Students’ perceptions of a project-based Organic Chemistry laboratory environment: a phenomenographic approach
Nikita L.
Burrows
 ,
Montana K.
Nowak
and
Suazette R.
Mooring
,
Montana K.
Nowak
and
Suazette R.
Mooring
 *
*
Department of Chemistry, Georgia State University, Atlanta, Georgia, USA. E-mail: smooring@gsu.edu
First published on 5th July 2017
Abstract
Students can perceive the laboratory environment in a variety of ways that can affect what they take away from the laboratory course. This qualitative study characterizes undergraduate students’ perspectives of a project-based Organic Chemistry laboratory using the theoretical framework of phenomenography. Eighteen participants were interviewed in a semi-structured format to collect their perspectives of the Organic Chemistry lab. Eight qualitatively different ways in which students perceived the lab were uncovered and an outcome space was derived. The findings of this work are intended to inform the design of the undergraduate laboratory curriculum in chemistry that facilitate better student learning. Implications and suggestions for design of laboratory courses based on the results of this work are also presented.
Introduction and background
The chemistry laboratory is considered an essential component of chemistry education for undergraduates (Johnstone, 1991; Dechsri et al., 1997; Loucks-Horsley and Olson, 2000; Johnstone and Al-Shuaili, 2001; Hofstein and Lunetta, 2004; Stephens and Arafa, 2006; Elliott et al., 2008; American Chemical Society, 2015). The American Chemical Society (ACS) Guidelines on Undergraduate Professional Education in Chemistry (2015) recommends 400 laboratory contact hours beyond the introductory chemistry laboratory for students to receive an ACS-certified undergraduate degree. Despite this emphasis on the laboratory, the usefulness of laboratory experiences to students’ education in chemistry has been challenged. Chemistry education researchers have called for a more comprehensive look at the laboratory environment in chemistry (Nakhleh et al., 2003; Hofstein and Lunetta, 2004). For instance, Hofstein and Lunetta (1982, 2004) have reviewed several empirical studies on the laboratory and arrived at the conclusion that there is “sparse data from carefully designed studies to support faculty claims of the value of the laboratory.”In response to the need for increased research on the laboratory, a few recent studies have examined students’ goals, perspectives, and experiences in the chemistry laboratory. How students experience and perceive the laboratory is an important factor towards understanding the laboratory environment and in making curriculum decisions. Nakhleh has noted that there is a need to more fully understand what is taking place in the laboratory and that quantitative measures may not be the most valuable when attempting to do so (Nakhleh et al., 2003). The majority of studies on the laboratory have been on students in traditional or expository laboratory settings. Therefore, the purpose of this study is to investigate students’ perspectives of a non-traditional, project-based Organic Chemistry laboratory using a qualitative, phenomenographical approach. Given the high cost of laboratory instruction, and the large amounts of time invested in laboratory education in chemistry, an examination of what kinds of laboratories produce desired student outcomes is of keen interest to the chemistry education community (Nakhleh et al., 2003; Singer et al., 2012).
The project-based laboratory
For the past fifteen to twenty years, many science educators have promoted the positive impact of inquiry-based laboratory curriculums and have argued against expository or “cookbook” styled laboratories (Domin, 1999; Loucks-Horsley and Olson, 2000; Monteyne and Cracolice, 2004; Horowitz, 2007). One method that has been adopted for Organic Chemistry is the project-based approach. There are many examples of project-based laboratories in the literature over the last five years (Kiefer et al., 2012; Bliss and Reid, 2013; Graham et al., 2014; MacKay and Wetzel, 2014; Slade et al., 2014; Marchetti and DeBoef, 2015; Pontrello, 2015; Raydo et al., 2015; Mistry et al., 2016; Weaver et al., 2016; Schellhammer and Cuniberti, 2017). Project-based laboratories require students to work on multi-week, research-like projects that allow them to modify and adapt existing procedures to solve synthetic problems. In some cases, the students are free to choose their own synthetic problems, while in others the instructor chooses the synthetic target or research question. Another aspect of project-based labs is that the instructor and students may not always know the outcome of the experiment and therefore it provides students the opportunity to think about the procedures and the chemical principles behind them more deeply. Regardless of the approach, project-based labs require students to conduct a form of research. In contrast, expository or cookbook styled labs usually include weekly experiments that are not necessarily connected to each other. Also, these expository labs typically provide step-by-step instructions and the outcome of the experiment is usually known or a so-called ‘right’ answer is expected. An understanding of students’ perceptions of project-based labs will provide the chemistry community with additional evidence for how to restructure laboratory curricula with the aim of enhancing student learning.Thus far, there are limited research studies on students’ perspectives and experiences in project-based labs or their effects on learning. In one study, phenomenology was used to examine students’ experiences in a cooperative project-based General Chemistry laboratory (Sandi-Urena et al., 2011; Sandi-Urena et al., 2012). This work found that students initially experienced confusion since this new laboratory environment was unfamiliar to them. However, through increased metacognitive awareness, students were able to better understand this laboratory environment. Another study examined students experiences in an open-ended, project-based Organic Chemistry course compared with students in a more conventional “cookbook” laboratory course (Cooper and Kerns, 2006). Students in this study viewed the laboratory as a place to make mistakes and to engage in experimentation, while those in the traditional section had a more passive view of what the lab was all about. Closely related to project-based labs are research-based labs such as described for the CASPiE (The Center or Authentic Science Practice in Education) curriculum. The CASPiE curriculum involved students in an authentic research experience that provided them the opportunity to engage in scientific process skills, such as designing experiments and using experimental evidence to draw conclusions. Students who participated in CASPiE were better able to explain what they did in the lab, had a greater sense of accomplishment, and understood the application of the laboratory to their lives compared to students who were enrolled in a traditional lab (Szteinberg and Weaver, 2013).
In the section below we summarize the results of more recent studies regarding students’ perspectives of the undergraduate chemistry laboratory in more traditional laboratory settings.
Studies on students’ perspectives of the undergraduate chemistry laboratory
Recent work by DeKorver and Towns (2015, 2016) has offered insight into students’ goals for the laboratory. A meaningful learning framework was used to examine students’ cognitive, affective, and psychomotor goals for the traditional General Chemistry (2015) and Organic Chemistry laboratories (2016). In both studies, the authors concluded that students often sought out correct answers, tried to avoid mistakes in the lab, and had a major goal of finishing the lab early. Some students also expressed the goal to learn laboratory skills; however, this goal conflicted with their goal of getting out of the lab quickly.A series of quantitative studies by Galloway and Bretz used the Meaningful Learning in the Laboratory Instrument (MLLI), also based on a Novak's theory of meaningful learning, to examine students’ cognitive and affective perceptions of chemistry laboratory (2015a, 2015b). The major findings from these studies were that students had diverse affective expectations for the chemistry laboratory. In addition, many students had high cognitive expectations but their experiences in the laboratory left those expectations unfulfilled.
Galloway and Bretz also used additional qualitative research to further explore their findings in the quantitative studies (2015, 2016). Students were video recorded doing experiments and were later asked to describe what they were doing and why they were doing it. The students in this study primarily focused on the hands-on components of the lab experiments and fewer students discussed chemical concepts. Only a few students could explain the purpose of or the concepts behind the experiments. Also, it was discovered that students expressed many emotions regarding the lab, which included frustration, boredom, and enjoyment during their experiments. It is important to note that students who described similar affective experiences responded in different ways to those feelings.
A significant implication for this set of qualitative and quantitative studies is the need to better understand and incorporate the affective domain into the design of the laboratory curriculum and the need for laboratory curricula that focus on students’ decision making rather than just focus on the expected outcome of an experiment.
