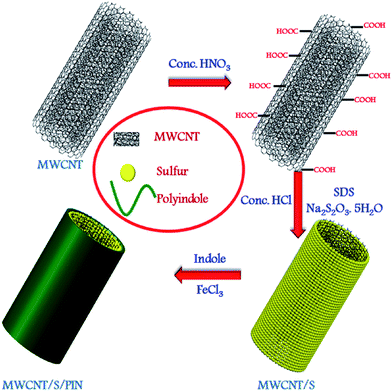
Enhanced cyclability using a polyindole modified cathode material for lithium sulphur batteries†
Reshma
Chulliyote
a,
Haritha
Hareendrakrishnakumar
a,
Murugan
Raja
b,
Joseph Mary
Gladis
 *a and
Arul Manuel
Stephan
b
*a and
Arul Manuel
Stephan
b
aDepartment of Chemistry, Indian Institute of Space Science and Technology, Thiruvananthapuram-695547, India. E-mail: marygladis@iist.ac.in; Tel: +91 4712568533
bElectrochemical Power Systems Division, CSIR-Central Electrochemical Research Institute, Karaikudi 630006, India
First published on 7th August 2017
Abstract
Here we report a novel multiwall carbon nanotube/sulphur/polyindole (MWCNT/S/PIN) nanocomposite as a cathode material for lithium–sulphur (Li–S) batteries to alleviate capacity decay. The nanocomposite cathode material is synthesized by chemical precipitation of sulphur onto functionalized MWCNTs followed by in situ polymerization of indole. The MWCNT/S/PIN nanocomposite exhibits an initial specific capacity of 1490 mA h g−1 and enhanced cycling stability compared to the binary nanocomposite (MWCNT/S). The MWCNT/S/PIN composite cathode displays a specific capacity of 1043 mA h g−1 after 100 cycles at 0.1C rate with 70% capacity retention. The better electrochemical performance of the ternary nanocomposite cathode material is attributed to the synergistic effect of the functionalized MWCNTs and polyindole which provides an improved conductivity and effective fencing of intermediate polysulphides.
Introduction
Rechargeable lithium ion batteries (LIBs) have become an important part of our day to day life in terms of their inevitable usage in smart phones, smart grids, electric vehicles, etc. LIBs use traditional cathode materials based on lithiated transition metal oxides, vanadates and phosphates with a relatively low energy density (∼200 W h kg−1), which is insufficient to satisfy the rapidly growing demand for energy.1–3 In addition, most LIBs are too expensive for large scale applications. Hence, great attention has been paid by researchers to the development of novel rechargeable systems with high energy density and low cost. Among the various cathode materials investigated for rechargeable lithium batteries,4 sulphur received significant attention due to the following reasons: (i) highest theoretical capacity (1675 mA h g−1), (ii) high energy density (2500 W h g−1),5–8 (iii) feeble toxicity, (iv) low cost, (v) natural abundance, (vi) environmental benignity, etc. However, Li–S batteries are associated with a number of issues which should be rectified to take a leap in energy storage technology.9 The challenges include overcoming the insulating nature of sulphur and its byproducts, volumetric changes in the electrodes, solubility of intermediate polysulphides (Li2Sx, 4 ≤ x ≤ 8), low utilization of active material due to polysulphide shuttling, etc., which in turn leads to low sulphur utilization, low coulombic efficiency and rapid capacity decay during repeated charging and discharging processes.10–14 In order to tackle these issues, various approaches have been adopted,2,3 which include combining sulphur with electrically conductive species such as metal oxides,16 carbon nanotubes,12 graphene,13 carbon nanofibers10,14 carbon onions,11 porous carbons,17–19 conductive polymers in core and yolk shell structures, etc.20–24 High conductivity of the cathode material is advantageous to improve the kinetics of Li–S redox reactions.25 Recently, attempts have been made to develop ternary cathode materials, where sulphur is trapped between two conductive elements, which improves the capacity and cycling performance. These include sulphur wrapped with carbon nanotubes and graphene,26,27 graphene and porous carbon,28,29 and carbon nanotubes and conductive polymers such as polyaniline,25,30 polypyrrole,5,31etc. The improved electrochemical performances of ternary composite cathodes are due to the enhanced conductivity and the ceiling effect of polysulfide in the cathodic region. On the other hand, achieving high sulphur loading and good performance simultaneously with a uniform polymer coating on sulphur is one more challenge to be addressed. Here, in this study, we present for the first time, polyindole (PIN) as one of the conductive layers in a ternary composite along with multiwall carbon nanotubes (MWCNTs) aiming to achieve a uniform coating and better sulphur loading. Polyindole is one of the well-known conductive electroactive polymers with an electrical conductivity of 0.1 S cm−1 and is stable under normal conditions, and therefore exhibits good electrochemical characteristics as a cathode material.32 Hence, a coating of polyindole on sulphur is beneficial to trap polysulphides. Owing to their good electrical conductivity, and thermal and mechanical properties,5 MWCNTs also play an advantageous role in the ternary system. Here, we report a MWCNT/S/PIN nanocomposite with a dual core shell structure as a ternary cathode material for Li–S batteries. The composite preparation involves simple chemical precipitation of sulphur onto MWCNTs followed by in situ polymerization of the monomer indole. An initial discharge capacity as high as 1490 m Ah g−1 at a rate of 0.1C, with a good cycling stability up to 100 cycles and a very feeble capacity fading rate (0.08%), is achieved. This demonstrates the enhanced electrochemical performance of the ternary composite due to the PIN coating.Experimental
