The effect of an ionic liquid on the rate of reaction at a phosphorus centre†
Abstract
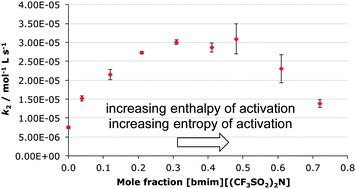
The effect of the ionic liquid 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide on the ethanolysis of diethyl chlorophosphate was investigated. The addition of the salt increases the rate constant, though the extent of this increase was dependent on the mole fraction used and the organic co-solvent. Temperature-dependent kinetic analyses were used to demonstrate that the rate enhancement was as a result of a decrease in the activation enthalpy offsetting a decrease in the activation entropy. Altering the salt used showed the importance of the balance of these enthalpic and entropic effects.

 Please wait while we load your content...
Please wait while we load your content...