Research question
This study examines students’ perspectives of a non-traditional, project-based, Organic Chemistry laboratory using a framework of phenomenography. The primary goal of our study is to contribute to data on laboratories, such that, chemical educators can design laboratories that meet the goals of instructors, promote student learning of content, and help students gain important skills. To this end, our overarching research question is: What are the different ways students perceive a project-based Organic Chemistry lab?Theoretical framework: phenomenography
This study employed phenomenography as the theoretical framework. Phenomenography was originally developed for the purpose of exploring research questions in an educational context. Phenomenography is used “to define the different ways in which people experience, interpret, understand, perceive or conceptualize a certain phenomenon or aspect of reality” (Orgill and Bodner, 2004). The epistemology of this methodology is that human experiences are based on the relationship between the person and the world around them (Marton, 1994). As such, we consider both the person and their experiences as a whole. The basic assumption of phenomenography is that there is no right or wrong in the phenomenon being investigated. The researcher is not interested in what is ‘real’ but only in how the person conceptualizes the phenomenon under investigation. The participants’ statements are regarded as truthful by the researcher. Marton proposed that regardless of the phenomena under investigation there are a limited number of qualitatively different ways, which can be described (Marton, 1981). This framework is especially suited for this research study since we are primarily interested in the perspectives of students regarding the laboratory environment. Phenomenography shaped the design of this study, the data collection, and the data analysis of this work.Description of laboratory course
Organic chemistry II laboratory setting
This study was conducted at a large, urban, research intensive institution in the South East United States. The second semester Organic Chemistry laboratory is the focus of our study. This lab enrolls four sections of 44 students each semester. To better facilitate the project-based laboratory experience and to maintain the continuity of a research-like experience, this laboratory course is a half-semester format. This means that students meet for five hours twice a week for seven weeks, instead of once per week for 13 weeks.The first hour of each laboratory session is used as a pre-laboratory lecture to provide students with guidance on chemistry concepts and theory, reaction mechanisms, procedures, and safety considerations for laboratory experiments. The pre-lab is followed by four hours of practical laboratory time. During each four-hour session, there are no scheduled breaks; however, students can enter and exit the lab at any point.
All experimental procedures listed in the manual are provided with the same wording format as they were reported in peer-reviewed journals. Objectives for the course are described in the laboratory manual as follows: (1) handling and characterization of solids (including safety concerns and procedures), (2) isolation techniques of solids, planning, and execution of chemical reaction, (3) connecting lab with literature search and (4) technical report preparation. Also, stated in the lab manual is a rationale for the design of the laboratory which includes: (1) emphasize the connection between observation in the laboratory and scientific statements in literature, (2) problem-solving, (3) having a sense of accomplishment, (4) mastery of the subject and (5) enjoyment of the discovery process.
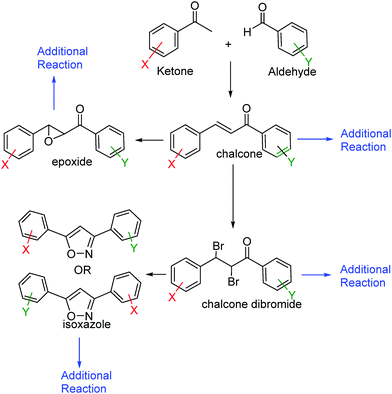
In this laboratory course, students synthesize a unique chalcone and its derivatives (Fig. 1). Each student has a unique combination of starting aldehydes and ketones, which create variations in the behavior of the synthesized compounds. Students do all of their work independently and at their own pace. They do not work in groups for any of their work. After synthesis of the chalcone, students are required to synthesize three additional derivatives – a dibromide, an epoxide, and an isoxazole (Stephens and Arafa, 2006) (Fig. 1). Additionally, students are required to synthesize two to three additional compounds of their choosing from any of the derivatives they have already made (Fig. 1). Students utilize Nuclear Magnetic Spectroscopy (NMR), Infrared (IR) Spectroscopy, and melting point data to fully characterize their compounds. They also perform their own melting point and IR spectroscopy. Literature searches using an online database (Reaxys) are also used (Tomaszewski, 2011).
At the end of the semester, students submit a comprehensive laboratory report describing the compounds they synthesized. The final report is graded on the required data collected for each compound, the quality of the writing, the quality of the discussion of the results, and how the data was used to draw conclusions about the structures of the synthesized compounds. The final exam has an essay and short answer format and assesses students’ proficiency in the following topics: reaction mechanisms, laboratory safety, general questions regarding laboratory procedures, yield calculations, proton NMR, Carbon NMR and knowledge of Hammett constants.
Methodology
Participants
A qualitative approach using purposeful sampling (Patton, 2002) was used to answer the research questions presented in this study. As is typical of phenomenographic studies, a purposeful homogeneous sample was chosen to address student perceptions of the project-based laboratory under investigation. It was important that our study participants represent students who have experienced the project-based Organic Chemistry lab and that can provide information-rich data. To qualify for the study, two requirements were met by each participant: (1) participants had to be 18 years or older, and (2) participants had to have taken both the G-Chem II and O-Chem I labs at the institution. After obtaining permission from our institution's Institutional Review Board (IRB), students were recruited within the first week of the course. During the Fall 2013 and Spring 2014 semesters, 18 students were voluntarily recruited on the first day of three standard O-Chem II lab sections to participate in the study. Once qualified, students signed consent forms and were given a $20 gift card at the completion of the pre- and post lab interviews. Students participated in one interview in the first week of the course and another interview one week before the end of the course. The majority of the study participants (14 students) described themselves as pre-med. The range of majors and student classification levels of participants in the study are shown in Table 1. Students self-reported G-Chem II and O-Chem I labs grades ranged from “A+” to “C” for both courses.| Participants (pseudonym) | Major | Classification | Research experience | |
|---|---|---|---|---|
| 1 | Anthony | Biology (Pre-med) | Junior | No |
| 2 | Catina | Biology (Pre-med) | Sophomore | No |
| 3 | Claire | Biology (Pre-med) | Senior | No |
| 4 | Denika | Biology (Pre-med) | Senior | No |
| 5 | Dominique | Biology(Pre-med) | Senior | Yes |
| 6 | Primrose | Biology (Pre-med) | Junior | No |
| 7 | Princess | Biology(Pre-med) | Senior | No |
| 8 | Shaquille | Biology(Pre-med) | Junior | No |
| 9 | Valorie | Biology (Pre-med) | Senior | Yes |
| 10 | Brandon | Chemistry | Junior | Yes |
| 11 | Cynthia | Chemistry (Pre-med) | Senior | No |
| 12 | Dali | Chemistry (Pre-med) | Junior | No |
| 13 | Edward | Chemistry (Pre-Pharm) | Sophomore | No |
| 14 | Meyers | Chemistry | Junior | Yes |
| 15 | Sterling | Chemistry | Sophomore | No |
| 16 | Daria | Psychology (Pre-med) | Senior | No |
| 17 | Dusk | Psychology (Pre-med) | Senior | No |
| 18 | Futurama | Spanish (Pre-med) | Sophomore | No |
Interviews
Interviews were conducted using a semi-structured interview protocol. The interviewer was a graduate student who had no previous contact with the students in the laboratory in any capacity. Also, the graduate student was not involved in the development of this project-based lab. During the interview, follow-up questions were used to clarify students’ responses to the initial questions. Some of the probing questions in the post interview were:(1) Please tell me about what you did in Organic Chemistry II Lab this semester
(2) Can you tell me a little bit about your experience in lab this semester?
(3) What did you think about the project?
(4) How do you think the project contributed to your learning?
The full interview protocol is shown in Appendix 1.
Participants were audio recorded in a private room removed from the laboratory environment. The post-interviews ranged from 18–41 minutes with an average time of 30 minutes. Interviews were conducted until data saturation was reached. As with qualitative studies, the number of participants required to reach saturation will vary from study to study. When we determined that no new perspectives of the laboratory were being uncovered, interviewing was stopped (Trigwell, 1994; Sandberg, 2000).