Composite synthesis
The MWCNT/S/PIN composite preparation is schematically represented in Fig. 1. Polyindole was synthesized by chemical oxidative polymerization33 of the indole monomer (Aldrich, 99% purity) using FeCl3 (Merck, 96%) as the oxidant. Oxidation of MWCNTs (Sigma Aldrich, 95% purity) was carried out using nitric acid (HNO3).34 The MWCNT/S composite was prepared by a chemical precipitation method. The oxidised MWCNTs were dispersed in a sodium dodecyl sulphate (SDS, Merck, 99%) aqueous solution in an ultrasonic bath. Sodium thiosulphate (Na2S2O3·5H2O) (Merck, 99.5%) was added and stirred for 10 minutes followed by dropwise addition of hydrochloric acid (HCl, Merck, 35%) and stirring for 1 hour. The product was separated and dried in a vacuum oven at 60 °C. To this material, 0.53 g of indole (Aldrich, 99%) in 10 ml of ethanol was added, stirred for 10 minutes, and then FeCl3 was added to the reaction mixture dropwise under a N2 atmosphere, and stirred for 24 hours at 0 °C. The pristine polymer was prepared by using indole and FeCl3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). Finally the product was dried in a vacuum at 70 °C for 24 hours. The preparation of the MWCNT/S/PIN composite and its preliminary characterization was reported in our previous work.35
5). Finally the product was dried in a vacuum at 70 °C for 24 hours. The preparation of the MWCNT/S/PIN composite and its preliminary characterization was reported in our previous work.35
Material characterization
The chemical transformations in the composite were characterized by using Fourier transform infrared spectroscopy (FTIR, Perkin Elmer) with the KBr pellet method. X-ray diffraction analysis (XRD, D8 Discover, Bruker) was carried out with Cu Kα radiation to confirm the components of the composite. The surface morphology and microstructure of the composite were studied by field emission scanning electron microscopy (HRSEM, Quanta FEG200, FEI, USA) coupled with elemental mapping and high resolution transmission electron microscopy (HRTEM, JEM-2100, JEOL, Japan). The thermal stability of the composite was determined by thermogravimetric analysis (TGA, Q-50, TA Instruments, USA) in the temperature range of 0–400 °C at a heating rate of 10 °C min−1 under a N2 atmosphere. The presence of polyindole in the composite was determined by using a Kratos-Analytical Axis Ultra X-ray photoelectron spectrophotometer (XPS) with a monochromatic Al-Kα-X-ray source.Electrochemical measurements
The electrochemical performance of the MWCNT/S/PIN composite was investigated using coin-type cells (CR2032) assembled in an argon filled Braun glovebox with lithium metal as the anode and a polypropylene membrane (Celgard 2042) as the separator. The cathode was prepared by mixing the 70 wt% MWCNT/S/PIN composite, 20 wt% acetylene black (MTI, 99.5% purity) as a conductive agent and 10 wt% polyvinylidene fluoride (PVDF) (Kynar, HSV900) as a binder in 1-methyl-2-pyrrolidone (NMP, Sigma-Aldrich, ≥ 99.5% purity). The resultant slurry was coated onto aluminium foil and dried in a vacuum for 12 h at 60 °C. The electrode was punched into circular disks with a diameter of 12 mm with an active material loading of about 2 mg cm−2. 0.7 M lithium bistrifluoromethanesulfonamide (Aldrich, 96% purity) in a solution of 1,3-dioxalane (Spectrochem) and dimethoxy ethane (Aldrich, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) was used as the electrolyte and 0.4 M LiNO3 (Aldrich) as an additive. The cells were galvanostatically charged and discharged between 1.6 and 3 V versus Li+/Li on a Land CT2001A battery tester. The specific capacities and current rates were calculated according to the mass of sulphur in the cathode. Cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) were performed by using a PGSTAT302N electrochemical workstation (Autolab, Metrohm, Netherlands). CV was carried out at a scan rate of 0.1 mV s−1 ranging from 1.6 to 3 V versus Li+/Li. EIS was performed at OCV, with a perturbation potential of 10 mV in a frequency range of 10 m Hz to 100 kHz. The sulphur cathodes were separated after 100 cycles and rinsed several times with pure DOL to remove the electrolyte residue. XPS analysis of the cathodes before and after 100 cycles was carried out.
1, v/v) was used as the electrolyte and 0.4 M LiNO3 (Aldrich) as an additive. The cells were galvanostatically charged and discharged between 1.6 and 3 V versus Li+/Li on a Land CT2001A battery tester. The specific capacities and current rates were calculated according to the mass of sulphur in the cathode. Cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) were performed by using a PGSTAT302N electrochemical workstation (Autolab, Metrohm, Netherlands). CV was carried out at a scan rate of 0.1 mV s−1 ranging from 1.6 to 3 V versus Li+/Li. EIS was performed at OCV, with a perturbation potential of 10 mV in a frequency range of 10 m Hz to 100 kHz. The sulphur cathodes were separated after 100 cycles and rinsed several times with pure DOL to remove the electrolyte residue. XPS analysis of the cathodes before and after 100 cycles was carried out.
