Aerobic Mukaiyama-type oxidation of essential oil of ortanic tangor from Reunion Island: an applied example of valorization on terpenes using salophen complexes
Keshika
Mahadeo
a,
Axelle
Pagis
a,
Jean-Baptiste
Harlé
a,
Arnaud
Marvilliers
a,
Hélène
Gérard
b,
Alain
Shum Cheong Sing†
a,
Anne
Gauvin-Bialecki
*a and
Richard
Villanneau
 *c
*c
aChembiopro, Laboratoire de Chimie et de Biotechnologie des Produits Naturels – UR, Faculté des Sciences et Technologies, Université de La Réunion, Campus du Moufia, 15 avenue René Cassin, 97744 Saint-Denis cédex 9, France. E-mail: anne.bialecki@univ-reunion.fr
bLaboratoire de Chimie Théorique, CNRS UMR 7616, Sorbonne Université, CNRS, Campus Pierre et Marie Curie, 4 Place Jussieu, F-75005, Paris, France
cInstitut Parisien de Chimie Moléculaire, CNRS UMR 8232, Sorbonne Université, CNRS, Campus Pierre et Marie Curie, 4 Place Jussieu, F-75005 Paris, France. E-mail: richard.villanneau@sorbonne-universite.fr
First published on 19th December 2025
Abstract
The work described in this comprehensive study is based on valorising essential oils from citrus originated from Réunion Island, ortanic tangor, using Mukaiyama-type aerobic oxidation of terpenes, in particular limonene. This innovative work combines extraction, fractionation and catalytic oxidation to transform an agricultural residue into valuable compounds, while respecting the principles of green chemistry. The research highlights the use of metal salophen complexes based on cheap and abundant transition metals to achieve mild epoxidation at room temperature. In addition, a theoretical study using DFT provided a better understanding of the mechanism of this reaction for the best catalyst (molybdenum containing), suggesting that the activator could be a molybdenum oxo-cis-{MoVIO2} species resulting from the evolution of the initial catalyst.
1. Introduction
Today, food processing in the agri-food industry is a major challenge in a society that increasingly favors organic farming. The use of inedible agricultural waste, such as citrus peels, is therefore receiving increasing attention. Only half of the fruit is used during citrus juice production, leaving 8 to 20 million tons of residues per year.1,2 These peel wastes are often burned, landfilled, or used as animal feed, incurring environmental and financial costs. However, citrus peels’ essential oils contain mainly non-oxygenated hydrocarbon terpenes, including 60–90% of limonene and 1–10% of pinenes. In a general way, many authors have reported the antimicrobial, antifungal, antioxidant and radical-scavenging properties of the essential oils of citrus.3 Due to their antimicrobial properties, the presence of these essential oils is the main problem for organic management options. Biocidal substances are highly ecotoxic and could therefore lead to imbalances in the environment.2,4Among the different citrus species, the ortanic tangor (Fig. 1) is a hybrid species, the result of a cross between two varieties of citrus fruit: the tangerine (citrus reticulata, a citrus close to the mandarin) and the orange (citrus sinensis). It was introduced to Reunion Island in the 1970s and accounts now for 60% of the island's total citrus-growing area.5 The composition of juices of tangor species has already been described in the literature,6 but equivalent studies on the essential oils of tangor peels are scarce.7,8
 | ||
| Fig. 1 Tree of ortanic tangor (citrus reticulata Blanco × citrus sinensis (L.)) from the Réunion Island. | ||
Valuing citrus essential oils is furthermore complicated by the fact that limonene is on the list of allergenic products for cosmetics and perfume manufacturers.9 At the same time, limonene is also considered a versatile and promising chemical precursor.10 Its transformation into valuable products paves the way for possible applications in flavors and fragrances, cleaning agents and polymer components. One possible way to value a molecule derived from biomass,11 and particularly limonene,12 is to convert it into oxygenated compounds, such as epoxides. In this respect, the most common derivative, 1,2-limonene oxide, is considered as a platform molecule which, after reaction with CO2, acids or water, can lead to the formation of compounds of interest to the perfume, cosmetics, aromatics, pharmacology and sustainable chemistry industries. By way of example, limonene epoxide is a key compound in the production of biodegradable polycarbonates.13 If we refer to the literature, an extensive study of metal-catalyzed oxidation reactions of terpenes (among which the limonene) was conducted in the last decades.12,14 In this context, preference should be given to the use of benign and abundant first-row transition metal catalysts rather than stoichiometric rare and potentially toxic reagents (chromium, osmium…), and of more atom-efficient oxidants such as dioxygen or hydrogen peroxide. Direct consumption of molecular dioxygen was first used as an oxygen source for olefin epoxidation by Mukaiyama and colleagues in the early 1990s, initially using nickel(II) complexes (1,3-diketonato).15 These compounds were rapidly replaced by other first-row transition metal catalysts,16 including chiral Mn(III)-salen complexes,17 in the presence of sacrificial aldehydes (generally pivalaldehyde or isobutyraldehyde).
In this paper, a systematic study of the aerobic oxidation of essential oil from ortanic tangor peels rich in limonene will be described. The oxidation was conducted using low-cost transition metal catalysts based on salophen ligands. An epoxidation strategy derived from the conditions used by Mukaiyama was implemented. This work follows on from our previous work on a chromium salophen derivative, which demonstrated the efficiency of such systems down to room temperature.18 However, these initial studies showed that the selectivity of the target epoxide species was low, mainly due to the instability of the complex in the presence of dioxygen and of the sacrificial aldehyde. In the present manuscript, the extraction and the fractionation of the essential oil found in the peels of ortanic tangor of Reunion Island were first described. The careful analysis of the essential oil and its hydrocarbon fraction was also detailed. In a second time, the benefits of carrying out the Mukaiyama-type oxidation of the hydrocarbon fraction of the essential oil in the presence of a stabilizing co-ligand such as N-methylimidazole were demonstrated. Finally, we also attempted to propose by a DFT study a plausible mechanism for the epoxidation of a model substrate using the best metal (molybdenum) catalyst determined in the course of this study.