Data analysis
We decided to focus our data analysis on the post-interviews. The pre-interviews did not provide any information about how students perceive the lab they were currently in. The pre-interviews addressed their prior lab experiences and many students had a difficult time recalling their experiences in prior chemistry labs in detail. Since we were primarily interested in students’ perceptions of the Organic Chemistry lab course, the post-lab interviews were most relevant and provided the richest data for our investigation.The interviews were coded in several stages. First, interview transcripts were read and re-read and then coded by the first author via an open coding approach using the qualitative data software, NVivo 10. Codes were then revisited, revised, and elaborated as necessary using the constant comparison method (Glaser, 1965). The second author then examined the transcripts with the developed codes to validate the initial codes further. The percent agreement for these initial codes generated was 94% (Säljö, 1988; Sandbergh, 1997). Codes were then further collapsed to organize the data into themes. The organization of codes was also discussed to ensure reliability. This first approach in analyzing the data helped sort and organize the data, which ultimately provided a base for more insightful data analysis. The interviews were then analyzed for semantic themes. Semantic themes attempt to identify the explicit overall meanings of interviews. This analysis involved a summarized interpretation of the data which attempted to theorize the significance of the patterns and their broader meanings and implications (Bruck et al., 2008) compared to previous literature. We were able to describe distinct student perspectives in the lab through this data analysis method. These student perspectives are valuable products of phenomenography research because they describe the various ways students perceive labs. The final element of the analysis was compilation of a series of categories and description which were then fitted to an outcome space to reflect the increasing complexity of perspectives.
Findings and discussion
We have uncovered eight distinct ways that students perceived the project-based lab (Table 2). It is important to emphasize that individual study participants do not belong to just one category of description since they expressed their views among several (2–4 perspectives each). In the results presented in this paper, we chose to describe each of the eight categories of description using one student who exemplified that perspective.| Student perspectives | Students’ focus |
|---|---|
| Explorer perspective | Exploring the unknown in science |
| Independent researcher perspective | Cultivating independence |
| Mastery perspective | Practical understanding of concepts |
| Socialite perspective | Social interactions |
| Skill developer perspective | Developing technical skills for future career |
| Detail oriented perspective | Gathering details of lab and experiments |
| Time saver perspective | Efficiency and saving time |
| Apathetic perspective | Uninterested in the lab |
Explorer perspective
This student perspective focused on the unexplored aspects of science and embraced the unknown ambiguity of science. Sterling, a senior chemistry student who tutors chemistry and is a teaching assistant in other General Chemistry labs, typified a student who embraces the unknown nature of science. In these project-based labs, neither the professor nor the student knows the outcome of the experiment or if the synthesis will be successful. Sterling's enjoyment of the lab was based on this element of the lab. Hence, discovering that there was more information to be uncovered in the world of chemistry fueled his excitement. Expanding knowledge and applying it to the unknown was a focus of Sterling:It was nice to know that there's – it's nice to know that I don't know as much as I thought I knew. So, there's more to learn. Learning is fun to me because then you can use that information and have fun in laboratories.
Sterling gained enjoyment through the application of knowledge to new situations. Many of his decisions in lab were based on interest and discovering the unfamiliar. As noted previously, towards the end of the lab students are given the freedom to select two synthesis procedures of their choice. Sterling, driven by exploring the unknown, expressed his reasoning for choosing a procedure when he said:
The structure looked interesting. Yeah, the structures looked interesting, and then the synthesis routes required me using some things I’d never done before, ever seen before . And then I discussed with Professor X before I did it and he said they may work or they may not work. So, I should just see if I can try my hands on them. I went for it .
Ambiguity in the lab was seen as a positive challenge to Sterling. To that extent, Sterling perceived working in the lab as a basis for exploration and questioning the science he was performing. In each experiment, Sterling was able to change the procedure and adjust chemicals based on his own decisions. This freedom in the lab not only allowed him to develop independence but it also allowed for him to assess the scientific knowledge that he had learned:
I got to learn that I can't just take science as it's – just accept science as it is. I have to look into it myself or look into it further than what is explained to me to see how it really is or if that's just a theory that hasn't been supported enough yet. Because so far, I think that most of science is just a theory.
Independent researcher perspective
This student perspective focused on the cultivation of independence and thrived in the quasi-research aspect of this lab environment. Meyers, a junior chemistry student with prior research experience in biochemistry, provided an ideal example of this student perspective. Technical skills used in this lab were developed in previous G-Chem and O-Chem labs. Meyers focused on her ability to relate past lab techniques to the current O-Chem II project-based lab. The previous exposure to techniques allowed her to focus on the cultivation of independence in the lab. This point was highlighted when she said:I don’t know um they [the instructor] expect you [the student] to remember a lot of things from Orgo I lab and like how to run a reaction under reflux. Like they don’t teach you that again in the pre-lecture. So, it's like, ok, we are going to run a reaction under reflux, and it's like, oh, now I have to remember what that is. So, it's like a lot of independence stuff that you should know already. So, I just felt independent because you set up your own thing and you kind of know what you’re doing already. They’re not showing you.
Meyers discussed that she noticed how independent she was becoming in various activities inside and outside of the lab. When selecting a procedure, Meyers saw an opportunity to gain additional independence in the lab and further hone the lab skills taught to her over the years. This point was explicitly made when she discussed her views on the knowledge she gained from the lab by saying:
I mean it [lab] made you think. Like [Professor X] made it – us think. Most of the quizzes and stuff that we had to do of the reaction or the mechanism wasn’t necessarily taught. He just gave like a general path that it would go down, but we would have to like read about it, and kind of figure it out for ourselves. I think a lot of the independence came in there… the two [experiments] where you actually have to learn it yourself. So, it was challenging enough that you have to figure out what's happening in the reaction .
The unique reactant combinations each student received was perceived as one of the most valuable aspects in the lab. This difference was perceived as valuable because Meyers valued not having interference from other students in regards to her lab work. Meyers liked the separate nature of working on her own project. She explained this point when she said: I did… I liked that everybody had something different because everybody was focused on their own stuff and not peeking over at what you’re doing.
In summary, a student with an ‘independent researcher’ perspective develops understanding and finds enjoyment in the lab based on the ability to cultivate independence, unassisted by instructors.
Mastery perspective
This student perspective focused on using the laboratory to deepen their understanding of concepts. Valorie, a post-baccalaureate biology major with prior research experience in statistics, exemplified this perspective: “So, you know, I have a real good understanding – an understanding as well as being able to apply it.”One skill that was a central focus of the lab was NMR interpretation. NMR facilitated students’ ability to identify and characterize synthesized compounds, therefore Valorie focused on understanding the concept of NMR. This point was highlighted when she said:
In Orgo I lab and lecture, you know, they tell us about NMR and we have to learn it. But I didn’t really appreciate it or understand what – I mean, I understood why I was learning it and what it's used for. But I guess I didn’t understand how effective it could be until I got to the lab. So, to me, that kind of connected the dots, okay, like so NMR, especially the difference between H-NMR and C-NMR, like I know they’re different because you’re looking at the hydrogens, how many hydrogens and how many carbons you have. But in my mind, I kind of always kept them like kind of isolated from each other. So, it kind of put it together for me…
As a post-baccalaureate student, Valorie was adamant about the importance of knowledge and concept mastery in lab. Valorie's focus on understanding was also reflected in her perspective of lab work. This lab required a final report at the end of the course that assesses students’ ability to synthesize all the information they have gathered throughout the lab. Also, this report allows students to put their projects into perspective. Upon doing the final report, Valorie recognized her connection to the information beyond surface understanding. This point was brought to light when she said:
But I actually understood, like I didn’t – like with the other Organic classes, I understood the information, but I understood it just enough to do the final report. But with this class, it's like I understand it to do the report, and it was easy. I mean, I was shocked because normally it would take me days to do an orgo report just because I would have to gather all my notes and figure out, you know, this and this and figure out all this stuff. But it was just so much easier.
Students who expressed this ‘mastery’ perspective were concerned about applying previous knowledge to solve problems in the laboratory and enhance their overall learning experience.
