Efficient synthesis of α-alkyl-β-amino amides by transaminase-mediated dynamic kinetic resolutions†
Abstract
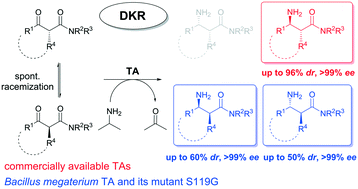
The biocatalytic stereocontrolled synthesis of various acyclic anti- or syn-α-alkyl-β-amino amides through a dynamic kinetic resolution strategy is demonstrated. A series of commercially available and in-house transaminases (TAs) were employed to perform the transamination of a series of chemically synthesized racemic α-alkyl-β-keto amides. Among them, commercial (R)-selective TAs showed the best activities and selectivities, giving access preferentially to the anti-diastereoisomers with low to high diastereomeric ratios (up to 96%) and excellent enantiomeric excess (>99%). The stereoselective biotransamination experiments were successfully demonstrated at a semi-preparative scale (25 mM, 100 mg substrate), leading to the corresponding optically active α-alkyl-β-amino amides in 45–90% isolated yields after a simple liquid–liquid extraction protocol.


 Please wait while we load your content...
Please wait while we load your content...