Results and discussion
FTIR spectroscopy is used to determine the presence of polyindole in the composite. The characteristic peaks in the IR spectra of polyindole (Fig. 2a) are in good agreement with the literature reports.33,36,37 The strong and broad peak at 3437 cm−1 is the characteristic absorption of the N–H bond. The band at 1368 cm−1 is related to modes involving the C8–N–C2–C3 group. The band at 741 cm−1 indicates that the benzene ring is not affected during the polymerization process of indole. The single peak located at 1450 cm−1 is assigned to the stretching of the benzene ring. The band at 1621 cm−1 is ascribed to the C–C vibration of the indole ring. The above-mentioned characteristic peaks of polyindole are also observed in the MWCNT/S/PIN which confirms the formation of polyindole on the surface of the MWCNT/S composite. | ||
| Fig. 2 (a) FTIR spectrum of the PIN and MWCNT/S/PIN, (b) TGA of PIN, MWCNT-COOH, MWCNT/S/PIN, MWCNT/S and sulphur and (c) XRD of sulphur, MWCNT/S/PIN and PIN. | ||
Carboxyl functionalized MWCNTs are characterized using Raman spectroscopy, IR spectroscopy and TGA, to confirm the functionalization of the MWCNTs (Fig. S1–S3†). Fig. 2b shows the TGA of polyindole, functionalized MWCNTs, sulphur, MWCNT/S and MWCNT/S/PIN under a nitrogen atmosphere. The composites display a weight loss from 118 °C to 240 °C due to the evaporation of sulphur from the composites. Polyindole undergoes a continuous weight loss up to 400 °C due to its gradual decomposition. The sulphur content is calculated after eliminating the weight loss due to polyindole and functionalized MWCNTs. The sulphur content of MWCNT/S and MWCNT/S/PIN is about 83.4 wt% and 79.06 wt%, respectively. Fig. 2c shows the XRD patterns of sulphur, and the as-prepared polyindole and MWCNT/S/PIN. The broad peak at 2θ = 20° exhibits the amorphous nature of the polymer. The peaks at 2θ values of 23.08°, 25.73°, 27.66°, 28.75°, 31.36°, 34.10° and 36.98° are in good agreement with the (222), (026), (117), (135), (044), (137) and (317) reflections of the Fddd orthorhombic phase (JCPSD no.: 00-001-0478) of sulphur. The sharp and intense diffraction signals indicate the crystal lines of sulphur. MWCNT/S/PIN exhibits an XRD pattern similar to those of sulphur and MWCNT/S (Fig. S4†), but with low intensity, evidencing that sulphur is coated with PIN. Fig. 3 and S5† depict the surface morphology of the MWCNT/S/PIN and MWCNT/S composites and their corresponding elemental mapping (c–f for MWCNT/S/PIN and S5(b–d)† for MWCNT/S). It is observed that the MWCNT/S and MWCNT/S/PIN composites have a tubular structure with no agglomerated sulphur particles. It is difficult to differentiate between sulphur and polymer coatings from the micrograph. The elemental distribution maps assert that polyindole is coated onto the MWCNT/S composite. It is further confirmed by the TEM images of the MWCNT/S and MWCNT/S/PIN composites as shown in Fig. 4. It is observed that the sulphur is coated over the surface of the MWCNTs and an increase in the tube diameter affirms the information obtained from SEM. A thin layer of polyindole is wrapped over the surface of MWCNT/S attributed to the coating of polyindole onto MWCNT/S, which further confirms the dual core shell structure. The thickness of the ternary composite is increased after polymerization. Moreover, the TEM image displays (Fig. 4d) a thickness increase of ∼2 nm in the ternary composite after the polyindole coating is observed. The EDS analysis shows the presence of sulphur and nitrogen in the composite (Fig. 4f).