Socialite perspective
This student perspective focused on the social aspects and interactions with others in the lab. Anthony, a junior in biology, embodied this type of student. As previously noted in the lab design section above, individualized projects were given in this lab, which prevented students from relying on others when carrying out experiments. Despite this element of the lab's design, Anthony sought to develop friendships inside of the lab. He focused on the “fun” social side of the lab and this point was highlighted when he said: It was just a fun lab in General,I made a lot of friends in that lab – it was just fun to be in there.It's just the reports and stuff that were killer.Anthony also saw student-student interactions as a way to learn more. The impact of communicating with other students in the lab provided him with new perspectives on his experiments. As such, the ability to compare and contrast different compounds and observe other student's results provided insight into other possibilities for the same experiments. Anthony highlights this in his advice for incoming students by saying:
Make friends, like for sure, make friends [is my advice]. Don't be afraid to you know put yourself out there and compare yourself to other peoples. To find out, you know, more about other people's substituents. I feel the biggest thing about working in Orgo II is being able to not only make all of your compounds and understand them but to be able to see the wide variety of compounds that you can form and what they look like.
In his interview, Anthony frequently referred to opportunities available in the lab to socialize with other students. This ability to socialize also affected his viewpoint of the lab work. Even though students had individualized work, Anthony's perception of lab work was group based and perceived through the amount of social interaction that was involved in the activity. When asked to describe what reactions he found easiest he replied by saying:
But along with [this lab being easy], it was just fun reactions I guess , in general, almost all the reactions are fun to do. There wasn’t anything too tedious. I think I did do the alpha bromination under reflux for like an hour or something. But again, we did the melting point and stuff like that, and we had our group, so we all sat there watched our stuff together .
Most of Anthony's interview focused on what he did in the “group,” even though he was aware of the lab projects required independent work (students did not work in groups). However, he decided to pursue social interactions:
So, I think even if it is not necessarily group-based, maybe I still think that the lab sets up ample opportunities for you to be able to make groups and work with people that are friendly and stuff, so I think it was good in that regard.
Anthony and other students like him perceived the lab as a social environment for pursuing social interactions. Their decisions in the lab were influenced by the volume and quality of interactions with other students. Also, this type of student gained understanding in the lab by comparing and contrasting with the experiences of other students.
Skill developer perspective
This perspective focused on building technical skills for future employment. Shaquille, a post-baccalaureate biology major, represented a student focused on developing hands-on lab skills for his future career. O-Chem II lab is often the last laboratory course for non-chemistry majors, and as such, some students expect to learn more “real world” lab skills in this course. Throughout his lab experience, Shaquille sought out direct applications and transferability of the skills he was learning in this lab to careers outside of school. His excitement for transferability of these practical skills was highlighted in the following exchange:Shaquille: Well if you remembered we talked about the professor's part in the lab and giving out more practical aspects of the lab.
Interviewer: Such as field trips and stuff?
Shaquille: Right. I don’t know if you talked to him or not [the interviewer did not talk to the professor], but right after our interview he started talking about how we could use our, you know, understanding of this lab and the experience in our future careers. And he just introduced some kind of uses that would be useful that you know – and it's related to what you are doing now. So, it's not just a regular lab. You are actually going to use these techniques in the future. And it was exciting , yeah. And that's it.
Shaquille's focused on relating lab skills to future employment was also reflected in his perception of the lab. Shaquille was concerned about how the knowledge he was learning could be directly applied to future careers. His concern for skill development was geared towards skill sets that were easy to quantify (NMR interpretation, recrystallizing, using the rotary evaporator, etc.) and readily applicable to the workforce. Shaquille explicitly expressed how he paid attention to technical skills directly related to the workforce below:
Yeah exactly, because I knew that there are some careers by just names. But making a connection with what you’re doing and what is your potential to do in the future, that was exciting. It was really exciting for me, and actually after that, I tried to, you know, ask more questions and be more practical about what I’m doing, you know. Because maybe I may use this stuff, you know, that I’ve learned today.
He then later goes on to describe how these skills can be used to improve his resume:
If you are working in a chemistry lab, you have to be able to read NMRs, IRs. That's something that you’d probably have to do a lot [in a chemistry lab], and that's not what everyone can do because – just ask my classmates. But if you learnt that then that's a really big plus on your resume.
Even when questioned on what he learned in the lab, Shaquille directly mentions employment:
When I did the synthesis procedures I actually learned that, ok, even if you can’t connect those, you know the information that you have, that ok now that I have the [compounds] that can react with something like water. And… aldehyde don’t react for example or then if you add like water, solvent and the product it's going to be like separated from the other products or reactants. So, I could make that connection with what I learned in lecture. And the other thing, it was kind of basic, but it prepared you for your future career .
Shaquille's focus on the practicality of lab work for future employment also affected his perception of lab work in many aspects. Particularly, Shaquille saw lab as a place to develop skills for future employment and believed that assessment of these labs skills should be done in the lab course:
I prefer to be tested on those [lab skills] rather than the basic knowledge of what you know. Now, you definitely have to know your stuff from the lecture, but being tested basically on that – I don’t think that's going to help for evaluating the students for the lab portion. I’d prefer that we left it in the lecture course.
The ‘skill developer’ perceived lab work through the lens of practicality and applicability of technical skills. Conceptual connections were developed based on the teachable skills that are applied to future careers.
Detail oriented perspective
This student is focused on the details of the experimental procedures and as such is overwhelmed by the ambiguity of the course. Primrose, a junior in biology with no prior research experience, epitomized the student who has difficulty dealing with the ambiguity of science and searches experimental details to avoid mistakes. As stated in the lab design section above, the outcome and results of the experiments were not outlined or defined. This element of the lab had a huge impact on Primrose's ability to cope with the unknown. Primrose focused on the mistakes that could be made in lab due to the lack of details provided for the students. Primrose expressed her disdain for the ambiguity of lab procedures and the possibility of making mistakes and failure:Interviewer: Did [your experiment] fail?
Primrose: It failed and then, that was also at the time that I was doing two reactions, the isoxazole and the isoxazoline… So, I had those on the same time and I just heated it just a little bit, but not boiling, and then added the KOH. And the instructor came over, and said that the reaction could fail and it did fail!
Interviewer: And how did you feel about that?
Primrose: I felt very sad, very mislead and just… kind of just frustrated that it [the lab manual] didn’t say to boil. It didn’t even have a note saying that this reaction could fail if you don’t heat it properly.
Interviewer: So what particularly you felt mislead you?
Primrose: The word heat. And even again she [the instructor] had printed out an extra – some extra instructions that she had come up with. And it still said just heat, it didn’t say boil , and I don’t recall anytime that she said boil in the lab.
Interviewer: So did that experience affect anything you did later on inside of the lab?
Primrose: That experience, yes, because that put me behind. Now I was two reactions behind, I had less chalcone to make, so I couldn’t just take the chalcone and just start all over again.
Primrose was slow in her lab work due to the uncertainty she experienced in the lab. From her experience in previous labs, Primrose found that focusing on details was a mechanism for avoiding mistakes. However, this current lab simulated a more research-like experience. As such, the ambiguity, uncertainty, and trial/error aspect of science hindered Primrose from using her detail-oriented approach to tackling labs to help avoid mistakes. She felt unable to rely on her abilities to interpret the correct way to carry out an experiment. Primrose spent time asking fellow students and the instructor about the procedures before she would carry out her experiments:
So, [the lab manual is] not really divided out into steps . So, for making the epoxide out of the chalcone, for example, it just says, add chalcone, add 20 ml of ethanol, add 12 ml of acetone, add 1.9 ml of NaOH, add 2.9 ml of hydrogen peroxide; but it doesn’t say that you need to dissolve the compounds right after you add each one individually. So…and I found that out through other students throughout the lab that are like, oh wait, and you guys need to add them and then shake it, and dissolve it, and just don’t pour it all in at one time . So, I was like okay.
