Deactivation mechanisms of tin-zeolites in biomass conversions†
Abstract
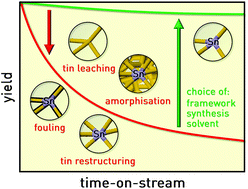
Metal-containing zeolites comprise outstandingly active and selective catalysts for a multitude of Lewis-acid and redox catalysed reactions that valorise renewable substrates into chemicals. Herein, focussing on tin-zeolites applied to the isomerisation of dihydroxyacetone (DHA) and xylose, we systematically study the influence of the framework type (MFI, MOR, BEA and FAU), preparation method (hydrothermal synthesis or alkaline-assisted metallation), hydrophobicity and nature of the solvent (water or methanol) on their intrinsic activity and, especially, on their stability. BEA and FAU zeolites were more active than MFI and MOR stannosilicates in aqueous and, particularly, in methanol-based tests owing to the larger relative amount of tetrahedral tin sites and/or superior mass transfer properties. Hydrothermally-prepared zeolites generally exhibited higher turnover frequencies than those obtained by a post-synthetic approach in view of their higher tin quality and less hydrophilic character. Remarkably, continuous-flow tests in a fixed-bed reactor indicated that exposure to reaction conditions for 24 hours could provoke dramatic changes in performance. Mainly due to amorphisation and modification of the tin structure (from tetra- to hexacoordination), the activity and selectivity of MOR, BEA and FAU zeolites were substantially, if not fully, depleted in the aqueous isomerisation of DHA, while MFI zeolites, especially the post-synthetically stannated sample, better preserved their initial performance. These deactivation phenomena were alleviated through the use of methanol, an industrially more amenable solvent, but, due to the greater retention of activity, fouling was more pronounced, particularly for MFI zeolites. In contrast, more extensive metal leaching was detected owing to the higher solubility of tin in the alcohol rather than water, where it hydrolyses into insoluble hydroxide species. In xylose isomerisation, hydrothermally-prepared MFI and BEA stannosilicates displayed reasonably stable operation. Deactivation mechanisms partly resembled those of the aqueous DHA isomerisation, but the chelating properties of the substrate played a greater detrimental role in tin loss.
- This article is part of the themed collection: 3rd International Symposium on Green Chemistry

 Please wait while we load your content...
Please wait while we load your content...