2. Experimental
(See SI for the synthesis of the catalyst and Table S1 for details about the origin of the reagents and solvents used).2.1. Plant material
Solvents and reagents were obtained from commercial sources and used as received. Ortanic tangor epicarps were collected from SETAA (Société Exploitation De Techniques Agro-Alimentaires), a subsidiary of CILAM (Compagnie Laitière des Mascareignes).2.2. Essential oil extraction
Ortanic tangor epicarps were hydrodistilled in a Clevenger-type apparatus for 3 h. The oil was taken up thanks to a liquid–liquid extraction using CH2Cl2. The organic phase was recovered and traces of water were removed with Na2SO4. The solvent is removed under reduced pressure using a rotary evaporator. The essential oil was conditioned under N2 and stored in a refrigerator at +4 °C. The extraction yield (0.2%) was calculated based on the weight of fresh plant material.2.3. Fractionation of the essential oil
The essential oil (2 g) was placed at the top of a silica column (Fluka Analytical silica gel 60), 3 cm in diameter and 20 cm long, prepared with isohexane as the eluent. Using a vacuum pump, the hydrocarbon fraction (Fraction H) was first eluted with 500 mL of isohexane and then the oxygenated fraction (Fraction O) was eluted with 500 mL of ethyl acetate. The solvent was removed for both fractions using a rotary evaporator and stored under N2 in a refrigerator at +4 °C. 1.206 mg and 0.807 mg were obtained for Fraction H and Fraction O, respectively.2.4. Identification and quantification of volatile compounds by gas chromatography analyses
Gas chromatography (GC) analyses using a non-polar SPB-5 capillary column (60 m, 0.32 mm i.d., film thickness 0.25 µm) were carried out with an Agilent 8890N GC system equipped with a flame ionization detection (FID) system for quantitative analysis and a mass spectrometer (MS) system (Agilent 5977B) for qualitative analysis. GC-MS instrument was operated at 70 eV in the EI mode over the m/z range 20–400 uma. The oven temperature was programmed from 60 to 250 °C at 4 °C min−1 and then isothermal at 250 °C for 30 min. The transfer line was at 250 °C. Injector and detector temperatures were maintained at 250 °C and 300 °C, respectively. Helium was used as the carrier gas at a flow rate of 1 mL min−1. Samples were injected in split mode (split ratio 1/50; injection volume 1 µL).Constituents of the samples were identified by comparison of their retention indices and their mass spectral fragmentation pattern with those reported in the literature (Adams, 2017) and stored on MS Libraries (NIST 2.4). Retention indices of all the constituents were determined by Kovats method using n-alkanes as standards. The mixture of n-alkanes (C8–C22) was prepared from pure chemicals at 5% concentration in pentane.
The quantification of the components was performed based on their GC peak areas on the SPB-5 column without FID response factor correction.
2.5. Catalytic oxidation of fraction H of the essential oil
- Preparation of the reaction mixture: In a pillbox, were added 1 mL of Fraction H of the essential oil (corresponding to ca. 5.71 mmol of limonene), 30 µL (0.35 mmol) of N-methylimidazole, 0.9 mL (7.3 mmol) of p-xylene (as internal standard) and 5 mL of solvent at T = 20 °C.- t0 sample: 0.5 mL of the previous mixture was introduced into a 10 mL volumetric flask, filled up to 10 mL with CH2Cl2, and analysed by GC/MS-FID (see paragraph 2.4 for details).
- Addition of the catalyst and start of the catalytic reaction: 0.07 mmol of catalyst (ca. 44 mg) was weighed and added to the previous mixture, the whole being introduced into a three-neck flask topped with a condenser, a glass stopper and a glass cannula in which the air had been replaced by O2. Then, 2 mL (21 mmol) of isobutyraldehyde (IBA) was added to the reaction medium and the system was kept under stirring for 24 hours at T = 20 °C.
- t24 sample (end of reaction): After 24 hours, 0.5 mL of the reaction medium was removed and diluted with 2 mL CH2Cl2. Then, 2 mL of an aqueous solution of NaHCO3 was added to neutralize the acids present. The organic phase was recovered and passed over a silica plug to remove the catalyst. The organic phase was eluted with 2 × 2 mL CH2Cl2, transferred to a 10 mL volumetric flask which was filled with CH2Cl2, and analyzed by GC/MS-FID (see paragraph 2.4 for details).
2.6. Computational methods
All calculations, minimum structures and transition states, were obtained at DFT level using the B3LYP19 functional implemented in GAUSSIAN 09 program package Revision D.01.20 No dispersion correction was applied along with the geometry optimizations. The 6-31G** basis set was used for all atoms except Mo, for which the LANL2DZ effective core potential and associated basis sets are used.21 This computational level was chosen to allow direct comparison with previous studies carried out for Cr.18 As this basis set is known to be outdated for second row transition metals,22 energies were corrected by an additional single point computations23 using a larger basis set (def2-TZVPP)24 and adding empirical dispersion corrections using the D3 version of Grimme's dispersion with Becke–Johnson damping.25 However, only minor changes were obtained, which justifies our approach not to reoptimize the geometry with this computational level. The connectivity of the transition state was ensured by imposing a small displacement to the TS along the imaginary frequency mode and carrying out optimization. The Gibbs free energies were calculated using the standard procedure implemented in Gaussian (ideal gas, rigid rotor approximation, T = 298 K and P = 1 atm as standard conditions) and then corrected to a standard state of 1 mol L−1 using the goodvibes26 post-processing software.3. Results and discussion
3.1. Composition of ortanic tangor peels essential oil and the hydrocarbon fraction
In traditional medicine, like other citrus fruits such as tangerine, tangor essential oil is particularly useful for relieving digestive problems, soothing stress, and improving metabolic disorders.27 As mentioned in the introduction, only two studies carried out on two different varieties of tangor fruits have been published in the literature so far. Thus, Cassab Azevedo and coworkers on the one side,7 and Choi and coworkers on the other side,8 identified up to 43 compounds in the essential oil of two varieties of tangor. Monoterpene hydrocarbons such as limonene (>85%), β-myrcene or sabinene, account for the majority of compounds identified. However, many oxygenated compounds were also identified; octanal and linalool, for example, are often encountered in the latter family of compounds. However, the authors also showed that the composition of essential oils was significantly affected by fruit maturity, the vegetative stage of the plant, storage conditions as well as the extraction method. In particular, Choi and coworkers showed that during the cold pressing method, oxidation occurred easily because the sample was agitated with air.8 This specific extraction method was therefore discarded in our study in favor of hydrodistillation, a technique that some of us have been using for decades,28 so as not to distort the results of the catalytic oxidation of the different fractions that we carried out in a second phase.The essential oil of the peels of ortanic tangor was first obtained by hydrodistillation for three hours. Its fractionation was performed by preparative chromatography on a silica column. Two fractions were thus separated after successive elution with isohexane and acetonitrile, designated as the hydrocarbon (fraction H) and oxygenated fractions (fraction O). The Mukaiyama-type oxidation of fraction H was then performed in a second step (see Paragraph 3.2), leading to an oxidized hydrocarbon fraction (fraction OH). The general outline of all these stages is shown in Scheme 1.
 | ||
| Scheme 1 Representation of the different stages of the extraction/oxidation process used in this work. | ||
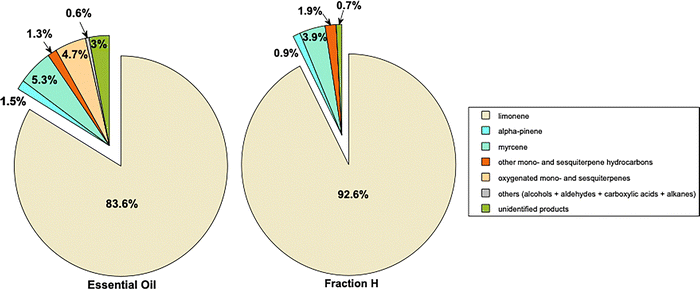
The results of the GC/MS-FID analyses of the essential oil and fraction H are presented in Scheme 2 (see SI Fig. S1 and S2 for the corresponding chromatograms). In addition, SI Table S2 details the contents of each chemical compound identified and listed by chemical family and in order of increasing Linear Retention Indices (see SI Method for Identification of the volatile compounds paragraph for details). Thus, 17 compounds were identified in the essential oil and 21 compounds in fraction H, giving identification rates of 97.0% and 99.3% respectively. Limonene was confirmed as the major component of the essential oil and the hydrocarbon fraction. By way of comparison, Cassab Azevedo and coworkers7 quantified 26 components and found 92.4% limonene in the essential oil of Murcott tangor (C. reticulata Blanco x C. sinensis). In their study, the other main components were linalool (2.4%), octanal (1.5%) and myrcene (1.4%).7 It is noteworthy that the content of myrcene was particularly high in the present work (5.3%). As expected, fraction H was enriched in hydrocarbon monoterpenes (total ca. 99.0%) and sesquiterpenes (0.3% vs. 0.1% in the essential oil) and no oxygenated species were detected. It is noteworthy that extraction of the essential oil from the leaves of the ortanic tangor was also performed in parallel, the results being showed in SI.