Primrose's attention to detail was reflected in her perception of the lab manual. As mentioned previously in the lab design section above, the procedures in the lab manual were taken from experimental procedures in peer-reviewed journals. Therefore, the language used in the lab manual reflected the language chemists use to communicate experimental procedures to other chemist in the community. This means that the amount of detail was for that of a chemist and not a novice student. This presented another level of scientific ambiguity that Primrose did not like, which resulted in her having a negative perception of the lab manual. When asked about her opinion on the lab manual, Primrose responded by saying:
There was a need for more detail. It was necessary to know that your reaction could fail if you don’t boil it. It was necessary to know that if you don’t dissolve it you could have impurities. And it was necessary to know that if you just dump the bromine in it could overheat and your flask could crack and, you know, you could go back to your desk and your compound fall everywhere from your flask breaking. That was kind of important, because the instructor put emphasis on safety procedure but they didn’t really tell us things could fail . I mean that kind of looks to me if things fail and you have a job you could get fired or sued. That's really important.
This type of student perceived the project-based lab as a cornucopia of pitfalls due to its ambiguous nature. Also, this type of student's perspective seemed hindered by the lack of explicit detail.
Time saver perspective
This student perspective focused on efficiency, thus resulting in a lab experience centered on saving time. Edward, a junior chemistry student pursuing a career in pharmaceuticals, personified the type of student who experienced the laboratory environment with a focus on saving time. This aspect of saving time was not a prominent theme for participants in this study; however, Edward focused on how much time he devoted to an experiment. He even described his work in the lab based on how efficient he thought he was:Like in the beginning, I felt like I was really slow at like all the experiments, like especially with the chalcone experiment, creating your chalcone. And as the labs progressed, I actually like picked up speed and I was able to, you know, do my experiments on time.
Edward saw his progression in the lab through the lens of efficiency, which he expressed through his recollections of experiments. Many of his decisions in lab were based on saving time in lab. This was noted towards the end of the lab when students were given the freedom to select two synthetic procedures to carry out independently. Edward's decision on which procedure to select was driven by time-saving factors:
There were other [student selected additional] experiments, but I chose the 4-hydroxy because I had epoxide that I could use that was good for that experiment. And plus, it was really fast. Well, just the preparation for everything was fast .
His perception of lab work was also based on time spent performing experiments or procedures and on how much “involved” work he was required to do. Edward described the “involved” lab work as time-consuming and unfamiliar. This was viewed as his least enjoyed aspect of the lab. This point was brought to light in his description of his least favorite experiments described below:
[Epoxide was my least favorite experiment] because the epoxide experiment, that one was I think – yeah, that one was the one that took… for it you had to be really patient because you had to heat the experiment in a water bath for like 40 minutes, or 40 to 45 minutes for at about 40 degrees Celsius I think it was. And so, that took a while because you had to titrate… I forgot what solution into the compound…But you just had to keep continuously heat and watch the compound from overheating or being under – yeah. So, you had to like make sure that you kept it about 40 degrees Celsius. And yeah, if it went over, you had to put in an ice bath and put it back in. It just took a lot of time. And the other one [least favorite experiment], yeah , I think that was the one I didn’t like the most. Also, creating the chalcone, because mine did not dissolve properly in just one flask, so I had to put it several other flasks and it took a lot of time . But yeah. You know, the epoxide was the one I didn’t like the most .
Apathetic perspective
This student perspective is described as a lack of interest in all aspects of lab work. Dominique, a senior biology student, exemplified a student that was disinterested in most aspects of the lab. Dominique had no focus in the laboratory and was unable to define a purpose for the lab. Her indifference to lab work was extended to all chemistry laboratories. She viewed chemistry laboratories as purposeless, which she described by saying:Dominique: I don’t know . Like I said I think chemistry labs are like purposeless. So, I don’t know. I just kind of went and did it and left.
Interviewer: So, were there any purposes that the teacher emphasized?
Dominique: I can’t remember. Like I literally went to class, reviewed my notes so I could do well on the exam and quizzes and left. Like I’m not a chemistry person, so I kind of went in there like “oh gosh, got to get this over with” and then I left.
Dominique's perception of the lab work presented itself as something that had to be done and over with. This perception influenced multiple aspects of her lab experience.
Interviewer: Can you tell me your experience you had with the Orgo II lab this semester?
Dominique: It was cool. Like it was a lot of tedious work , but it was cool.
Interviewer: So, what about it was tedious to you?
Dominique: Just waiting for everything. Like I told you before, I don’t like to wait for labs. I don’t like it. But yeah.
Interviewer: So, is there any…When you decided to do a new lab, what type of things were you thinking about when you were making that choice?
Dominique: Just so that I don’t have to do it the next week . That's it…
Her avoidance of lab work and her need to get work done was also reflected in her perception of the lab work itself. When asked about how she felt about lab work she replied, “How do I feel about it? It was required, like I had to do it. That's about it”. Based on her explanations for her decisions above, it could appear that Dominique's motivation was simply a matter of saving time. However, when probed further, Dominique revealed that science was not a career she was interested in and that her major was simply a choice made by her parents. When asked about the purpose of labs she responded:
Dominique: I think I wasn’t trying to understand the lab. I just wanted to get it done, but I didn’t go in there like I wanted to learn something because I don’t think lab really benefits what I’m trying to do. So.
Interviewer: That's dental school, right?
Dominique: It was, but now I don’t know what I want to do. So, I don’t think it really benefits to anything that I want to do. So, I’m trying, I just want to get it done, like I graduate soon. So, I just want to get it done, so I can move on to that next step.
Interviewer: So, you have any direction you want to move in. Are you going to stay in science?
Dominique: I don’t know. I have no idea. I might get in my Masters. I was going to get it in public health but I was like why waste money if I’m not sure if that's what I want to do. Just like this biology degree it was just something my parents thought of. I’m not sure if I would have chosen biology, but then I’m not sure what I would have chosen at this point. So, I’m just going to think about, reevaluating my life, and figure out what I want to do.
Ultimately, Dominique had no critique for the lab or any explanation as to why she was trying to save time on experiments. Her focus, decisions, and perceptions of the lab all linked back to her lack of motivation for the lab and science in general. She also stated that the lab was not for her. She highlighted this point when she discussed improvements for the lab:
No [I do not have any improvements], because there's certain people who are actually genuinely interested in the stuff and it would be perfect for those people. So, I wouldn’t say there is anything to be improved on, because I think it's pretty ok for the chemistry majors and people interested in doing research for the rest of their lives or even if you are interested in going to dental or medical. So, it's great for those people, just not me.
Discussion of the outcome space
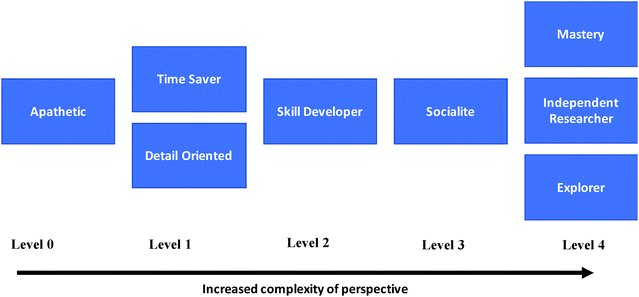
An important part of using a phenomenographical framework is to define an outcome space that describes how the student perspectives are related and fit into the laboratory experience as a whole (Marton, 1994). There are several ways to develop an outcome space. Some outcome spaces are hierarchical in nature in which the categories are logically arranged so that higher or more sophisticated perceptions also include lower ones (Laurillard, 1993). Laurillard also describes an outcome space that is arranged as a developmental progression in which each successive perception is more expansive or may be seen as better or more desirable that the one before it (1993).In this study, we define the outcome space as a developmental progression. We examined the primary focus of each category of description and arranged them in levels based on how limited or expansive their perspective of the lab is (Fig. 2). Student perspectives that are on the higher levels have a more complex or broader view of the lab, while those on the lower levels are more limited do not have the same range. We also compared each of the eight perspectives to the objectives and goals of the lab noted in the description of the lab section above to arrive at the different levels. These goals included: development of certain lab skills and techniques, report preparation, connection between observation in the laboratory and scientific statements in literature, problem-solving, mastery of the subject and enjoyment of the discovery process.
