Bragg diffraction effects.Another obstacle to be considered is non-linear interference in the electron–optical imaging process, which might well break the correspondence between image intensity and a structural feature such as the molecular shell. In other words, in images taken at different defocus values, the same atomic layer might be represented once as a dark fringe and once as a bright fringe, preventing a meaningful reconstruction. However, when a specific focal range is kept during the acquisition of the tilt series, fringes in the reconstruction of the nanostructures truly correspond to shell layers. A simulated focal-series of a WS2 four-shell nanotube was produced in order to give evidence for the latter. The model of a nanotube was chosen for this simulation because its one-dimensional periodicity helps to avoid excessive calculation time. Still the conclusion remains the same for the onion-like IF structures with their two-dimensionally curved layers. The atomic structure of the model INT taken for calculation is based on previously reported experimental evidence.11 In a practical experiment on nanotubes it is important that the tube axes are aligned parallel to the tilt rotation axis. This setup will ensure that the full length of the nanotube stays within a narrow focus range, preventing contrast reversals across the object. This is important, however, not just for bright-field tomography, but also for any high-resolution electron tomography.
Fig. 1a–c shows the model of the nanotube with an outer diameter of 19 nm, the projected electrostatic potential map, an image simulated for the conventional Scherzer defocus and an image for an underfocus of 160 nm in a 200 kV FEG-TEM. The effects of continuous focal variation on the fringe image (representing the shells of the nanotube) is shown in Fig. 1d. The high-density projected potential along each of the four walls of the nanotube is represented as a dark fringe in the Scherzer image, marked with red arrows. In contrast, the simulated image for an underfocus of 160 nm serves as an example to the possible contrast reversals. Four red arrows, located in the corresponding position to the four dark fringes on the Scherzer image, now point to two white fringes and two gray ones. In contrast to solid crystalline structures, dynamical diffraction effects are unlikely for the curved layered structures even for particle sizes of more than 10 nm. Moreover, the fringe contrast shows no contrast reversal within a range of defocus between −40 nm and −130 nm underfocus, around the Scherzer defocus of −63 nm. Keeping the focus in this range during the acquisition of the tilt series will henceforth avoid artefacts in the tomographic reconstruction due to contrast reversal of the shell fringes with respect to the underlying structure. As a result, an object feature which is represented as a dark fringe in one image, can be kept dark in the rest of the tilt series as long as the defocus range mentioned above is maintained.
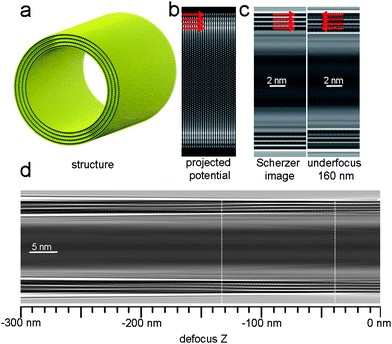 | ||
| Fig. 1 Imaging of WS2 nanotubes by TEM: (a) The ball and stick model of a multiwall WS2 nanotube, 19 nm in diameter, based on experimental finding16. W in blue, S in yellow. The projected electrostatic potential is presented in (b) alongside simulated TEM images of the same tube section (accelerating voltage 200 kV, spherical aberration constant: 1.2 mm, Scherzer defocus: −63 nm and an underfocus of 160 nm) (c). The red arrows mark the location of high-density potential, where the tube walls are located, and the corresponding areas in the simulated images. In (d), a simulated TEM image of the tube in various defoci is presented, from zero focus to −300 nm defocus. In white dashed line, two regions of contrast reversals are marked. | ||
Once identified for a particular structure of interest and a particular microscope, the setting of the favourable conditions for each image in the tilt-series is a precondition for a reliable tomographic reconstruction. In all application examples given below, the tilt series of bright-field images were taken according to the favourable conditions identified above.
