Determination of newly synthesized lipoic acid–fasudil dimer in rat plasma by LC-MS/MS and its application to pharmacokinetics study
Abstract
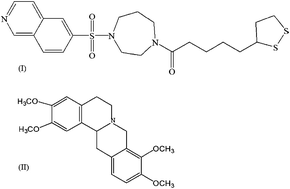
The aim of the present work was to develop and validate an LC-MS/MS method with electrospray ionization (ESI) for the quantification of lipoic acid–fasudil dimer (L–F 001), a novel multi-functional neuroprotective drug in rat plasma, using tetrahydropalmatine as an internal standard. The separation was carried out on a C18 column using a mobile phase consisting of acetonitrile and water (containing 5 mmol l−1 ammonium acetate adjusted to pH 5.0 with formic acid). The detection was performed by multiple reactions monitoring (MRM) mode via ESI source operating in the positive ionization mode. An linear calibration curve was obtained over the concentration range of 1.0–1000.0 ng ml−1 for lipoic acid–fasudil dimer (L–F 001). Accuracy, precision and extraction recovery of the analyte was satisfactory. The validated method was successfully applied to monitor the pharmacokinetic profile of the lipoic acid–fasudil dimer (L–F 001) after oral administration to rats.

 Please wait while we load your content...
Please wait while we load your content...