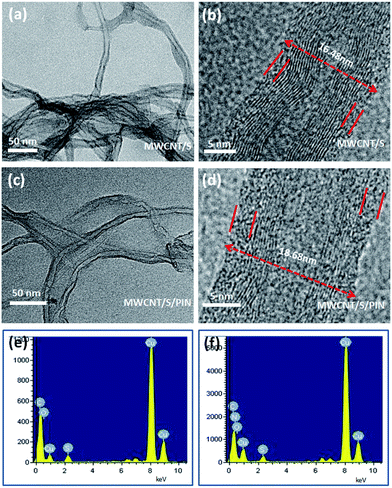 | ||
| Fig. 4 TEM images of (a, b) MWCNT/S and (c, d) MWCNT/S/PIN, and EDS analysis of (e) MWCNT/S and (f) MWCNT/S/PIN. | ||
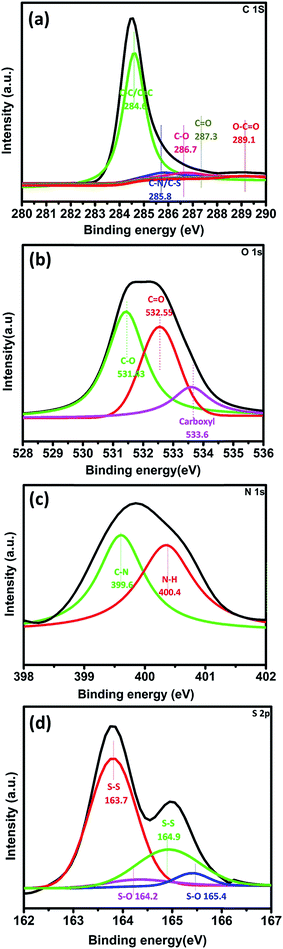
A wide scan energy spectrum of the MWCNT/S/PIN indicates the presence of C, O, N and S which agrees well with the elemental mapping. The survey scan (Fig. S6†) reveals the presence of C 1s, O 1s, N 1s, S 2s and S 2p in the composite. The peaks at 164 eV, 240 eV, 285.1 eV, 400 eV and 533.1 eV correspond to the S 2p, S 2s, C 1s, N 1s and O 1s, respectively. The high resolution C 1s (Fig. 5a) peak is divided into 5 components: C–C bond (284.6 eV) of sp2, C–N, C–S bond (285.8 eV), C–O bond (286.7 eV) and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (289.1 eV).38,39 The high resolution O 1s spectrum (Fig. 5b) is subdivided into three components: C–O bond (531.43 eV), C
O (289.1 eV).38,39 The high resolution O 1s spectrum (Fig. 5b) is subdivided into three components: C–O bond (531.43 eV), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond (532.55 eV) and carboxyl group (533.6 eV).39,40 The N 1s spectrum (Fig. 5c) contains two contributions at 399.6 eV and 401.68 eV (ref. 41) which are assigned to the N–C and N–H bonds. These results are in good agreement with the FTIR spectra. The presence of pyrrolic nitrogen may influence the electrochemical properties. The basic pyrrolic nitrogen (N–H) in the composite enhances the adsorption of polysulphides through its interaction with nitrogen atoms. Moreover, these five membered pi systems with lone pairs enhance the surface adsorption sites for polysulphides.42,43,47 The high resolution spectra of S 2p (Fig. 5b) are subdivided into four peaks. S 2p3/2 (163.7 and 164.9 eV) and S 2p1/2 (164.2 and 165.4 eV) with an energy separation of 1.2 eV correspond to the S–S and S–O bonds in the composite.43,44
O bond (532.55 eV) and carboxyl group (533.6 eV).39,40 The N 1s spectrum (Fig. 5c) contains two contributions at 399.6 eV and 401.68 eV (ref. 41) which are assigned to the N–C and N–H bonds. These results are in good agreement with the FTIR spectra. The presence of pyrrolic nitrogen may influence the electrochemical properties. The basic pyrrolic nitrogen (N–H) in the composite enhances the adsorption of polysulphides through its interaction with nitrogen atoms. Moreover, these five membered pi systems with lone pairs enhance the surface adsorption sites for polysulphides.42,43,47 The high resolution spectra of S 2p (Fig. 5b) are subdivided into four peaks. S 2p3/2 (163.7 and 164.9 eV) and S 2p1/2 (164.2 and 165.4 eV) with an energy separation of 1.2 eV correspond to the S–S and S–O bonds in the composite.43,44
This binding energy shift ascribed to the changes in the electronic distribution on the sulphur atom evidenced the binding between the sulphur and functionalized MWCNTs via S–O bonds, which helps to prevent active material loss by trapping the polysulphides during the charge–discharge process.45,46
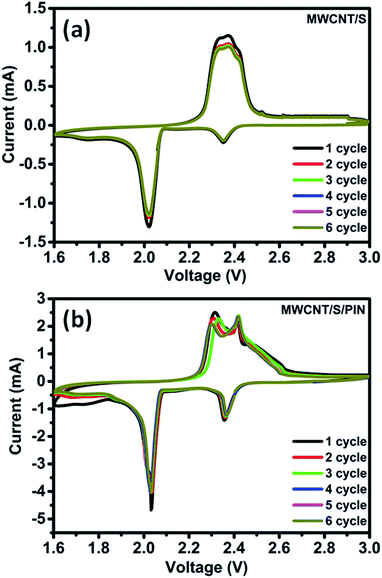
The cyclic voltammetry studies of the MWCNT/S and MWCNT/S/PIN composite electrodes were carried out at a scan rate of 0.1 mV s−1 to understand the electrochemical reaction of the nanocomposite cathode. Two reduction and one oxidation peaks are observed (Fig. 6a and b) in both the composites due to multiple reactions between sulphur and lithium, as reported previously.6 The cathodic peak at 2.37 V is due to the conversion of octaatomic sulphur to higher order lithium polysulphides (Li2Sn, 4 ≤ n ≤ 8); further reduction at 2.0 V is due to the conversion of these higher order polysulphides to lower order polysulphides (Li2Sn, 3 ≤ n ≤ 4) and finally to insoluble Li2S2 and Li2S.6–11
The subsequent oxidation process provides a broad oxidation peak at 2.42 V mainly due to the conversion of Li2S2 and Li2S to higher order polysulphides and finally to sulphur.15–17 The increase in the peak intensity of the MWCNT/S/PIN composite is attributed to good electrochemical contact which in turn boosts the utilisation of active material.6–11 The increment in the peak area compared to the binary composite proves the increase in active material utilisation.5,25,47 Also, the CV curves are more stable in the subsequent cycles indicating better cyclability.