Tomographic reconstruction of WS2 nanotubes
In practice, WS2 nanotubes (INT-WS2) are often found in bundles. The different structures are entangled in such a way, that identifying a single tube and interpreting its internal structure from a single 2D projection is complicated or even impossible. In contrast, a tomographic reconstruction enables manipulation of the structures in different viewing directions, including cross-sectional views. Such a case is presented in Fig. 2. A bundle of INT-WS2 is seen in a conventional 2D projection in Fig. 2a. The isolation of a single tube is not feasible, and the question is also raised, whether residual tungsten oxide, one of the reactants, is present within the tube or as an external contaminant on the outer walls of the tube. Figs. 2b and 2d–e show tomogram slices of the bundle. The different tubes can be resolved as well as their shell spacing of 0.62 nm. The number of shells can be identified for each of the tubes from the cross-section of the tomogram, unlike in the 2D image where only the number of layers of the outermost tube is unambiguous. The 3D voxel projection of the semi-transparent volume in Fig. 2c visualizes that the tube is still partially filled with residual material, by the opaqueness inside the tube. These nanotubes thus contain residual tungsten oxide in their interior due to incomplete oxide consumption during the reduction–sulfidation reaction. When slicing through the tomogram in the tube direction, slices with inner voids (such as the one in Fig. 2d) alternate with slices showing amorphous material inside the tube (marked by the arrows in Fig. 2e), in accordance with the alternating density seen in the 2D images. However, in the tomogram it is possible to identify the filled and empty segments for each of the tubes at the same time, when in the 2D images only the outermost ones can be resolved. Thus the tomographic reconstruction can be used for the reliable assessment of the fraction filling of composite tubes, where different compounds were crystallized within the inner space of tubes. This is an important issue for technological applications affecting the tube properties as well as an obstacle for further synthesis steps, meant to produce core–shell tubes from template WS2 tubes such as metal–halide core–shell tubes.7 If an oxide is present within or on the tube, additional procedures need to be applied in order to empty and clean the internal surface of the tube. Additional material, including the tilt series and tomogram slice and projection movies, is available in the ESI (movies S1 and S2†). | ||
| Fig. 2 Single-axis tomographic reconstruction of a bundle of WS2 nanotubes. An image from the tilt series, around zero tilt, is presented in (a). A corresponding slice of the reconstruction matching the frame marked in (a) is presented in (b), with the density inverted. The voxel view, representing the 3D data, is shown in (c). Two slices of the reconstruction, showing the cross-section of several tubes, are presented in (d) and (e), once with the lower tube empty (d) and once where the same tube is partially filled, probably with residual WOx, marked by the arrows (e). | ||
Tomographic reconstruction of onion-like inorganic fullerenes (IF)
The difficulty in isolating a single particle out of an agglomerate is also a concern when IF are examined. Snapshots of the tomographic recording and reconstruction of agglomerated IF-MoS2 nanoparticles are shown in the ESI.† In Fig. S1, a single image from the tilt series shows the superposition of information from multiple structures one on top of the other. Using the full tilt series, a 3D reconstruction of the agglomerate volume was carried out (see movies S3 and S4). From this 3D volume, one particle circled in red was carved out and visualized by 3D rendering (see movie S5).Working in a low-dose mode, which allows the addition of a second tilt axis to the reconstruction, leads to a further improvement of the tomogram with respect to a single-tilt axis reconstruction. The dual-axis tomography vastly decreases distortions due to the missing information in the transmission direction.21 The tracking of the object and the focusing in a low-dose mode are done on another part of the sample, enabling minimal exposure of the object to the irradiating beam and large number of acquisitions. The reconstruction of an isolated IF particle shown in Fig. 3 was based on dual-axis tilt series. Fig. 3a is a single image out of one tilt series, around zero tilt. Fig. 3b is the corresponding slice from the tomogram, showing the fringes of the atomic layers and the void of the particle. Additional slices of the tomogram, showing all three possible viewing directions, are available in the ESI as Fig. S2.† The 3D reconstruction in a voxel view is presented in Fig. 3c. The voxel representation is a visualization of the volumetric data by 2D projections of a colored, semi-transparent volume, allowing the understanding of the 3D structure of the particle without segmentation of the slices, retaining all the information of the tomogram. The particle and the gold markers are visible (see also movie S6) and the internal void and the surrounding shells are resolved and can be traced throughout the particle. At both edges of the particle in the axis of the missing wedge, elongation and deterioration of the information are still seen (marked by white arrows in Fig. 3d) as expected since this cannot be avoided given the restrictions on the tilt range. In order to draw the outer shell of the particle, the tomogram slices were segmented manually and the resulting surface is shown in Fig. 3d. While in Fig. 3c the estimation of the internal structure is facilitated, the outer surface of the particle is produced in Fig. 3d, showing an apex and edges which connect the particle's facets.
