Co-delivery of 5-fluorocytosine and cytosine deaminase into glioma cells mediated by an intracellular environment-responsive nanovesicle†
Abstract
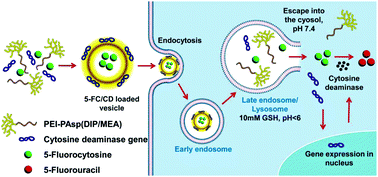
The diblock copolymer (PEI-PAsp(DIP/MEA)) of branched polyethyleneimine (PEI) and biodegradable poly(2-diisopropylamino/2-mercaptoethylamine) ethyl aspartate (PAsp(DIP/MEA)) was synthesized and assembled into nanovesicles for the co-delivery of cytosine deaminase (CD) gene and 5-fluorocytosine (pCMVCD/5-FC) into glioma cells. The nanovesicle comprises a hydrophilic outerlayer of branched PEI for pCMVCD complexation, a hydrophilic inner cavity for prodrug 5-FC loading, and a pH-sensitive membrane crosslinked by disulfide to prevent drug leakage into the bloodstream-simulating environment with neutral pH and without reducing agent. Yet, once the PPDM (PEI-PAsp(DIP/MEA)) nanovesicle was internalized into cancer cells and entrapped inside lysosomes featuring a low pH (∼5) and enriched reducing agent (∼10 mM GSH), it dissociated as a result of tertiary amine protonation and disulfide bond breakage to release the loaded drug. The in vitro release studies showed that less than 10% of 5-FC was released from the nanovesicle in 24 h at pH 7.4, whereas about 98% of 5-FC was released in the presence of 10 mM GSH at pH 5.0. The prodrug 5-FC was converted into its toxic active metabolite 5-fluorouracil (5-FU) by cytosine deaminase inside C6 glioma cells, owing to the effective CD gene transfection and expression. As a result, the 5-FC/pCMVCD-loaded nanovesicle induced effective apoptosis and generated a significant cytotoxic effect in the cancer cells. Our results indicated that this multifunctional nanovesicle is a promising carrier for the co-delivery of gene and drug or prodrug in cancer therapy.

 Please wait while we load your content...
Please wait while we load your content...