Bio-proton coupled semiconductor/metal-complex hybrid photoelectrocatalytic interface for efficient CO2 reduction†
Abstract
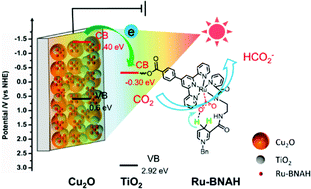
Aimed at high-efficiency biomimetic CO2 photoelectrochemical conversion, a bio-proton coupling metal-complex/semiconductor hybrid photoelectrocatalytic interface (Ru-BNAH/TiO2/Cu2O) was constructed by covalently modifying an in situ proton-transfer functionized molecular catalyst (Ru-BNAH) on the surface of a TiO2/Cu2O composite semiconductor substrate electrode. Due to the excellent proton coupling of the bio-proton carrier, the light current density in a CO2 atmosphere of the prepared Ru-BNAH/TiO2/Cu2O photoelectrocatalytic interface was twice as high as that without a proton carrier under the same conditions. Simultaneously, based on the excellent photosensitivity of the metal oxide substrate, the photogenerated electrons could rapidly transfer to the molecular catalyst for efficient CO2 reduction in a water medium. After 8 h irradiation at −0.9 V potential, the Ru-BNAH/TiO2/Cu2O photoelectrocatalytic interface produced 409.5 μmol formic acid, which was 2.44 times more than that without a proton transfer carrier. In addition, the in situ UV-visible absorption spectra and in situ Raman spectra indicated that the proton transport carrier supplied protons during CO2 reduction. Moreover, the generation of HCOO− in CO2-saturated D2O medium confirmed the proton (H) originated from the proton transfer carrier rather than the solvent (D2O).


 Please wait while we load your content...
Please wait while we load your content...