![Dual-axis tomographic reconstruction of a single IF-WS2 particle. An image from the tilt series, around zero tilt, is presented in (a). A corresponding slice of the reconstruction is presented in (b). [Additional views are available as Fig. S2 of the ESI]. The voxel view, representing the 3D data, is shown in (c). The gold particles seen in the surrounding of the particle were added to the sample as markers for alignment. The outer surface of the particle is shown in (d). The white arrows mark the degradation of the information as a result of the missing wedge.](/image/article/2010/NR/b9nr00251k/b9nr00251k-f3.gif) | ||
| Fig. 3 Dual-axis tomographic reconstruction of a single IF-WS2 particle. An image from the tilt series, around zero tilt, is presented in (a). A corresponding slice of the reconstruction is presented in (b). [Additional views are available as Fig. S2 of the ESI†]. The voxel view, representing the 3D data, is shown in (c). The gold particles seen in the surrounding of the particle were added to the sample as markers for alignment. The outer surface of the particle is shown in (d). The white arrows mark the degradation of the information as a result of the missing wedge. | ||
The lattice fringes resolved for the IF-WS2 show the imperfect internal structure of the particle. An internal flat facet is seen in the middle of the void, not in accordance with previous observations, resulting in a wide crack and many defects surrounding it. These cracks and defects are the fingerprints of the reaction paths which control the morphology of the products.
The cross-sectional analysis of the internal structure of the particle gives useful insight into the processes by which these particles gain their superior lubrication qualities, e.g. by defect formation and exfoliation. On the other hand, non-continuous layers and edges in the structure of IFs (like the particle in Fig. 3), might be weak points in terms of mechanical strength, accelerating plastic deformation processes under mechanical load.
Prospects of high-resolution bright-field tomography
The experimental data proves that bright-field tomography can be performed with a resolution on the scale of layer spacings in inorganic fullerenes, i.e. with a resolution of 0.6 nm. As the obtained resolution of the electron tomography enters the sub-nm range, it is worthwhile discussing the attainable limit and possible approaches to overcome these problems. Missing wedge, tilt accuracy and accurate image alignment are common problems in electron tomography. The missing wedge artefacts can be reduced by using high-angle tilt holders22 or by dual-axis tomography as shown above. The effect of random and systematic errors in specimen tilt and image alignment demands for refinement steps during the reconstruction process. As such, the image alignment can be improved: for example for the nanotube reconstruction in Fig. 2 the alignment has been iteratively refined by cross-correlation to the re-projected tomogram.Furthermore, when aspiring to higher resolutions and to atomic scale, controlling the focal range is not sufficient, since the fact that the image is an interference pattern hinders the direct interpretation of the location of the atoms. One approach to overcome this problem was suggested by the authors.23 Acquiring the images in a Cs-corrected microscope and setting negative spherical aberration imaging (NCSI) conditions,24 yields images which are close representations of the projected potential for weakly scattering objects. These images show high sensitivity even for light elements such as oxygen.24,25 In the case of moderate density of the atomic nucleus, excellent linearity and superior signal-to-noise ratio can be achieved compared to more conventional imaging techniques. Therefore, under these conditions, the locations of the atoms can be directly interpreted from the image, opening the way to atomic scale tomography for weakly scattering objects. The use of aberration-corrected microscopes also eliminates the delocalization, which produces “ghost” fringes outside of the object. All of these are achieved for lower voltage operation modes, where the knock-on radiation damage is far less prominent, which is in many cases the practical limiting factor.
When applications and large-scale production are concerned, such ultra-high atomic scale resolutions are not always required and a reliable, fast technique, which can be achieved by the use of conventional electron microscopes, is appreciated.
In the present paper it was shown that the desired 3D investigation of inorganic fullerene-like particles and nanotubes is feasible through the use of conventional electron microscopes. The reliability of the reconstruction depends on imposing the right conditions during acquisition to eliminate contrast reversals, conditions which should be calculated prior to the experiment.
Experimental
Synthesis
WS2 inorganic fullerene-like particles were produced in the fluidized bed reactor.26 The precursors, in this case WO3 nanoparticles, were reacted with H2S at elevated temperatures (about 850 °C) to produce IF-WS2 closed-cage structures and WS2 nanotubes with a 95![[hair space]](https://www.rsc.org/images/entities/char_200a.gif) :
:![[hair space]](https://www.rsc.org/images/entities/char_200a.gif) 5% yield, respectively. IF-MoS2 nanoparticle synthesis and characterization is described in detail elsewhere.27,28
5% yield, respectively. IF-MoS2 nanoparticle synthesis and characterization is described in detail elsewhere.27,28
Lacey carbon (PLANO GmbH Wetzlar, Germany) or Quantifoil (Quantifoil GmbH, Jena Germany) grids were covered by a thin deposited film of amorphous carbon (a few nm thick). The grids were plasma etched and then decorated from both sides by 4 nm gold markers. The particles were suspended by sonication in ethanol and then spread on the grid.