Level 0
We placed the ‘apathetic’ perspective at the lowest level of the outcome space because this was the most limited perspective of the lab. The focus of this student perspective is one that is on the outside looking in. There is a perceived lack of interest in almost all aspects of the lab and there is little to no interest in learning concepts or developing skills in the lab.Level 1
The ‘time saver’ and the ‘detail oriented’ perspectives are both on level 1. Both of these represent perspectives that were limited in focus. For the ‘time saver’ it was limited to being more efficient and finishing quickly, while for the ‘detail oriented’ the focus was to avoid mistakes by trying to get more details on the lab procedures. In addition, these perspectives are not particularly focused on any of the desirable goals of the lab such as building laboratory skills, understanding content or problem-solving. In other words, both of these perspectives are occupied with aspects that are external to the goals of the lab.Level 2
The ‘skill developer’ perspective was viewed as more sophisticated that those on levels 0 and 1 because it focused on a prominent goal of the lab, which is to develop skills. However, this perspective still has limitations because there is less emphasis about other desirable aspects of the lab that are also important such a problem-solving and mastery.Level 3
The ‘socialite’ perspective is on this level because there is a focus on interaction with others. Students with this focus perceived that their interactions with other students increased their opportunity for learning, gaining essential skills, and further understanding concepts presented in the lab. They also suggested that these interactions led to enjoyment of the discovery process, which is also a goal of this lab.Level 4
We chose to put the ‘independent,’ ‘mastery,’ and ‘explorer’ perspectives on the highest level of the outcome space because these perspectives are more expansive or complex than any of the other perspectives. All three are focused on some of the ideal goals of the lab. Their perspectives extended beyond just skill development to problem-solving, mastery of content and skills, enjoyment of the discovery process and making connections between theory and practice.Validity
Primarily, interview transcripts informed the development of various student perspectives. Explicitly detailed quotations were used to support all points made when classifying the lab perspectives and development of the outcome space. In the analysis, broader meanings and implications were discussed in relation to previous literature.Implications and conclusions
In this study, we have uncovered eight distinct ways in which students perceive a project-based Organic Chemistry lab. Again, it is important to note that several students in this study were found to possess more than one of the identified perspectives. Many of the students described perspectives related to cognitive, affective, and skill-based components of the laboratory. Designing teaching laboratories with these student perspectives in mind, can help move students into a deeper level of learning and more enjoyable and meaningful experiences. Below we describe the implications of and suggestions for laboratory design based on the student perspectives. We are not including any suggestions for the apathetic perspective because we do not believe that specific changes to the design of the laboratory course could alter a student's lack of intrinsic motivation.From the explorer perspective
Students with the ‘explorer’ perspective saw value in the opportunity to explore their experimental interests. The ‘explorer’ perspective seem to suggest that introducing elements of freedom and experimental ambiguity may be beneficial. This element of freedom allowed them to explore various experimental routes. One way to incorporate freedom into a lab is to provide students with the ability to choose an aspect of their project, as is the case for the project-based lab described in this work. Galloway and Bretz also noted that labs should be designed with increased opportunity for students to make decision and explore other possibilities without penalty (Galloway et al., 2015).From the mastery perspective
The ‘mastery’ perspective focused on using the laboratory to deepen their understanding of concepts and skills that were previously only theoretical in nature to them. This student perspective suggests that explicit emphasis on how knowledge gained from lecture courses can be used to solve problems in the laboratory and the ‘real world’ is of benefit to students and improve their enjoyment of the lab. Examples of organic chemistry labs in the literature that have the goal of providing such context for students include, the synthesis of biologically relevant compounds for treating diseases (Cheung et al., 2010), exploring green chemistry concepts (Fennie and Roth, 2016; Silverman, 2016) and everyday life applications of organic chemistry (Tami et al., 2017).From the independent researcher perspective
Based on the independent researcher perspective, labs can be designed with an individual component to them. This student perceived that not having to depend on anyone else and working on their own as a benefit. They were more focused on their own work and trying to solve any problems on their own without interference from others. DeKorver and Towns showed that when students are paired for lab experiments, they sometimes share tasks or rely on one student to do a particular task (DeKorver and Towns, 2015). This separation of tasks creates a problem where one student focuses on a task, and the other student rarely gets to perform that task. As a result, students learning of certain skills is decreased. Individualized project-based labs, as described in this work, may help to eliminate this issue because each student has to carry out all of the experiment rather than delegating parts to their lab partner. Independent work also gives students an opportunity to self-assess their work and skills and build responsibility and independence.From the socialite perspective
Professional agencies, industry, and academia describe the ability to collaborate as a desirable characteristic for a future employee (Lowden et al., 2011). The ‘socialite’ gained enjoyment and furthered their learning in the lab based on their interactions with others. It is well known that peer interaction can facilitate construction of knowledge (Vygotsky, 1980). Of note, the project-based lab described in this study does not have a structured cooperative component and this is something we need to consider for future improvement. Some ways to facilitate group work in the lab are group presentations, peer review, and group worksheets. Peer interviews can also facilitate collaboration among students. These peer interviews can consist of students comparing and contrasting their projects with each other and writing a summary or recording and uploading the conversation for credit.From the skill developer perspective
At minimum, chemistry labs have a goal of hands-on development of lab skills (Bruck et al., 2010). In the case of the ‘skill developer,’ they were excited to learn that the skills obtained in the laboratory can have practical significance for the future. Therefore, instructors may choose to provide context to students about how what they are learning in the lab can be used for their future careers. Keeping in mind that the majority of students are not chemistry majors, instructors can also encourage students to expand their thinking beyond just laboratory skills to others professional skills that can be developed in project-based labs such as problem-solving, independence, and the ability to work with others.From the time saver perspective
The ‘time saver’ was driven by being more efficient in lab. In this study, only four students expressed this view of finishing quickly to get out of the laboratory faster. In their studies, DeKorver and Towns (2015, 2016) described that many students were heavily driven by the incentive of leaving lab early. They alluded to the possible benefits students can have if the time factor was removed. Project-based labs are intended to simulate a continuous project that a student would face in a research lab. As such, there are no clear starting and stopping points for the project. We believe that this format of project-based laboratory presented in this study helped to reduce this time factor, even with the lengthy lab time. Although there were students that focused on leaving lab early, this was only a small number of students. Providing students with ambiguous finishing points can produce other motivational aspects that put the time factor into the background.From the detail oriented perspective
The ‘detail oriented’ student was occupied with avoiding mistakes and was frustrated by the lack of detail and ambiguity presented in the project-based lab. In science, there are not always clear cut answers and mistakes and uncertainty are sometimes unavoidable; however, “cookbook” labs rarely show this reality to students since they are designed to be successful (Galloway et al., 2015). Instructors can consider providing lab manuals with less specific procedures similar to those in the research literature and having students analyze real data. Educators also need to think of ways to show students that feelings of frustration are part of the scientific process and these feelings can give students an opportunity for deeper learning and exploration. Of additional importance, is the need for instructors to provide appropriate scaffolding that will help students work through the lack of detail in the laboratory manual and give them guidance of how to read and interpret experimental procedures to reduce frustration.Summary
The outcome space derived from the results of this work allows chemical educators to consider ways in which a well-designed lab curriculum can move students from limited to more diverse and complex perspectives of the laboratory. Thus far, we have considered implications for each of the student perspectives on laboratory design. However, it is also important that we discuss how these student perspectives operate holistically when laboratory design elements such as task, autonomy, ambiguity, and logistics are considered (Table 3).| Student perspective | Lab elements | Lab course design elements examples |
|---|---|---|
| Independent | Task | Balancing group versus individual tasks |
| Socialite | ||
| Explorer | Autonomy | Freedom of choice in laboratory tasks |
| Mastery | Ambiguity | Incorporation of context and ‘real life’ examples |
| Skill developer | ||
| Detail oriented | Incorporation of authentic research elements | |
| Scaffolding by instructor | ||
| Timesaver | Logistics | Continuity, length of lab session |
For instance, the ‘independent researcher’ and ‘socialite’ perspectives, though on opposite sides of the spectrum, does emphasize the need to design a laboratory that balances both of these. The inclusion of individual and group tasks in the laboratory design is important since both elements are critical for student development regardless of their career path. Students need to be able to work on their own without much guidance, but they also need to know how to work together with and learn from others. The need for group interaction as exemplified by the ‘socialite’ perspective can also extend to the ‘explorer’ perspective. Although the ‘explorer’ enjoys the freedom to explore a variety of synthetic pathways, their desire to learn can be enhanced by opportunities to interact with other students through assigned group activities.