3.2 Catalytic oxidation of the hydrocarbon fraction (fraction H) through Mukaiyama-type epoxidation
 | ||
| Scheme 3 Representation of the Mukaiyama-type epoxidation of alkene with O2 and a sacrificial aldehyde. | ||
In the present study, we used a series of salophen-based metal catalysts, based on the previously synthetized N,N′-bis(3,5-di-tert-butylsalicylidene)-1-carboxy-3,4-phenylene-diamine ligand (salophenH2).29,30 The choice of metal precursors was initially based on their use in conventional oxidation processes described in the literature (MnIII, CrIII, {VI![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O}, {MoIV
O}, {MoIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O}).31 In addition, CoII and NiII complexes were added to this study. We have shown that these same complexes exhibit good activity in CO2 cycloaddition reactions on epoxides, reactions that can be coupled with alkene epoxidation reactions to form cyclic carbonates from alkenes.
O}).31 In addition, CoII and NiII complexes were added to this study. We have shown that these same complexes exhibit good activity in CO2 cycloaddition reactions on epoxides, reactions that can be coupled with alkene epoxidation reactions to form cyclic carbonates from alkenes.
Thus, six different complexes were prepared according to the protocols initially described by Luts and Papp,29 with respectively {MoI![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O}2+ ([Salophen-{MoIVO}]), NiII ([Salophen-{NiII}]), {VIV
O}2+ ([Salophen-{MoIVO}]), NiII ([Salophen-{NiII}]), {VIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O}2+ ([Salophen-{VIVO}]), {CrIII–Cl}2+ ([Salophen-{CrIII–Cl}]), {MnIII–Cl}2+ ([Salophen-{MnIII–Cl}]) and CoII ([Salophen-{CoII}]) ions (see Fig. 2). Indeed, ([Salophen-CrIIICl]) complex previously proved to be a convenient pre-catalyst at room temperature for the Mukaiyama-type epoxidation of limonene, and for which we have proposed a mechanism based on the formation of several successive {salophen-CrV
O}2+ ([Salophen-{VIVO}]), {CrIII–Cl}2+ ([Salophen-{CrIII–Cl}]), {MnIII–Cl}2+ ([Salophen-{MnIII–Cl}]) and CoII ([Salophen-{CoII}]) ions (see Fig. 2). Indeed, ([Salophen-CrIIICl]) complex previously proved to be a convenient pre-catalyst at room temperature for the Mukaiyama-type epoxidation of limonene, and for which we have proposed a mechanism based on the formation of several successive {salophen-CrV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O} active species.16 Furthermore, to improve the catalytic efficiency of the catalyst, N-methylimidazole was added to the reaction mixture as a co-ligand. Indeed, as in the case of iron porphyrin complexes,32 the presence of imidazole ancillary ligands showed a significant effect on the reactivity of the catalysts in the presence of O2. The use of this co-ligand in the Mukaiyama type epoxidation process was also mentioned very early on in the literature.17,33
O} active species.16 Furthermore, to improve the catalytic efficiency of the catalyst, N-methylimidazole was added to the reaction mixture as a co-ligand. Indeed, as in the case of iron porphyrin complexes,32 the presence of imidazole ancillary ligands showed a significant effect on the reactivity of the catalysts in the presence of O2. The use of this co-ligand in the Mukaiyama type epoxidation process was also mentioned very early on in the literature.17,33
As showed earlier,18 this reaction hardly proceeded in the absence of catalysts at room temperature (blanks 2 and 4, see SI Table S3). Indeed, this reaction involves the formation of organic radicals from the sacrificial aldehyde, such as the corresponding peracyl radical. The latter results from the initial breaking of the C–H bond of the aldehyde (IBA) and the subsequent reaction of the acyl radical with molecular dioxygen (see Scheme 4). Regarding the initial breaking of the C–H bond, which is the rate-determining step, the activation Gibbs energy without a metal catalyst was found rather high (ΔrG# = 26.6 kcal mol−1),18 so the use of metal catalysts seemed unavoidable. Finally, the comparison of blank 1 with the reference experiment (including all the possible mixture components, see Table S3) was also instructive as it allowed the contribution of the methyl-imidazole co-ligand to be assessed. Under the reaction conditions used, the presence of this co-ligand appears particularly interesting as it results in gains both in terms of substrate conversion (80 vs. 99.5%) and monoepoxide yield (45 vs. 57%).
 | ||
| Scheme 4 Simplified aldehyde autoxidation pathway according to Nam and Valentine.23 | ||
The catalytic epoxidation reaction conditions were then optimized using the Jacobsen catalyst again by varying the solvents and the reaction temperatures, assuming that the limonene: N-methylimidazole![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cat ratio remained identical in all experiments (Fig. 3). At T = 20 °C, it was possible to observe complete limonene conversion at t = 24 h for the tests carried out in toluene, ethanol and acetonitrile, with the test in anisole showing a slightly lower conversion rate (91%). However, in ethanol, acetonitrile and anisole, a large amount of the reaction products (between 30 and 50%) was not detected by GC/MS-FID. It is likely that in this case, the formation of polymeric species or at least oligomers was responsible for this deficit in the mass balance, as previously observed by some of us.30b In brief, the solvent that gave the best conversion rate and the best mass balance was toluene (mass balance deficit <9%).
cat ratio remained identical in all experiments (Fig. 3). At T = 20 °C, it was possible to observe complete limonene conversion at t = 24 h for the tests carried out in toluene, ethanol and acetonitrile, with the test in anisole showing a slightly lower conversion rate (91%). However, in ethanol, acetonitrile and anisole, a large amount of the reaction products (between 30 and 50%) was not detected by GC/MS-FID. It is likely that in this case, the formation of polymeric species or at least oligomers was responsible for this deficit in the mass balance, as previously observed by some of us.30b In brief, the solvent that gave the best conversion rate and the best mass balance was toluene (mass balance deficit <9%).
The reaction temperature was also adjusted around the initially tested temperature of 20 °C (reactions carried out in toluene). As expected,34 a decrease in temperature (T = 10 °C) led to a lower conversion (93.0% vs. 99.5%) associated with a slight increase in the yield of epoxides (60% compared with 56.5% at 20 °C). Conversely, a moderate increase (in this case T = 35 °C) quickly resulted in a significant fall in monoepoxide yield (45%). Consequently, all the reactions presented in the following paragraph will be carried out in toluene at T = 20 °C.