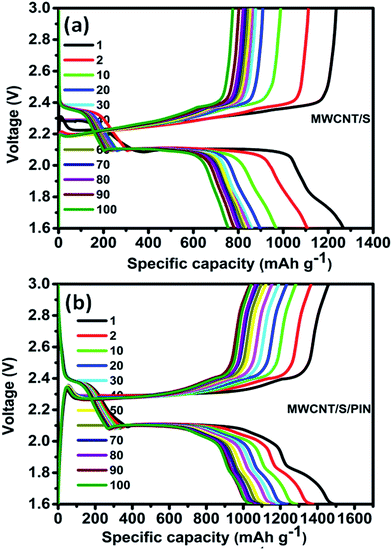
The result obtained from the CV is in good agreement with the galvanostatic charge–discharge curves. Fig. 7a and b represent the typical charge–discharge behaviour of the MWCNT/S and MWCNT/S/PIN composite electrodes in rechargeable Li–S batteries at room temperature. The figures show the charge–discharge capacities of the 1, 2, 10, 20, 30, 40, 50, 60, 70, 80, 90 and 100th cycles of ternary and binary composites. The performance of the MWCNT/S/PIN composite is enhanced due to the polyindole coating compared to that of MWCNT/S. The ternary composite delivered an initial capacity of 1490 mA h g−1 with an average capacity degradation of 0.08% per cycle and 70% capacity retention with respect to the initial discharge capacity, and 97% with respect to the 50th discharge capacity up to 100 cycles. But in the case of the binary composite, an initial discharge capacity of 1267 mA h g−1 with 61% capacity retention with respect to the initial discharge capacity is observed. The polyindole coating onto the composite prevents the loss of active material by blocking the polysulphides. The oxygen atoms in the functionalised MWCNTs and the nitrogen atoms in the polyindole are helpful to prevent the loss of sulphur as polysulphides during the cycling and continuous utilization of sulphur in the composite.45,47
The coulombic efficiency of the MWCNT/S/PIN cathode (∼100%) is higher than that of MWCNT/S (∼97%), due to the reduction in the shuttling behaviour of polysulphides (Fig. 8a).
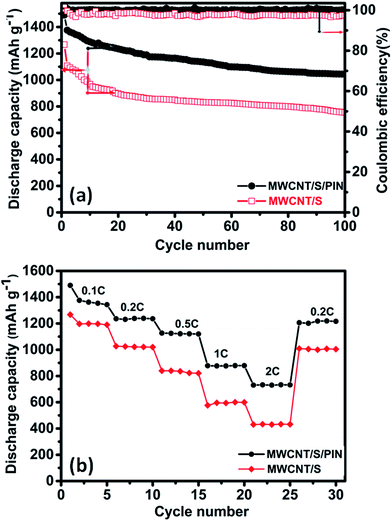 | ||
| Fig. 8 (a) Cycle performance and coulombic efficiency of MWCNT/S and MWCNT/S/PIN at 0.1C rate and (b) rate capability studies of MWCNT/S and MWCNT/S/PIN. | ||
The electrochemical performances of the binary and ternary composites at different current densities were investigated in detail, which are shown in Fig. 8b. The ternary composite exhibits excellent performance at different C rates when compared with the binary composite. The initial discharge capacity is 1490 mA h g−1 at 0.1C rate. It exhibits 1236, 1124, 878 and 730 mA h g−1 discharge capacities at the current densities of 0.2C, 0.5C, 1C and 2C respectively. When returned to 0.2C rate its discharge capacity was retained at 1218 mA h g−1 with 98.5% capacity retention which indicates robustness and stability of the ternary composite.