The ‘mastery’ and ‘skill developer’ perspectives suggest the incorporation of context and ‘real life’ aspects into the laboratory design. Whereas, the ‘detail oriented’ perspective indicates the need for scaffolding by instructors at the beginning of the lab course so that students can better deal with ambiguity in experimental procedures and tasks. Finally, the ‘timesaver’ perspective suggests the consideration of logistical aspects of laboratory design. Logistical design aspects include the length of lab session and providing continuity in laboratory activities that encourage students to think more about concepts and discourage a focus on leaving the lab as quickly as possible.
The student perspectives uncovered in this study also encourages future research on chemistry laboratories that employ direct observations of students in lab and assessment of their learning. A situated cognition theoretical framework may be ideal to explore these ideas. Another aspect for future research is to investigate various patterns for students who display more than one of the perspectives.
Limitations
The study investigated a project-based lab at a single institution. Thus, the data collected might not be a reflection of all institutions; however, the diversity of this university lends to a wider scope that can capture a variety of experiences. The study also had a small number of participants (N = 18) compared to the number of students that enroll in this course per semester (N = 176). Hence, this may not be a representative sample; however, data saturation was reached for the 18 interviews conducted. Information on student learning based on each interaction was not collected. This was beyond the scope of our study, and it is something we hope to address in the future.Appendix: interview protocol
Beginning of the semester
Thank you for agreeing to participate in a study on student experiences in general and organic chemistry labs. The purpose of this interview is to learn about what students do in the laboratory, how they complete the laboratories, and what they have learned from the laboratory experiences. It is our hope that through this study we can improve general and organic chemistry lab activities to better promote student learning. Data from this and other interviews will be used to evaluate the laboratory experience you had and to inform the development and improvement of labs for future students.(1) Can you describe to me what you did inside of you past laboratory courses?
(2) What do you see as the purpose of your chemistry lab courses?
(3) What do you define as success in your laboratory courses?
(4) How successful were you in your past chemistry laboratory courses?
(a) Describe some of the most successful things that happened in the lab.
(b) Describe some of the most unsuccessful things that happened in the lab.
(5) Describe some of the things that made your past chemistry laboratory courses easy.
(6) Describe some of the things that made your past chemistry laboratory courses hard.
(7) Ask student to recall all pre-lab and lab experiences inside of Organic Chemistry I
(8) For Organic Chemistry I lab, you had a project that you had to complete, what did you think about the project?
(a) Can you describe the project you had in Organic Chemistry I Lab?
(b) What were some of the easy parts of the project?
(c) What were some of the hard parts of the project?
(d) How do you think the project contributed to your learning?
(9) If you could offer advice to a student taking either General Chemistry II Lab or Organic Chemistry I Lab, what you suggest they do to be successful in the courses? Follow up question: Why?
(10) If you could change anything about your past lab experiences, what would it be?
(11) The Organic Chemistry II Lab has a project that will last the entire semester. How do you think you will do in this lab?
(12) How do you think your experiences in General Chemistry II Lab and Organic Chemistry I Lab have prepared for the project this semester?
End of the semester
Thank you for agreeing to participate in the second part of a study on student experiences in Organic Chemistry labs. The purpose of this interview is to learn about what students do in the laboratory, how they complete the laboratories, and what they have learned from the laboratory experiences. It is our hope that through this study we can improve Organic Chemistry lab activities to better promote student learning. Data from this and other interviews will be used to evaluate the laboratory experience you had and to inform the development and improvement of labs for future students.(1) Please tell me about what you did in Organic Chemistry II Lab this semester.
(2) Describe your project in Organic Chemistry II lab this semester?
(a) Probe: Can you tell me a little bit about your experience in lab this semester?
(3) What did you see as the purpose of your Organic Chemistry II Lab?
(4) How successful were you in Organic Chemistry II Lab?
(a) Describe some of the most successful things that happened in the lab.
(b) Describe some of the most unsuccessful things that happened in the lab.
(5) For Organic Chemistry II Lab, you had a project that you had to complete, what did you think about the project?
(a) What were some of the easy parts of the project?
(b) What were some of the hard parts of the project?
(c) How do you think the project contributed to your learning?
(d) How do you think your experiences in Organic Chemistry I Lab helped prepare you for Organic Chemistry II Lab?
(6) If you could offer advice to a student taking Organic Chemistry II Lab, what would you suggest they do to be successful in the course?
(7) If you could change anything about your Organic Chemistry II Lab experience, what would it be?
(8) Since your first interview, have you participated in any additional research projects outside of your coursework?
(a) If so, what did you do?
(b) What were some of the easy aspects of doing research?
(c) What were some of the hard aspects of doing research?
Acknowledgements
We would like to acknowledge Professor A. L. Baumstark for his contributions to the development of this project-based lab. We thank Dr. Jeffery Raker for discussions that initiated this research project. We would also like to thank the Molecular Basis of Disease fellowship for providing support to NLB.References
- American Chemical Society, (2015), Undergraduate professional education in chemistry: ACS guidelines and evaluation procedures for Bachelor's degree programs, American Chemical Society.
- Bliss J. M. and Reid C. W., (2013), Medical Mycology and the Chemistry Classroom: Germinating Student Interest in Organic Chemistry, J. Chem. Educ., 90(10), 1320–1324.
- Bruck L. B., Bretz S. L. and Towns M. H., (2008), Characterizing the level of inquiry in the undergraduate laboratory, J. Coll. Sci. Teach., 38(1), 52–58.
- Bruck L. B., Towns M. and Bretz S. L., (2010), Faculty perspectives of undergraduate chemistry laboratory: Goals and obstacles to success, J. Chem. Educ., 87(12), 1416–1424.
- Cheung L. L., Styler S. A. and Dicks A. P., (2010), Rapid and convenient synthesis of the 1, 4-dihydropyridine privileged structure, J. Chem. Educ., 87(6), 628–630.
- Cooper M. M. and Kerns T. S., (2006), Changing the Laboratory: Effects of a Laboratory Course on Students' Attitudes and Perceptions, J. Chem. Educ., 83(9), 1356.
- Dechsri P., Jones L. L. and Heikkinen H. W., (1997), Effect of a labratory manual design incorporating visual information-processing aids on student learning and attitudes, J. Res. Sci. Teach., 34(9), 891–904.
- DeKorver B. K. and Towns M. H., (2015), General chemistry students’ goals for chemistry laboratory coursework, J. Chem. Educ., 92(12), 2031–2037.
- DeKorver B. K. and Towns M. H., (2016), Upper-level undergraduate chemistry students’ goals for their laboratory coursework, J. Res. Sci. Teach., 53(8), 1198–1215.
- Domin D. S., (1999), A review of laboratory instruction styles, J. Chem. Educ., 76(4), 543.
- Elliott M., Stewart K. and Lagowski J., (2008), The role of the laboratory in chemistry instruction, J. Chem. Educ., 85(1), 145.
- Fennie M. W. and Roth J. M., (2016), Comparing Amide-Forming Reactions Using Green Chemistry Metrics in an Undergraduate Organic Laboratory, J. Chem. Educ., 93(10), 1788–1793.
- Galloway K. R. and Bretz S. L., (2015a), Measuring meaningful learning in the undergraduate chemistry laboratory: a national, cross-sectional study, J. Chem. Educ., 92(12), 2006–2018.