Electron tomography
Tilt-angle series of WS2 nanoparticles and nanotubes were acquired with a Tecnai F20 (FEI, Cs = 1.2 mm) operated at 200 kV. For the NT-WS2 reconstruction, a microscope with a Super-Twin lens was used and bright-field single-tilt images were recorded manually at 2° intervals over a range from −56° to 64°. The 3D reconstruction was carried out using a weighted additive backprojection algorithm and the simultaneous iterative reconstruction technique (SIRT) provided by the FEI Xplore3D tomography package. Custom-written software was used for the refinement of the image alignment to the re-projection of the tomogram. For the IF-WS2 and IF-MoS2 reconstruction, a Tecnai F20 (FEI) microscope with a TWIN lens (Cs = 2.0 mm) was used and bright-field double-tilt images were recorded at 1.5° intervals over a range of ±65°, using the SerialEM program for automated tilt series collection.29 The low-dose mode of the SerialEM program was pre-set to acquire images with a dose of about 60 e·A−2 per image, a factor of 5 less in comparison to regular acquisition conditions. Image alignment and dual-axis 3D reconstruction using a weighted additive backprojection algorithm were done with the IMOD tomography package.30–32The background was removed and the contrast was normalized for each image in the tilt series prior to reconstruction. A first image alignment was done by cross-correlation of successive images in the tilt series followed by manual fine-tuning. Three-dimensional visualizations were performed using isosurface and voxel representations of the tomograms, created with the AMIRA visualization software (Mercury Computer Systems).
Image simulation
Simulated focal series of nanoparticles were calculated from supercell data containing the atomic coordinates of the structures. The supercell data for the multishell WS2 nanotubes were produced by geometrically rolling sheets of WS2. The supercells were divided into stacks of 0.2 nm thick slices for the multislice calculation of the interaction of the electron wave with the sample. The multislice iteration and the electron optical imaging were calculated using the EMS image calculation software.33 Microscope parameters for an instrument equipped with a Super-Twin lens were chosen. An aperture was applied to cut off the signal beyond the Scherzer-point resolution of the instrument of about 0.25 nm.Conclusion
High-resolution tomography based on TEM bright-field images was used to characterize the three-dimensional structure of inorganic fullerene-like nanostructures and nanotubes. Appropriate imaging conditions for the investigation in the transmission electron microscope were identified by image calculation in order to retain the correspondence between structural features and image intensities in the electron-optical imaging process. Experimental tomograms of inorganic nanostructures show their cross-sectional properties with sub-nm resolution. Remaining fillings and defects in the cross-section of the structures reveal details of the growth mechanism and relate to mechanical properties.Acknowledgements
The authors thank Prof. R. Tenne from the Weizmann Institute of Science for valuable discussions and Dr R. Rosentsveig and Dr L. F. Deepak for the sample preparation. M. B. S. acknowledges the Minerva Fellowship program funded by the German Federal Ministry for Education and Research, for the support of the G. M. J. Schmidt Minerva Center for Supramolecular Chemistry, the support of the Harold Perlman Foundation and ERC grant INTIF No. 226639. L. H. and M. B. S. thank the German Science Foundation (DFG) for their support of sub-Ångström microscopy at the Ernst Ruska Centre. Part of the electron microscopy studies were conducted at the Irving and Cherna Moskowitz Center for Nano and Bio-Nano Imaging at the Weizmann Institute of Science.References
- P. A. Midgley and R. E. Dunin-Borkowski, Nat. Mater., 2009, 8, 271 CrossRef CAS.
- H. Friedrich, P. E. de Jongh, A. J. Verkleij and K. P. de Jong, Chem. Rev., 2009, 109, 1613 CrossRef CAS.
- R. Tenne and G. Seifert, Annu. Rev. Mater. Res., 2009, 39, 387 CrossRef CAS.
- R. Tenne, Nat. Nanotechnol., 2006, 1, 103 CrossRef CAS.
- R. Tenne, L. Margulis, M. Genut and G. Hodes, Nature, 1992, 360, 444 CrossRef CAS.
- L. Margulis, G. Salitra, R. Tenne and M. Talianker, Nature, 1993, 365, 113 CrossRef CAS.