The binary composite shows the discharge capacities of 1232, 1024, 840, 590 and 430 mA h g−1 at 0.1C, 0.2C, 0.5C, 1C and 2C, respectively, and when switched back to 0.2C rate the discharge capacity becomes 996 mA h g−1. The high solubility of lithium polysulphides in the electrolytes, insulating nature of sulphur and lowest end discharge products are negatively affecting the performance of the composites when we switch to higher current rates. Here in the case of the ternary composite both MWCNTs and polyindole provide an effective conductive network to move electrons, thereby enhancing the rate performance. The superior performances of the ternary composite are due to the polyindole coating and MWCNTs that confine the polysulphides from dissolving in the electrolyte. Here, sulphur is sandwiched between polyindole and MWCNTs. Such a protective thin layer coating is absent in the case of the binary composite, and sulphur is deposited over the surface of MWCNTs. Consequently, there exist more chances to lose sulphur as polysulphides due to dissolution in the electrolyte, thereby reducing its performance. The XPS spectra of the MWCNT/S/PIN cathode before and after 100 cycles were recorded to support this rationalization as shown in Fig. 9. The presence of aluminium and fluorine in the survey scan spectrum (Fig. 9a) is due to the Al collector and PVDF binder in the cathode. The high resolution spectra of Li 1s are subdivided into two components (Fig. 9b). The peak at 54.9 eV is ascribed to the electrons in the lithium sulphide. The interaction between lithium and nitrogen in the polyindole is proved by the peak at 56.1 eV which corresponds to the Li–N bond.47,48 These interactions favour polyindole to restrain the discharge products. Furthermore, the conductive nature of polyindole helps to enhance the utilization of active material.50,51
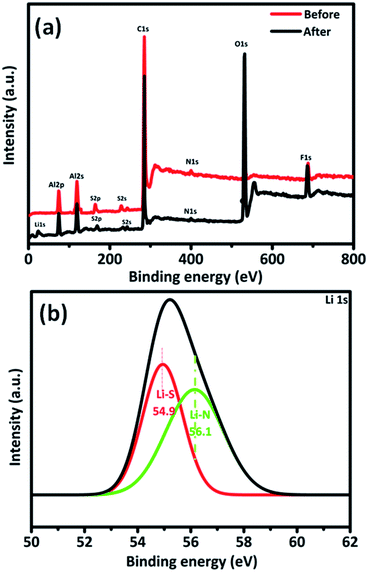 | ||
| Fig. 9 (a) Survey scan of the cathode before and after cycling and (b) high resolution Li 1s spectra of the cathode after 100 cycles. | ||
The electrochemical performance of the ternary composite is significantly greater than those of the control material – MWCNT/S, and other ternary composites reported elsewhere.25–31 Wu et al. reported a coating of polyaniline over the binary composite MWCNT/S (932.4 mA h g−1 after 80 cycles, exhibiting 56.3% capacity retention w.r.t the initial discharge capacity at 0.1C rate).25 Similarly, Li et al. studied sulphur supported with MWCNTs and coated with polyaniline (∼600 mA h g−1 after 100 cycles with 61.8% capacity retention at 0.2C rate).30 A dual core shell structured cathode material using MWCNTs and polypyrrole exhibited a capacity of 917 mA h g−1 after 60 cycles and 60% capacity retention at a current density of 200 mA g−1 was described by Wang et al.5 Another type of material based on MWCNTs and polypyrrole reported by Zhang et al. displayed a reversible capacity of 751 mA h g−1 after 100 cycles, with 60% capacity retention at 0.2C rate.31
To investigate the superior performance of MWCNT/S/PIN over MWCNT/S, EIS measurements were carried out at OCV. Fig. 10 shows the Nyquist plots of the ternary composite (inset) and binary composite before and after 100 cycles, which consist of a depressed semicircle in the high frequency region and a straight line in the low frequency region. The largest diameter of an impedance semicircle in the high frequency region is associated with higher charge transfer resistance and the straight line in the low frequency region corresponds to ion diffusion resistance within the cathode.16–23 The resistance of the electrolyte (Re) determined from the intersection of the front end of the semicircle on the Z′ axis is zero in both the cases. The diameter of the impedance semicircle is related to the charge transfer resistance which is the measure of the difficulty in the transfer of electrons/charge at the electrode and electrolyte interface. The ternary composite has a much lower charge transfer resistance than the binary composite, which is expected due to its higher initial discharge capacity than the binary composite before and after cycling. In the case of the ternary composite, sulphur is sandwiched in between conductive MWCNTs and polyindole, facilitating an easy pathway to electron transfer. After 100 cycles the charge transfer resistance of the binary composite is increased due to the dissolution of polysulphides in the electrolyte and the irreversible deposition of discharge products (Li2S/Li2S2) leading to the loss of active materials.13,49 But the charge transfer resistance of the ternary composite cathode is almost the same after 100 cycles, indicating that the conductive architecture surrounded by sulphur maintains its integrity during the charge–discharge process.50,51 Moreover, the porous structure of conductive polyindole helps easy transport of Li+ ions. Hence, the ion diffusion resistance is reduced for MWCNT/S/PIN compared to MWCNT/S. It is further supported by the identical morphology of the ternary composite cathode even after 100 cycles (Fig. S7†). The changes on the surface of the binary composite cathode are attributed to the impact of electrochemical reactions resulting in the increased charge transfer resistance.5,49 The reason for the better rate capability and cyclability of the ternary composite is attributed to the low charge transfer and diffusion resistances. Also, there is sufficient interaction among MWCNTs and polyindole with sulphur which enhances the electronic conduction.