The CoII and {MnIIICl} complexes were both interesting since the cumulative yields in epoxides (mono + diepoxides) were satisfactory (ca. 70% and 81% respectively). However, the significant loss of mass observed with these two catalysts (undoubtedly linked to the formation of polymeric/oligomeric species as indicated above) means that they cannot be used for larger-scale applications. The best selectivity in monoepoxide was obtained with the [salophen-{MoIVO}] complex (noted complex Mo0 in the following) yield of 69.5% corresponding to a selectivity of 71%, bearing in mind that the secondary product of the reaction is the diepoxide (yield of 26.6%, selectivity of 27.2%). We therefore have here a particularly efficient catalyst for the epoxidation of the endocylic double bond of the limonene since the sum of identified reaction products in which this double bond has undergone epoxidation exceeds 96% after 24 h at room temperature. This catalyst was also highly selective in that the mass balance was complete (all the compounds were detected by GC/SM-DIF), and in that only small amounts of secondary products (1.7%) were detected and quantified.
Although limonene is not the only constituent of the hydrocarbon fraction of the essential oil, the low initial concentration of the other monoterpenes prevents us from really quantifying the other compounds oxidized during the reaction. However, it is possible to observe the persistent presence of myrcene and α-pinene at the end of the reaction, a sign at best of incomplete oxidation of these substrates, which are the two most abundant among the minority species in fraction H. At this stage, the oxidation products of these compounds have not yet been identified, let alone quantified, because they are not present in sufficient amounts in the final mixture.
Regarding the specific behavior of Mo0, the literature suggests that Schiff base-{MoIVO} complexes are in fact precatalytic species, which under oxidizing conditions led to the formation of a Mo(VI) complex.31 The first idea would be that the active species corresponded to the simple transformation of the {MoIVO} complex Mo0 into the corresponding cis-{MoVIO2} dioxo species (namely [Salophen-{MoVIO2}], complex Mo1). It is noteworthy that such complexes containing cis-{MoVIO2} entity associated with one or more Schiff bases were regularly used in the past for epoxidation reactions with various oxidants (H2O2, TBHP, cumene hydroperoxide…).35
We thus synthesized Mo1 according to the procedure described in the literature for similar Mo(VI) salen or equivalent complexes (see SI synthesis of the catalysts paragraph for details). The catalytic study under the same experimental conditions as above showed that this complex exhibited performances very close to those observed for the Mo(IV) Mo0 complex after 24 h of reaction at room temperature: the conversion of limonene after 24 h was 95.4%, while the corresponding selectivity in mono- and diepoxide was respectively 65.4% and 29.4% (see SI Fig. S4). Another interesting common point is that in both cases, an induction period of around one hour was observed before the onset of catalytic activity. In order to elucidate or at least to give some clues about the catalytic behavior of the Mo0/Mo1 system, we tried to identify by a DFT study the aerobic epoxidation mechanism.
3.3. Computational investigation of the [Salophen-{MoIVO}] mono-oxo complex (Mo0) and its oxidized forms for the epoxidation of alkene
To our knowledge (but the literature is particularly rich in this field), the present study is the second one in which aerobic epoxidation of alkenes was implemented using a molybdenum complex associated to Schiff base ligand,36 the present work being the first example at room temperature. In the continuity of this work, we thus have undertaken a DFT study in order to understand the oxidation pathway of the Mo(IV) precursor Mo0 as well as to determine the mechanism of the alkene epoxidation catalyzed by the oxidized form Mo1. | ||
| Fig. 5 Gibbs energy profiles (in kcal mol−1) for oxidation of Mo0 with O2 for the triplet (red) and singlet (black) spin states. | ||
The most stable spin state for complex Mo0 is a low spin 0 (namely Mo0S) with a d2 electronic structure in which the two electrons are paired. However, another electronic configuration with a triplet state (Mo0T) was also found only 3.4 kcal mol−1 higher in Gibbs energy. In the latter case, the spin population analysis is consistent with a partial reduction of the salophen ligand associated with a d1 electronic configuration at Mo (Mulliken spin population (pop) = 1.15 e) characteristic of Mo(V) oxidation state, the second unpaired electron being spread all over the salophen ligand (see SI Table S4).
We then examined the interaction of dioxygen with complex Mo0 (Fig. 5) The coordination of triplet O2 with singlet Mo0S to lead to [Salophen-{MoVIO(η1-O2)}] in a triplet state (complex Mo2η1T, see Fig. 6 and SI Table S5) is found to be quasi athermic (+0.9 kcal mol−1). The two electrons are located on both the metal (0.76 e) and the two oxygen atoms of the O2 ligand (0.74 and 0.45 e respectively), therefore suggesting a Mo(V) with a d1 configuration and a superoxo O2−˙ ligand. Formation of this adduct goes through a low Gibbs energy TS (TS0-2T, +10.4 kcal mol−1 above separated reactants, see SI Table S6). A more stable (by −19.4 kcal mol−1 compared with Mo0S) η2-O2 complex was obtained, but in the singlet state, namely [Salophen-{MoVIO(η2-O2)}] (complex Mo2η2S). This complex can thus be described as a diamagnetic oxo-η2-peroxo d0 MoVI complex (see SI Table S5), which can be obtained from the η1-O2 singlet complex Mo2η1S, which is +0.6 kcal mol−1 below Mo0S. Connection between the η1 and η2 singlet complexes takes place in the singlet state viaTS2η1-2S (see Table S6) which is +13.4 kcal mol−1 above Mo2η1S. A TS for formation of Mo2η1S from the separated reactant could be obtained (TS0-2S, see SI Table S6), but very high in energy (see SI Fig. S6). Given the proximity in energy of the Mo0 and the two Mo2η1 complexes respectively, we can safely assume that the most stable Mo2η2S complex is reached through changes of the ground spin state along the mechanism.
3.3.2.1. Direct oxygen transfer by Mo1 and Mo2. We first evaluated the direct epoxidation of ethylene (taken here as the model substrate) by the lowest energy complex Mo2η2S, according to path a in Scheme 5. This reaction was found to take place in a one-step mechanism, with a high activation Gibbs energy of +38.2 kcal mol−1 (see Fig. 7). The arrival of C2H4 at the complex Mo2η2S yields the epoxide by direct transfer of an oxygen atom of the η2-peroxo ligand, the products of the reaction, i.e.Mo1 (see Fig. 6) and the 1,2-epoxyethylene, being very stable by −39.8 kcal mol−1 (see SI Table S7). It is noteworthy that the same reaction, i.e. the approach of the ethylene, at the triplet state complex Mo2η1T also led to TS located 21.2 kcal mol−1 above Mo2η1T, and thus 41.4 kcal mol−1 above Mo2η2S, due to strong stability of the latest with respect to Mo2η1T (see Table S7). Thus, even if access to the dioxo Mo1 complex can be achieved by direct reaction between the ethylene substrate and the Mo2 complex, whatever the spin state considered, the latter can hardly be proposed as the true active species for ethylene epoxidation, with regard to the energy of the corresponding transition states. On the other hand, it is fully compatible with an initiation step that allows the compound Mo1 (which proves to be particularly stable) to be obtained from Mo0, as has been demonstrated experimentally (see paragraph 3.2.3).