- Galloway K. R. and Bretz S. L., (2015b), Measuring meaningful learning in the undergraduate general chemistry and organic chemistry laboratories: a longitudinal study, J. Chem. Educ., 92(12), 2019–2030.
- Galloway K. R. and Bretz S. L., (2016), Video episodes and action cameras in the undergraduate chemistry laboratory: eliciting student perceptions of meaningful learning, Chem. Educ. Res. Pract., 17(1), 139–155.
- Galloway K. R., Malakpa Z. and Bretz S. L., (2015), Investigating affective experiences in the undergraduate chemistry laboratory: Students’ perceptions of control and responsibility, J. Chem. Educ., 93(2), 227–238.
- Glaser B. G., (1965), The constant comparative method of qualitative analysis, Soc. Probl., 12(4), 436–445.
- Graham K. J., Jones T. N., Schaller C. P. and McIntee E. J., (2014), Implementing a Student-Designed Green Chemistry Laboratory Project in Organic Chemistry, J. Chem. Educ., 91(11), 1895–1900.
- Hofstein A. and Lunetta V. N., (1982), The role of the laboratory in science teaching: neglected aspects of research, Rev. Educ. Res., 52(2), 201–217.
- Hofstein A. and Lunetta V. N., (2004), The laboratory in science education: foundations for the twenty-first century, Sci. Educ., 88(1), 28–54.
- Horowitz G., (2007), The state of organic teaching laboratories, J. Chem. Educ., 84(2), 346.
- Johnstone A. and Al-Shuaili A., (2001), Learning in the laboratory; some thoughts from the literature, Univ. Chem. Educ., 5(2), 42–51.
- Johnstone A. H., (1991), Why is science difficult to learn? Things are seldom what they seem, J. Comput. Assist. Lear., 7(2), 75–83.
- Kiefer A. M., Bucholtz K. M., Goode D. R., Hugdahl J. D. and Trogden B. G., (2012), Undesired Synthetic Outcomes During a Project-Based Organic Chemistry Laboratory Experience, J. Chem. Educ., 89(5), 685–686.
- Laurillard D., (1993), Rethinking University Teaching: A Framework for the Effective use of Educational Technology, London: Routledge.
- Loucks-Horsley S. and Olson S., (2000), Inquiry and the National Science Education Standards: A Guide for Teaching and Learning, Washington, D. C.: National Academies Press.
- Lowden K., Hall S., Elliot D. and Lewin J., (2011), Employers’ perceptions of the employability skills of new graduates, London: Edge Foundation.
- MacKay J. A. and Wetzel N. R., (2014), Exploring the Wittig Reaction: A Collaborative Guided-Inquiry Experiment for the Organic Chemistry Laboratory, J. Chem. Educ., 91(5), 722–725.
- Marchetti L. and DeBoef B., (2015), Solution-Phase Synthesis of Dipeptides: A Capstone Project That Employs Key Techniques in an Organic Laboratory Course, J. Chem. Educ., 92(9), 1536–1538.
- Marton F., (1981), Phenomenography: describing conceptions of the world around us, Instr. Sci., 10, 177–200.
- Marton F., (1994), Phenomenography, in Husen T. and Postlethwaite T. N., (ed.), The international encyclopaedia of education, Oxford: Pergamon, pp. 4424–4429.
- Mistry N., Fitzpatrick C. and Gorman S., (2016), Design Your Own Workup: A Guided-Inquiry Experiment for Introductory Organic Laboratory Courses, J. Chem. Educ., 93(6), 1091–1095.
- Monteyne K. and Cracolice M. S., (2004), What's Wrong with Cookbooks? A Reply to Ault, J. Chem. Educ., 81(11), 1559–1560.
- Nakhleh M. B., Polles J. and Malina E., (2003), Learning chemistry in a laboratory environment, in Gilbert J. K., de Jong O., Justi R., Treagust D. F. and van Driel J. H., (ed.), Chemical education: Towards research-based practice, New York: Springer, pp. 69–94.
- Orgill M. and Bodner G., (2004), What research tells us about using analogies to teach chemistry, Chem. Educ. Res. Pract., 5(1), 15–32.
- Patton M. Q., (2002), Qualitative Research and Evaluation Methods, Thousand Oaks: Sage Publications.
- Pontrello J. K., (2015), Metalloprotease Peptide Inhibitors: A Semester-Long Organic Synthetic Research Project for the Introductory Laboratory Course, J. Chem. Educ., 92(5), 811–818.
- Raydo M. L., Church M. S., Taylor Z. W., Taylor C. E. and Danowitz A. M., (2015), A Guided Inquiry Liquid/Liquid Extractions Laboratory for Introductory Organic Chemistry, J. Chem. Educ., 92(1), 139–142.
- Säljö R., (1988), Learning in educational settings: Methods of inquiry, Improving learning: New perspectives, London: Kogan Page, pp. 32–48.
- Sandbergh J., (1997), Are phenomenographic results reliable? High. Educ. Res. Dev., 16(2), 203–212.
- Sandberg J., (2000), Understanding human competence at work: an interpretative approach, Acad. Manage. J., 43(1), 9–25.
- Sandi-Urena S., Cooper M. and Stevens R., (2012), Effect of Cooperative Problem-Based Lab Instruction on Metacognition and Problem-Solving Skills, J. Chem. Educ., 89(6), 700–706.
- Sandi-Urena S., Cooper M. M., Gatlin T. A. and Bhattacharyya G., (2011), Students’ Experience in a General Chemistry Cooperative Problem Based Laboratory, Chem. Educ. Res. Pract., 12, 434–442.
- Schellhammer K. S. and Cuniberti G., (2017), Competence-Based, Research-Related Lab Courses for Materials Modeling: The Case of Organic Photovoltaics, J. Chem. Educ., 94(2), 190–194.
- Silverman J. R., (2016), Biobased Organic Chemistry Laboratories as Sustainable Experiment Alternatives, J. Chem. Educ., 93(10), 1679–1681.
- Singer S. R., Nielsen N. R. and Schweingruber H. A., (2012), Discipline-Based Education Research, Washington, D. C.: National Academies Press.
- Slade M. C., Raker J. R., Kobilka B. and Pohl N. L. B., (2014), A Research Module for the Organic Chemistry Laboratory: Multistep Synthesis of a Fluorous Dye Molecule, J. Chem. Educ., 91(1), 126–130.
- American Chemical Society, (2015), Undergraduate professional education in chemistry: ACS Guidelines and Evaluation Procedures for Bachelor's Degree Programs, Washington, D.C., March 1.
- Stephens C. E. and Arafa R. K., (2006), 3,5-Diarylisoxazoles: Individualized Three-Step Synthesis and Isomer Determination Using 13C NMR or Mass Spectroscopy, J. Chem. Educ., 83(9), 1336.
- Szteinberg G. A. and Weaver G. C., (2013), Participants' reflections two and three years after an introductory chemistry course-embedded research experience, Chem. Educ. Res. Pract., 14(1), 23–35.
- Tami K., Popova A. and Proni G., (2017), Engaging Students in Real-World Chemistry through Synthesis and Confirmation of Azo Dyes via Thin Layer Chromatography To Determine the Dyes Present in Everyday Foods and Beverages, J. Chem. Educ., 94(4), 471–475.
- Tomaszewski R., (2011), A Science librarian in the laboratory: A case study, J. Chem. Educ., 88(6), 755–760.
- Trigwell K., (1994), The first stage of a phenomenographic study of phenomenography, in Phenomenographic research: Variations in Method, The Warburton Symposium, Melbourne, Australia: Royal Melbourne Institute of Technology Australia.
- Vygotsky L. S., (1980), Mind in society: the development of higher psychological processes, Boston, MA: Harvard University Press.
- Weaver M. G., Samoshin A. V., Lewis R. B. and Gainer M. J., (2016), Developing Students’ Critical Thinking, Problem Solving, and Analysis Skills in an Inquiry-Based Synthetic Organic Laboratory Course, J. Chem. Educ., 93(5), 847–851.
| This journal is © The Royal Society of Chemistry 2017 |