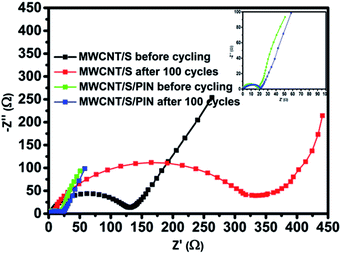 | ||
| Fig. 10 Electrochemical impedance spectra of MWCNT/S and MWCNT/S/PIN before and after 100 cycles at 0.1C rate. | ||
Conclusion
In summary, a novel ternary composite MWCNT/S/PIN was prepared by in situ oxidative polymerisation of indole onto MWCNT/S. It is a promising cathode material for Li–S batteries. The electrochemical performance has been improved, especially specific capacity, cyclability and rate capability, by wrapping polyindole on sulphur. This may be due to reduced charge transfer resistance of the material, which facilitates easy transport of lithium ions into the cathode material, and reduced shuttling effects. The MWCNT/S/PIN composite attains an initial capacity of 1490 mA h g−1 and a reversible capacity of 1043 mA h g−1 after 100 cycles at 0.1C with 100% coulombic efficiency. The good electrochemical performance is due to the functionalized MWCNT/S/PIN dual core shell type structure, with both MWCNTs and polyindole supporting the trapping of the polysulphides and the enhancement of the capacity retention during cycling.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors C. R., H. H. and J. M. G. gratefully acknowledge the financial support by the Indian Institute of Space Science and Technology (IIST), Thiruvananthapuram. India.References
- P. G. Bruce, S. A. Freunberger, L. J. Hardwick and J.-M. Tarascon, Nat. Mater., 2012, 11, 19–29 CrossRef CAS PubMed.
- L. Carbone, S. G. Greenbaum and J. Hassoun, Sustainable Energy Fuels, 2017, 1, 228–247 Search PubMed.
- A. Manthiram, Y. Fu and Y.-S. Su, Acc. Chem. Res., 2013, 46, 1125–1134 CrossRef CAS PubMed.
- D. Bresser, S. Passerini and B. Scrosati, Chem. Commun., 2013, 49, 10545–10562 RSC.
- C. Wang, W. Wan, J.-T. Chen, H.-H. Zhou, X.-X. Zhang, L.-X. Yuan and Y.-H. Huang, J. Mater. Chem. A, 2013, 1, 1716–1723 CAS.
- Y. Zhang, Y. Zhao and Z. Bakenov, Nanoscale Res. Lett., 2014, 9, 137 CrossRef PubMed.
- J. Jiang, J. Zhu, W. Ai, X. Wang, Y. Wang, C. Zou, W. Huang and T. Yu, Nat. Commun., 2015, 6, 8622 CrossRef CAS PubMed.
- Y.-X. Wang, S.-L. Chou, H.-K. Liu and S.-X. Dou, J. Power Sources, 2013, 244, 240–245 CrossRef CAS.
- N. Li, M. Zheng, H. Lu, Z. Hu, C. Shen, X. Chang, G. Ji, J. Cao and Y. Shi, Chem. Commun., 2012, 48, 4106–4108 RSC.
- K. Fu, Y. Li, M. Dirican, C. Chen, Y. Lu, J. Zhu, Y. Li, L. Cao, P. D. Bradford and X. Zhang, Chem. Commun., 2014, 50, 10277–10280 RSC.
- S. Choudhury, M. Zeiger, P. Massuti-Ballester, S. Fleischmann, P. Formanek, L. Borchardt and V. Presser, Sustainable Energy Fuels, 2017, 1, 84–94 Search PubMed.
- K. Xi, B. Chen, H. Li, R. Xie, C. Gao, C. Zhang, R. V. Kumar and J. Robertson, Nano Energy, 2015, 12, 538–546 CrossRef CAS.
- J. Feng, X. Qin, Z. Ma, J. Yang, W. Yang and G. Shao, Electrochim. Acta, 2016, 190, 426–433 CrossRef CAS.
- R. Singhal, S.-H. Chung, A. Manthiram and V. Kalra, J. Mater. Chem. A, 2015, 3, 4530–4538 CAS.
- J. Lee, T. Hwang, Y. Lee, J. K. Lee and W. Choi, Mater. Lett., 2015, 158, 132–135 CrossRef CAS.
- J. Pan, C. Wu, J. Cheng, Y. Pan, Z. Ma, S. Xie and J. Li, J. Power Sources, 2015, 293, 527–532 CrossRef CAS.
- S. Zhang, M. Zheng, Z. Lin, N. Li, Y. Liu, B. Zhao, H. Pang, J. Cao, P. He and Y. Shi, J. Mater. Chem. A, 2014, 2, 15889–15896 CAS.
- G.-L. Xu, Y.-F. Xu, J.-C. Fang, X.-X. Peng, F. Fu, L. Huang, J.-T. Li and S.-G. Sun, ACS Appl. Mater. Interfaces, 2013, 5, 10782–10793 CAS.
- D. Li, F. Han, S. Wang, F. Cheng, Q. Sun and W.-C. Li, ACS Appl. Mater. Interfaces, 2013, 5, 2208–2213 CAS.
- F. Wu, J. Chen, R. Chen, S. Wu, L. Li, S. Chen and T. Zhao, J. Phys. Chem. C, 2011, 115, 6057–6063 CAS.
- H. Chen, W. Dong, J. Ge, C. Wang, X. Wu, W. Lu and L. Chen, Sci. Rep., 2013, 3, 1910 CrossRef PubMed.
- Y. Fu and A. Manthiram, RSC Adv., 2012, 2, 5927–5929 RSC.
- Y. Fu and A. Manthiram, J. Phys. Chem. C, 2012, 116, 8910–8915 CAS.
- W. Zhou, Y. Yu, H. Chen, F. J. DiSalvo and H. D. Abruña, J. Am. Chem. Soc., 2013, 135, 16736–16743 CrossRef CAS PubMed.