- R. Kreizman, S.-Y. Hong, J. Sloan, R. Popovitz-Biro, A. Albu-Yaron, G. Tobias, B. Ballesteros, B. G. Davis, M. L. H. Green and R. Tenne, Angew. Chem., Int. Ed., 2009, 48, 1230 CrossRef CAS.
- R. D. Luttrell, S. Brown, J. Cao, J. L. Musfeldt, R. Rosentsveig and R. Tenne, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73, 035410 CrossRef.
- L. Rapoport, Y. Bilik, Y. Feldman, M. Homyonfer, S. R. Cohen and R. Tenne, Nature, 1997, 387, 791 CrossRef CAS.
- M. Redlich, A. Katz, L. Rapoport, H. D. Wagner, Y. Feldman and R. Tenne, Dent. Mater., 2008, 24, 1640 CrossRef CAS.
- M. Bar Sadan, L. Houben, A. N. Enyashin, G. Seifert and R. Tenne, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 15643 CrossRef CAS.
- M. Bar-Sadan, A. N. Enyashin, S. Gemming, R. Popovitz-Biro, S. Y. Hong, Y. Prior, R. Tenne and G. Seifert, J. Phys. Chem. B, 2006, 110, 25399 CrossRef CAS.
- A. N. Enyashin, S. Gemming, M. Bar-Sadan, R. Popovitz-Biro, S. Y. Hong, Y. Prior, R. Tenne and G. Seifert, Angew. Chem., Int. Ed., 2007, 46, 623 CrossRef CAS.
- J. Frank, in Electron tomography: three-dimensional imaging with the transmission electron microscope, Plenum, New York, London, 1992 Search PubMed.
- A. J. Koster, R. Grimm, D. Typke, R. Hegerl, A. Stoschek, J. Walz and W. Baumeister, J. Struct. Biol., 1997, 120, 276 CrossRef CAS.
- B. F. McEwen and M. Marko, J. Histochem. Cytochem., 2001, 49, 553 CAS.
- M. Weyland, Top. Catal., 2002, 21, 175 CrossRef CAS.
- P. A. Midgley and M. Weyland, Ultramicroscopy, 2003, 96, 413 CrossRef CAS.
- A. J. Koster, U. Ziese, A. J. Verkleij, A. H. Janssen and K. P. de Jong, J. Phys. Chem. B, 2000, 104, 9368 CrossRef CAS.
- K. P. de Jong, L. C. A. van den Oetelaar, E. T. C. Vogt, S. Eijsbouts, A. J. Koster, H. Friedrich and P. E. de Jongh, J. Phys. Chem. B, 2006, 110, 10209 CrossRef CAS.
- I. Arslan, J. R. Tong and P. A. Midgley, Ultramicroscopy, 2006, 106, 994 CrossRef CAS.
- N. Kawase, M. Kato, H. Nishioka and H. Jinnai, Ultramicroscopy, 2007, 107, 8 CrossRef CAS.
- M. Bar Sadan, L. Houben, S. G. Wolf, A. Enyashin, G. Seifert, R. Tenne and K. Urban, Nano Lett., 2008, 8, 891 CrossRef.
- C. L. Jia, M. Lentzen and K. Urban, Science, 2003, 299, 870 CrossRef.
- M. Lentzen, Microsc. Microanal., 2006, 12, 191 CrossRef CAS.
- Y. Feldman, A. Zak, R. Popovitz-Biro and R. Tenne, Solid State Sci., 2000, 2, 663 CrossRef CAS.
- F. L. Deepak, A. Margolin, I. Wiesel, M. Bar-Sadan, R. Popovitz-Biro and R. Tenne, Nano, 2006, 1, 167 CrossRef CAS.
- F. L. Deepak, H. Cohen, S. Cohen, Y. Feldman, R. Popovitz-Biro, D. Azulay, O. Millo and R. Tenne, J. Am. Chem. Soc., 2007, 129, 12549–12562 CrossRef CAS.
- SerialEM Homepage, http://bio3d.colorado.edu/SerialEM/.
- IMOD Homepage, http://bio3d.colorado.edu/imod/.
- J. R. Kremer, D. N. Mastronarde and J. R. McIntosh, J. Struct. Biol., 1996, 116, 71 CrossRef.
- D. N. Mastronarde, J. Struct. Biol., 1997, 120, 343 CrossRef CAS.
- P. A. Stadelmann, Ultramicroscopy, 1987, 21, 131 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Figs. S1 and S2 and movies S1–S6. See DOI: 10.1039/b9nr00251k |
| This journal is © The Royal Society of Chemistry 2010 |
