Synthesis and thermal behavior of tin-based alloy (Sn–Ag–Cu) nanoparticles†
Abstract
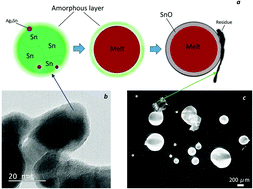
The prominent melting point depression of nanoparticles has been the subject of a considerable amount of research. For their promising applications in electronics, tin-based nano-alloys such as near-eutectic Sn–Ag–Cu (SAC) alloys have been synthesized via various techniques. However, due to issues such as particle aggregation and oxidation or introduced impurities, the application of these nano-size particles has been confined or aborted. For instance, thermal investigations by DTA/DSC in a large number of studies revealed exothermic peaks in the range of 240–500 °C, i.e. above the melting point of SAC nanoparticles, with different and quite controversial explanations for this unclear phenomenon. This represents a considerable drawback for the application of nanoparticles. Correspondingly, in the current study, the thermal stability of SAC nanoparticles has been investigated via electron microscopy, XRD, FTIR, and DSC/TG analysis. It was found that the nanoparticles consist mainly of a metallic β-Sn core and an amorphous tin hydroxide shell structure. The SnO crystalline phase formation from this amorphous shell has been associated with the exothermic peaks on the first heating cycle of the nanoparticles, followed by a disproportionation reaction into metallic Sn and SnO2.The results also revealed that the surfactant and reducing agent cannot only affect the size and size distribution of the nanoparticles, they might also alter the ratio between the amorphous shell and the crystalline core in the structure of particles.

 Please wait while we load your content...
Please wait while we load your content...