- F. Wu, J. Chen, L. Li, T. Zhao and R. Chen, J. Phys. Chem. C, 2011, 115, 24411–24417 CAS.
- L. Sun, W. Kong, Y. Jiang, H. Wu, K. Jiang, J. Wang and S. Fan, J. Mater. Chem. A, 2015, 3, 5305–5312 CAS.
- J. He, Y. Chen, P. Li, F. Fu, Z. Wang and W. Zhang, J. Mater. Chem. A, 2015, 3, 18605–18610 CAS.
- M. Yu, R. Li, Y. Tong, Y. Li, C. Li, J.-D. Hong and G. Shi, J. Mater. Chem. A, 2015, 3, 9609–9615 CAS.
- S. Liu, K. Xie, Y. Li, Z. Chen, X. Hong, L. Zhou, J. Yuan and C. Zheng, RSC Adv., 2015, 5, 5516–5522 RSC.
- X. Li, M. Rao, D. Chen, H. Lin, Y. Liu, Y. Liao, L. Xing and W. Li, Electrochim. Acta, 2015, 166, 93–99 CrossRef CAS.
- Y. Zhang, Y. Zhao, Z. Bakenov, M. Tuiyebayeva, A. Konarov and P. Chen, Electrochim. Acta, 2014, 143, 49–55 CrossRef CAS.
- C. Zhijiang, S. Xingjuan and F. Yanan, J. Power Sources, 2013, 227, 53–59 CrossRef.
- S. An, T. Abdiryim, Y. Ding and I. Nurulla, Mater. Lett., 2008, 62, 935–938 CrossRef CAS.
- I. D. Rosca, F. Watari, M. Uo and T. Akasaka, Carbon, 2005, 43, 3124–3131 CrossRef CAS.
- C. Reshma and J. Mary Gladis, Mater. Sci. Forum, 2015, 830–831, 604–607 CrossRef.
- C. Zhijiang and H. Chengwei, J. Power Sources, 2011, 196, 10731–10736 CrossRef.
- N.-W. Li, Y.-X. Yin and Y.-G. Guo, RSC Adv., 2016, 6, 617–622 RSC.
- S. Niu, W. Lv, C. Zhang, Y. Shi, J. Zhao, B. Li, Q.-H. Yang and F. Kang, J. Power Sources, 2015, 295, 182–189 CrossRef CAS.
- V. Datsyuk, M. Kalyva, K. Papagelis, J. Parthenios, D. Tasis, A. Siokou, I. Kallitsis and C. Galiotis, Carbon, 2008, 46, 833–840 CrossRef CAS.
- S. Roy, T. Das, Y. Ming, X. Chen, C. Y. Yue and X. Hu, J. Mater. Chem. A, 2014, 2, 3961–3970 CAS.
- S. P. Koiry, V. Saxena, D. Sutar, S. Bhattacharya, D. K. Aswal, S. K. Gupta and J. V. Yakhmi, J. Appl. Polym. Sci., 2007, 103, 595–599 CrossRef CAS.
- J. Yang, J. Xie, X. Zhou, Y. Zou, J. Tang, S. Wang, F. Chen and L. Wang, J. Phys. Chem. C, 2014, 118, 1800–1807 CAS.
- S. Zhu, Y. Wang, J. Jiang, X. Yan, D. Sun, Y. Jin, C. Nan, H. Munakata and K. Kanamura, ACS Appl. Mater. Interfaces, 2016, 8, 17253–17259 Search PubMed; G. Zhou, L.-C. Yin, D.-W. Wang, L. Li, S. Pei, I. R. Gentle, F. Li and H.-M. Cheng, ACS Nano, 2013, 7, 5367–5375 CrossRef CAS PubMed.
- L. Zhang, L. Ji, P.-A. Glans, Y. Zhang, J. Zhu and J. Guo, Phys. Chem. Chem. Phys., 2012, 14, 13670–13675 RSC.
- R. Demir-Cakan, M. Morcrette, F. Nouar, C. Davoisne, T. Devic, D. Gonbeau, R. Dominko, C. Serre, G. Férey and J.-M. Tarascon, J. Am. Chem. Soc., 2011, 133, 16154–16160 CrossRef CAS PubMed.
- W. G. Wang, X. Wang, L. Y. Tian, Y. L. Wang and S. H. Ye, J. Mater. Chem. A, 2014, 2, 4316–4323 CAS.
- X. Li and X. Sun, Front. Energy Res., 2014, 2, 1–9 RSC.
- Z. W. Seh, H. Wang, P.-C. Hsu, Q. Zhang, W. Li, G. Zheng, H. Yao and Y. Cui, Energy Environ. Sci., 2014, 7, 672–676 CAS.
- Y.-S. Su and A. Manthiram, Electrochim. Acta, 2012, 77, 272–278 CrossRef CAS.
- C. Wang, H. Chen, W. Dong, J. Ge, W. Lu, X. Wu, L. Guo and L. Chen, Chem. Commun., 2014, 50, 1202–1204 RSC.
- J. Zhang, Y. Cai, Q. Zhong, D. Lai and J. Yao, Nanoscale, 2015, 7, 17791–17797 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c7se00210f |
| This journal is © The Royal Society of Chemistry 2017 |