In the same manner, we also verified the possibility of direct oxygen transfer between the cis-dioxo Mo1 complex and ethylene, potentially leading to the formation of the reduced Mo0 complex, according to path b in Scheme 5. In that case, the mechanism takes place in two steps, the first one corresponding to the addition of the ethylene to one oxo ligand of Mo1, leading to a single state [Salophen-{MoVIO(![[O with combining low line]](https://www.rsc.org/images/entities/char_004f_0332.gif) C2H4)}] complex (Mo3S, see Fig. 7 and Table S8), in which the epoxy is coordinated to the metal. The corresponding TS, which has a very high activation Gibbs energy (+51.2 kcal mol−1, see SI Table S7), points out to the formation of a {MoVIO(O+–CH2CH2−)} moiety which results from an attack of one oxo ligand to one carbon of the ethylene. The corresponding Mo–O distance was found equal to 2.05 Å, significantly longer than the Mo
C2H4)}] complex (Mo3S, see Fig. 7 and Table S8), in which the epoxy is coordinated to the metal. The corresponding TS, which has a very high activation Gibbs energy (+51.2 kcal mol−1, see SI Table S7), points out to the formation of a {MoVIO(O+–CH2CH2−)} moiety which results from an attack of one oxo ligand to one carbon of the ethylene. The corresponding Mo–O distance was found equal to 2.05 Å, significantly longer than the Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O distance (1.72 Å) with the remaining terminal oxo ligand. The formation of Mo3S is found endergonic by +32.5 kcal mol−1 from the separated reagents and is followed by decoordination of the epoxide, which takes place with no energy barrier, leading to the formation of the Mo0S species. The global reaction in that case is found strongly endergonic by 28.3 kcal mol−1. The high activation barrier (around 50 kcal mol−1) combined with endothermicity and a very high-energy reaction intermediate mean that this route is totally inaccessible. In other words, regeneration of Mo0S from Mo1 by epoxidation of ethylene proves impossible.
O distance (1.72 Å) with the remaining terminal oxo ligand. The formation of Mo3S is found endergonic by +32.5 kcal mol−1 from the separated reagents and is followed by decoordination of the epoxide, which takes place with no energy barrier, leading to the formation of the Mo0S species. The global reaction in that case is found strongly endergonic by 28.3 kcal mol−1. The high activation barrier (around 50 kcal mol−1) combined with endothermicity and a very high-energy reaction intermediate mean that this route is totally inaccessible. In other words, regeneration of Mo0S from Mo1 by epoxidation of ethylene proves impossible.
3.3.2.2. Epoxidation using RC(
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/i_char_e001.gif) O)OOH peracid.
However, as mentioned previously, we described in a recent article the mechanism of ethylene epoxidation in Mukaiyama-type oxidation process using the [Salophen-{CrIIICl}] derivative.18 We showed for such a reaction which requires the use of a sacrificial reducing RCHO aldehyde, that the true oxidant was most likely the corresponding RC(
O)OOH peracid.
However, as mentioned previously, we described in a recent article the mechanism of ethylene epoxidation in Mukaiyama-type oxidation process using the [Salophen-{CrIIICl}] derivative.18 We showed for such a reaction which requires the use of a sacrificial reducing RCHO aldehyde, that the true oxidant was most likely the corresponding RC(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)OOH peracid. It may be worth reminding that in the case of Cr, this peracid enabled to obtain a {CrV
O)OOH peracid. It may be worth reminding that in the case of Cr, this peracid enabled to obtain a {CrV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O} function, responsible for the transfer of the oxygen atom during the epoxidation of the alkene.18 Obviously, this cannot be applied in the case of Mo(VI) oxo complexes, as we have shown that the Mo1 and Mo2 forms have activation barriers that are too high for catalytic reactions. We are therefore not in the case of redox catalysis.
O} function, responsible for the transfer of the oxygen atom during the epoxidation of the alkene.18 Obviously, this cannot be applied in the case of Mo(VI) oxo complexes, as we have shown that the Mo1 and Mo2 forms have activation barriers that are too high for catalytic reactions. We are therefore not in the case of redox catalysis.
On the other hand, epoxidation reactions catalyzed by cis-dioxo molybdenum(VI) complexes have been widely described in the literature using various oxidants, including peracids.35 Several mechanisms exist in which the interactions between the catalyst and the reaction substrates are variable. As part of this work, we thus also examined the behavior of complex Mo1 in the presence of CH3C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)OOH (used here as a model oxidant) for the epoxidation of ethylene. First, building direct covalent interaction between Mo1 and the organic reactants has to be rejected. Indeed, as mentioned above, we have seen that the initial reaction of ethylene with the oxo ligand is highly unfavourable (+51.2 kcal mol−1 for activation Gibbs energy). We then studied the direct reaction of Mo1 with the peracid (in the absence of ethylene) to form Mo2. We found that this reaction again implies a high transition state (+35.2 kcal mol−1 with respect to separated reactants, see Fig. 8A and SI Table S9), which indicates that it is not possible to epoxidize ethylene by this route.
O)OOH (used here as a model oxidant) for the epoxidation of ethylene. First, building direct covalent interaction between Mo1 and the organic reactants has to be rejected. Indeed, as mentioned above, we have seen that the initial reaction of ethylene with the oxo ligand is highly unfavourable (+51.2 kcal mol−1 for activation Gibbs energy). We then studied the direct reaction of Mo1 with the peracid (in the absence of ethylene) to form Mo2. We found that this reaction again implies a high transition state (+35.2 kcal mol−1 with respect to separated reactants, see Fig. 8A and SI Table S9), which indicates that it is not possible to epoxidize ethylene by this route.
Direct epoxidation of ethylene by peracetic acid is found to be highly exergonic (−41.8 kcal mol−1). At this point, comparison with the literature proved particularly instructive. Indeed, it has been shown on several occasions that epoxidation mechanisms mediated by peroxides can involve weak hydrogen bonds between the complex and the peroxide.35g,37 We therefore computed the epoxidation reaction of ethylene by peracetic acid, assisted by hydrogen bonds involving the complex Mo1 (see Scheme 5, path d) and the peracid. In these conditions, the epoxide is obtained through oxygen transfer from the peracid within a single step with a relatively low activation Gibbs energy at +25.6 kcal mol−1 (see SI Table S9). This TS is characterized by a short H⋯O distance (1.67 Å, see Fig. 8c) between one of the oxo ligand and the H atom of a {CH2CH2OH}+ intermediate, which results from the transfer of a {OH}+ from the peracid onto the ethylene. By comparison, the activation barrier for direct ethylene epoxidation by peracetic (without metal) acid was found equal to +24.3 kcal mol−1 in the absence of any metal at the same computational level.
We also found an alternate mechanism involving complex Mo1 in which no direct interaction was observed (see Scheme 5, path e). This mechanism is also characterized by a low activation energy calculated at +21.2 kcal mol−1 (see Fig. 8 and SI Table S9), in the same range than the two previous ones (i.e. that involving hydrogen bonds or in the absence of the catalyst).
As a matter of fact, all three energy profiles (including the value of final energy of the products) are virtually identical, which seems to indicate that in this case the complex Mo1 has little influence on this step. The particular efficiency of this catalyst must therefore be sought elsewhere.
3.3.2.3. Attempted epoxidation using RC(
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/i_char_e001.gif) O)OO− peracetate.
Thus, we also envisaged the initial deprotonation of the peracid by the N-methylimidazole initially used a co-factor in the catalytic oxidation process. We thus checked the direct interaction of Mo1 with the peracetate ion CH3C(
O)OO− peracetate.
