Using imidazolium-based ionic liquids as dual solvent-catalysts for sustainable synthesis of vitamin esters: inspiration from bio- and organo-catalysis†
Abstract
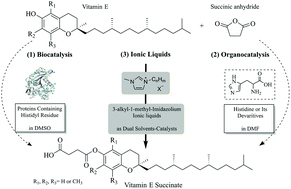
Vitamin E (VE) has significant biological activities and thus its acylation to increase its stability is of extreme interest. We developed an efficient and sustainable approach using imidazolium-based ionic liquids as dual solvent-catalysts for the esterification between α-tocopherol (the most active form of VE) and succinic anhydride. Although in literature it is reported that lipase can catalyze this reaction, hereby we demonstrate that the reaction observed in DMSO and DMF is catalyzed by the histidyl residues of the protein. Histidine and its analogue containing an imidazole ring were tested as organocatalysts for the production of α-tocopherol succinate. In light of the imidazole organocatalysis, commercially-available 3-alkyl-1-methyl imidazolium ILs [CnC1Im][X−] were investigated as dual solvent-catalysts for the esterification of α-tocopherol with succinic anhydride, and provided satisfactory yields and reaction rates. [C5C1Im][NO3−] can be recycled by water extraction, instead of organic solvent extraction to separate α-tocopherol succinate from [C5C1Im][NO3−], with an average yield of 94.1% for 4 subsequent batches, while the catalytic activity of the recycled ILs showed almost no loss after 4 batches. The developed protocol for the synthesis of α-tocopherol esters and IL recycling bears industrial potential due to the ease of use and the efficient recycling.

 Please wait while we load your content...
Please wait while we load your content...