Synthesis and self-assembly of a novel fluorinated triphilic block copolymer†
Abstract
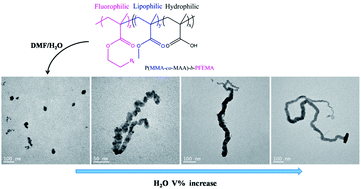
A novel fluorinated diblock tercopolymer was synthesized by the reversible addition fragmentation chain transfer method, incorporating methacrylic acid, methyl methacrylate, and perfluoroalkyl ethyl methylacrylate blocks as hydrophilic, lipophilic, and fluorophilic units, respectively. Depending upon the specific triphilic balance, under control solution conditions, the block copolymer self-assembled in dimethylformamide/water dispersions induced an evolution from spheres to wormlike structures, and finally to novel nail-shaped structures, as verified by transmission electron microscopy and dynamic light scattering. The control mechanism is explained in terms of the effect of each property on the forces that govern the formation of any given morphology, namely the core-chain stretching, strong incompatibility between the lipophilic blocks and the fluorophilic blocks, and interfacial tension.

 Please wait while we load your content...
Please wait while we load your content...