Thus, we also envisaged the initial deprotonation of the peracid by the N-methylimidazole initially used a co-factor in the catalytic oxidation process. We thus checked the direct interaction of Mo1 with the peracetate ion CH3C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)OO−. We found this ion able to link to the molybdenum(VI) cation leading to the formation of a penta-coordinated anionic cis-{MoO2} complex (Mo4S, Fig. 9 and SI Table S8). It is noteworthy that the two N atoms from the imidazole functions of the salophen ligand find themselves decoordinated from the metal (N–Mo distances greater than 2.6 Å), the salophen then playing the role of a bidentate ligand via the two phenolate oxygen atoms. The coordination sphere of the cation is also completed by the O− atom of the peracetate.
O)OO−. We found this ion able to link to the molybdenum(VI) cation leading to the formation of a penta-coordinated anionic cis-{MoO2} complex (Mo4S, Fig. 9 and SI Table S8). It is noteworthy that the two N atoms from the imidazole functions of the salophen ligand find themselves decoordinated from the metal (N–Mo distances greater than 2.6 Å), the salophen then playing the role of a bidentate ligand via the two phenolate oxygen atoms. The coordination sphere of the cation is also completed by the O− atom of the peracetate.
In the second step, the epoxide is formed in a single step with a fairly high activation barrier of +39.5 kcal mol−1. The transition state can be described as an adduct between ethylene, an acetate ion and a tris-oxo Mo(VI) complex in a meridional configuration, characterized by the de-coordination of the two N atoms of the salophen ligand. The reaction product corresponds to a neutral penta-coordinated cis-{MoO2} complex in which the metal is linked to the formed epoxide, after liberation of the acetate ion. The reaction is again found strongly exothermic at −32.0 kcal mol−1.
3.3.2.4. Concluding remarks about the excellent epoxidation properties of the Mo–salophen system. It is therefore clear that this last reaction pathway again fails to account for the excellent styrene conversion observed in this system. At this stage, the literature is particularly instructive and gives some clues to explain the reactivity observed in this case. Indeed, Hey-Hawkins and coworkers showed that the complex [Salophen-{MoIVCl2}], strongly related to Mo0 because it contains a {Mo(IV)Cl2} coordinated to the same salophen ligand as in our case, was also easily oxidable (see SI Fig. S5).38 Thus, they described its quick evolution in the concomitant presence of H2O and O2. The evolution product thus described on this occasion contained a penta-coordinated cis-{MoVIO2Cl} center and a bidentate ligand resulting from the rearrangement of the Salophen ligand (Fig. 10).
 | ||
| Fig. 10 Details of the published crystal structure of the evolution product of the [Salophen-{MoIVCl2}] complex in the presence of H2O and O2 leading to a {MoVIO2Cl} complex according to Hey-Hawkins and coworkers.38 | ||
The most likely hypothesis in our case is therefore based on a behavior that is probably similar to that observed by Hey-Hawkins and co-workers. At this point, it is worth remembering that there is an induction time of one hour for both complexes Mo0 and Mo1, which is thus in favor of a different active species. Unfortunately, efforts to crystallize and/or spectroscopically identify a potential true active species in our case have so far proved unsuccessful. Although the exact structure of this true catalyst in the present study could not be determined, there is little doubt that this species contains a cis-{MoO2} centre. To support this hypothesis, it is noteworthy that complexes containing the {MoVIO2} entity associated with one or more Schiff bases have been regularly used in the past for epoxidation reactions with various oxidants (H2O2, TBHP, cumene hydroperoxide).35
4. Concluding remarks
The work presented here aimed to transform agricultural wastes into useful and valuable products, to enhance the concept of sustainability through the recycling of resources. We have therefore developed a complete set of studies, from the initial treatment of wastes from the peel of a citrus fruit native to Réunion Island to the chemical valuation of the major constituent of the essential oil: limonene. This work has made it possible to:- Quantify and identify 17 compounds in the essential oil and 21 in the hydrocarbon fraction by GC/MS-FID, which corresponds to an identification of ca. 97% and 99% respectively.
- Implement an aerobic epoxidation protocol for the hydrocarbon fraction of the essential oil, under the mildest possible conditions, in particular at room temperature.
- After a screening of the catalytic properties of a family of complexes based on Salophen ligands, demonstrate that the {MoIVO}-containing Mo0 and cis-{MoVIO2}-containing Mo1 precatalysts allow access to an effective active species for the selective formation of the mono- and di-epoxides of limonene (cumulative yield of these species greater than 96%).
- Finally, through a complete DFT study, rule out the Mo1 and Mo2 complexes (and a fortiori the Mo0 complex) as true catalyst for the epoxidation reaction in the case of the molybdenum-based system, alone, in the presence of a peracid or a peracetate. Although no other active species has yet been identified, it is clear that it results from the evolution of the Mo1 complex, probably in the form of a cis-{MoVIO2} complex.
This work is particularly important as it has enabled the establishment of a process for upgrading the essential oil from a citrus fruit endemic to Réunion Island, the ortanic tangor, through the aerobic oxidation of its main constituent, limonene. After this treatment under very mild conditions (room temperature), it is possible to obtain an oxidized hydrocarbon fraction of the essential oil that is particularly rich in oxygenated compounds, with recognized olfactive and food science properties.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been introduced as part of the supplementary information (SI). Supplementary information: a table about the origin and purity of reagents and solvents used. A Table indicating the exact composition of the essential oil and fraction H of ortanic Tangor epicarps. A Table indicating the composition and limonene conversion of reaction blanks in Mukaiyama epoxidation reactions. The synthesis of the catalysts. A paragraph indicating the method used for Identification of the volatile compounds, as well as the calculation of the composition of the mixtures after catalytic oxidation reactions. Figures and schemes indicating the main oxidation products of the limonene, the catalytic performances of [Salophen-{MoVIO2}] and the proposed mechanism for the evolution of the [Salophen-{MoIVCl2}] complex in the presence of H2O and O2. Finally, tables detailing structures and structural data for all intermediates and transition states in the DFT calculation performed all along this study. See DOI: https://doi.org/10.1039/d5nj03975d.Acknowledgements
This study benefited from the support of the FEDER project TERPENOX funded by the European Regional Development Funds GURDTI 2020-0969-0022947, (EU-Région Réunion-French State national counterpart) for the post-doctoral fellowship of Drs Keshika Mahadeo and Jean-Baptiste Harlé, as well as for the financial reward of Mrs Axelle Pagis. The authors would also like to acknowledge the Centre National de la Recherche Scientifique (CNRS), Université de la Réunion and Sorbonne Université.References
- Y. Ma, P. Li, Y. Li, S. J.-P. Willot, W. Zhang, D. Ribitsch, Y. H. Choi, R. Verpoorte, T. Zhang, F. Hollmann and Y. Wang, Natural Deep Eutectic Solvents as Multifunctional Media for the Valorization of Agricultural Wastes, ChemSusChem, 2019, 12, 1310–1315, DOI:10.1002/cssc.201900043.
- D. A. Zema, P. S. Calabrò, A. Folino, V. Tamburino, G. Zappia and S. M. Zimbone, Valorisation of Citrus processing waste: A review, Waste Manag., 2018, 80, 252–273, DOI:10.1016/j.wasman.2018.09.024.
- (a) M. Chutia, P. Deka Bhuyan, M. G. Pathak, T. C. Sarma and P. Boruah, Antifungal activity and chemical composition of Citrus reticulata Blanco essential oil against phytopathogens from North East India, LWT, Food Sci. Technol., 2009, 42, 777–780, DOI:10.1016/j.lwt.2008.09.015; (b) M. Chutia, J. J. Mahanta, R. C. Saikia, A. K. S. Baruah and T. C. Sarma, Influence of leaf blight disease on yield of oil and its constituents of java citronella and in-vitro control of the pathogen using essential oils, World J. Agri. Sci., 2006, 2, 319–321 Search PubMed.
- P. S. Calabrò, L. Pontoni, I. Porqueddu, R. Greco, F. Pirozzi and F. Malpei, Effect of the concentration of essential oil on orange peel waste biomethanization: Preliminary batch results, Waste Manag., 2016, 48, 440–447, DOI:10.1016/j.wasman.2015.10.032.
- S. Joy Ondet, La tenue des Fruits sur Tangor Ortanique, Bulletin d’Information des Producteurs, 2001, 3, 1–7 Search PubMed.
- (a) L. Goldenberg, Y. Yaniv, A. Doron-Faigenboim, N. Carmi and R. Porat, Diversity among mandarin varieties and natural sub-groups in aroma volatiles compositions, J. Sci. Food Agric., 2016, 96, 57–65, DOI:10.1002/jsfa.7191; (b) S. Simón-Grao, V. Gimeno, I. Simón, V. Lidón, M. Nieves, R. M. Balal, A. A. Carbonell-Barrachina, F. J. Manera, F. Hernández and F. García-Sánchez, Fruit quality characterization of eleven commercial mandarin cultivars in Spain, Sci. Hortic., 2014, 165, 274–280, DOI:10.1016/j.scienta.2013.11.022.
- M. Cassab Azevedo, S. Marques, M. Cristofani-Yaly, F. A. D. Azevedo, M. Bastianel, R. L. Carneiro and S. Bogusz Junior, Essential oil profiling of six new Citrus hybrids from Murcott tangor and Pera sweet orange, J. Essent. Oil Res., 2019, 31, 400–408, DOI:10.1080/10412905.2019.1596845.
- M. K. Park, J. Y. Cha, M.-C. Kang, H. Won Jang and Y.-S. Choi, The effects of different extraction methods on essential oils from orange and tangor: From the peel to the essential oil, Food Sci. Nutr., 2024, 12, 804–814, DOI:10.1002/fsn3.3785.
- E. Rozas-Munoz, J. P. Lepoittevin, R. M. Pujol and A. Giménez-Arnau, Allergic Contact Dermatitis to Plants: Understanding the Chemistry will Help our Diagnostic Approach, Actas Dermosifiliogr, 2012, 103, 456–477, DOI:10.1016/j.adengl.2012.07.006.
- J. Hanssens, D. Meneses, J. M. Saya and R. V. A. Orru, Terpenes and Terpenoids: How can we use them?, Eur. J. Org. Chem., 2025, e202401151, DOI:10.1002/ejoc.202401151.
- (a) R. A. Sheldon, Catalytic Oxidations in a Bio-Based Economy, Front. Chem., 2020, 8, 132, DOI:10.3389/fchem.2020.00132; (b) J. Zhong, J. Pérez-Ramírez and N. Yan, Biomass valorisation over polyoxometalate-based catalysts, Green Chem., 2021, 23, 18–36, 10.1039/d0gc03190a.
- A. Denicourt-Nowicki, M. Rauchdi, M. Ait Ali and A. Roucoux, Catalytic Oxidation Processes for the Upgrading of Terpenes: State-of-the-Art and Future Trends, Catalysts, 2019, 9, 893, DOI:10.3390/catal9110893.
- (a) O. Hauenstein, M. Reiter, S. Agarwal, B. Rieger and A. Greiner, Bio-based polycarbonate from limonene oxide and CO2 with high molecular weight, excellent thermal resistance, hardness and transparency, Green Chem., 2016, 18, 760–770, 10.1039/C5GC01694K; (b) O. Hauenstein, S. Agarwal and A. Greiner, Bio-based polycarbonate as synthetic toolbox, Nat. Commun., 2016, 7, 111862, DOI:10.1038/ncomms11862.
- Y. Mahamat Ahmat, S. Madadi, L. Charbonneau and S. Kaliaguine, Epoxidation of Terpenes, Catalysts, 2021, 11, 847, DOI:10.3390/catal11070847.
- (a) T. Yamada, T. Takai, O. Rhode and T. Mukaiyama, Highly efficient method for epoxidation of olefins with molecular oxygen and aldehydes catalyzed by nickel (II) complexes, Chem. Lett., 1991, 1–4, DOI:10.1246/bcsj.64.2109; (b) T. Yamada, T. Takai, O. Rhode and T. Mukaiyama, Direct Epoxidation of Olefins Catalyzed by Nickel (II) Complexes with Molecular Oxygen and Aldehydes, Bull. Chem. Soc. Jpn., 1991, 64, 2109–2117, DOI:10.1246/bcsj.64.2109.
- (a) P. Mastrorilli, C. F. Nobile, G. P. Suranna and L. Lopez, Aerobic oxidations of unsaturated substrates under Mukaiyama's conditions: The role of the metal and of the sacrificial aldehyde, Tetrahedron, 1995, 51, 7943–7950, DOI:10.1016/0040-4020(95)00410-A; (b) R. I. Kureshy, N. H. Khan, S. H. R. Abdi, A. K. Bhatt and P. Iyer, Synthesis, physicochemical studies and aerobic enantioselective epoxidation of non functionalized olefins catalyzed by new Co(II) chiral salen complexes, J. Mol. Cat. A Chem., 1997, 121, 25–31, DOI:10.1016/S1381-1169(96)00452-9; (c) K. Muniz-Fernandez, Manganese catalyzed epoxidation, in Transition metals for organic synthesis: building blocks and fine chemicals, ed. M. Beller and C. Bolm, Wiley-VCH, Weinheim, 2004, Vol. 2, pp. 344–356 Search PubMed; (d) Z. Li, S. Wu, H. Ding, D. Zheng, J. Hu, X. Wang, Q. Huo, J. Guan and Q. Kan, Immobilized Cu (II) and Co (II) salen complexes on graphene oxide and their catalytic activity for aerobic epoxidation of styrene, New J. Chem., 2013, 37, 1561–1568, 10.1039/C3NJ00099K; (e) Z. Li, S. Wu, D. Zheng, H. Ding, X. Wang, X. Yang, Q. Huo, J. Guan and Q. Kan, Enhanced Aerobic Epoxidation of Styrene with Copper(II), Cobalt(II), Iron(III), or Oxovanadium(IV) Salen Complexes Immobilized onto Carbon-Coated Fe3O4 Nanoparticles Hybridized with Graphene Sheets, ChemPlusChem, 2014, 79, 716–724, DOI:10.1002/cplu.201300424; (f) S. Rayati and F. Nejabat, The catalytic efficiency of Fe–porphyrins supported on multi-walled carbon nanotubes in the oxidation of olefins and sulfides with molecular oxygen, New J. Chem., 2017, 41, 7987–7991, 10.1039/C7NJ01530E; (g) K. P. Bryliakov, Catalytic Asymmetric Oxygenations with the Environmentally Benign Oxidants H2O2 and O2, Chem. Rev., 2017, 17, 11406–11459, DOI:10.1021/acs.chemrev.7b00167; (h) S. Madadi, J.-Y. Bergeron and S. Kaliaguine, Kinetic investigation of aerobic epoxidation of limonene over cobalt substituted mesoporous SBA-16, Catal. Sci. Technol., 2020, 11, 594–611, 10.1039/D0CY01700K.
- (a) T. Yamada, K. Imagawa, T. Nagata and T. Mukaiyama, Enantioselective Epoxidation of Unfunctionalized Olefins with Molecular Oxygen and Aldehyde Catalyzed by Optically Active Manganese(III) Complexes, Chem. Lett., 1992, 2231–2234, DOI:10.1246/cl.1992.2231; (b) T. Yamada, K. Imagawa, T. Nagata and T. Mukaiyama, Aerobic Enantioselective Epoxidation of Unfunctionalized Olefins Catalyzed by Optically Active Salen–Manganese(III) Complexes, Bull. Chem. Soc. Jpn., 1994, 67, 2248–2256, DOI:10.1246/bcsj.67.2248.
- A. Marvilliers, J.-B. Harlé, F. Launay, S. Blanchard, A. Gauvin-Bialecki, H. Gérard and R. Villanneau, Investigating the Mukaiyama-type epoxidation reaction of alkenes with Cr(III) salophen catalysts: a confrontation between experiments and DFT calculations, J. Catal., 2024, 434, 115526, DOI:10.1016/j.jcat.2024.115526.
- (a) A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5662, DOI:10.1063/1.464913; (b) C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–789, DOI:10.1103/PhysRevB.37.785.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 16, Revision C.01, Gaussian Inc, Wallingford, CT, 2016 Search PubMed.
- W. R. Wadt and P. J. Hay, Ab initio effective core potentials for molecular calculations – potentials for main group elements Na to Bi, J. Chem. Phys., 1985, 82, 284–298, DOI:10.1063/1.448800.
- (a) A. D. Kulkarni and D. G. Truhlar, Performance of density functional theory and Møller–Plesset second-order perturbation theory for structural parameters in complexes of Ru, J. Chem. Theory Comput., 2011, 7, 2325 CrossRef CAS; (b) R. Robidas and C. Y. Legault, How to obtain accurate results with molecular iodine and density functional theory?, Int J Quant. Chem, 2024, 124, e27277, DOI:10.1002/qua.27277; (c) A. Siiskonen and A. Priimagi, Benchmarking DFT methods with small basis sets for the calculation of halogen-bond strengths, J. Mol. Model., 2017, 23, 50, DOI:10.1007/s00894-017-3212-4.
- M. Bursch, J.-M. Mewes, A. Hansen and S. Grimme, Best-Practice DFT Protocols for Basic Molecular Computational Chemistry, Angew. Chem., Int. Ed., 2022, 61, e202205735, DOI:10.1002/anie.202205735.
- (a) F. Weigend and R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305, 10.1039/B508541A; (b) F. Weigend, Accurate Coulomb-fitting basis sets for H to Rn, Phys. Chem. Chem. Phys., 2006, 8, 1057–1065, 10.1039/B515623H.
- S. Grimme, S. Ehrlich and L. Goerigk, Effect of the damping function in dispersion corrected density functional theory, J. Comp. Chem., 2011, 32, 1456–1465, DOI:10.1002/jcc.21759.
- G. Luchini, J. V. Alegre-Requena, I. Funes-Ardoiz and R. S. Paton, GoodVibes: Automated Thermochemistry for Heterogeneous Computational Chemistry Data, F1000Research, 2020, 9, 291, DOI:10.12688/f1000research.22758.1.
- P. Agarwal, Z. Sebghatollahi, M. Kamal, A. Dhyani, A. Shrivastava, K. K. Singh, M. Sinha, N. Mahato, A. K. Mishra and K.-H. Baek, Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms, Antioxidants, 2022, 11, 2374, DOI:10.3390/antiox11122374.
- (a) J. Smadja, D. Strasberg, G. Legoff and A. Gauvin-Bialecki, Volatile Compounds from Melicope obscura, Chem. Biodiversity, 2010, 7, 467–475, DOI:10.1002/cbdv.200900098; (b) A. Gauvin-Bialecki and C. Marodon, Essential oil of Ayapana triplinervis from Reunion Island: A good natural source of thymohydroquinone dimethyl ether, Biochem. Syst. Ecol., 2009, 36, 853–858, DOI:10.1016/j.bse.2008.09.006.
- T. Luts and H. Papp, Novel Ways of Mn-Salen Complex immobilization on modified silica support and their catalytic activity in cyclooctene epoxidation, Kinet. Catal., 2007, 48, 176–182, DOI:10.1134/S0023158407010235.
- (a) M. Balas, L. K/Bidi, F. Launay and R. Villanneau, Chromium-salophen as a soluble or silica-supported co-catalyst for the fixation of CO2 onto styrene oxide at low temperatures, Front. Chem., 2021, 9, 765108, DOI:10.3389/fchem.2021.765108; (b) M. Balas, A. Mayoufi, R. Villanneau and F. Launay, F, Revisiting the Mukaiyama-type epoxidation for the conversion of styrene into styrene carbonate in the presence of O2 and CO2, React. Chem. Eng., 2023, 8, 345–354, 10.1039/D2RE00330A; (c) M. Balas, R. Villanneau and F. Launay, Design of simple salophen chromium complexes for a two-steps oxidative carboxylation of styrene with CO2 and O2, J. CO2 Utili., 2023, 102497, DOI:10.1016/j.jcou.2023.102497.
- (a) W. Nam, H. J. Kim, S. H. Kim, R. Y. N. Ho and J. S. Valentine, Metal Complex-Catalyzed Epoxidation of Olefins by Dioxygen with Co-Oxidation of Aldehydes. A Mechanistic Study, Inorg. Chem., 1996, 35, 1045–1049, DOI:10.1021/ic950782a; (b) M. L. Kuznetsov and J. Costa Pessoa, Epoxidation of olefins catalysed by vanadium–salan complexes: a theoretical mechanistic study, Dalton Trans., 2009, 5460–5468, 10.1039/B902424G; (c) N. Zwettler, M. E. Judmaier, L. Strohmeier, F. Belaj and N. C. Mösch-Zanetti, Oxygen activation and catalytic aerobic oxidation by Mo(IV)/(VI) complexes with functionalized iminophenolate ligands, Dalton Trans., 2016, 45, 14549–14560, 10.1039/c6dt01692h.
-
(a) T. G. Traylor, W. A. Lee and D. V. Stynes, Model compound studies related to peroxidases. Mechanisms of reactions of hemins with peracids, J. Am. Chem. Soc., 1984, 106, 755–764, DOI:10.1021/ja00315a050;
(b) M. J. Beck, E. Gopinath and T. C. Bruice, Influence of Nitrogen Base Ligation on the Rate of Reaction of [5,10,15,20-Tetrakis(2,6-dimethyl-3-sulfonatophenyl)-porphinato]iron(III) Hydrate with -BuOOH in Aqueous Solution, J. Am. Chem. Soc., 1993, 115, 21–29, DOI:10.1021/ja00054a004;
(c) P. Battioni, J. P. Renaud, J. F. Bartoli, M. Reina-Artiles, M. Fort and D. Mansuy, Monooxygenase-like Oxidation of Hydrocarbons by H2O2 Catalyzed by Manganese Porphyrins and Imidazole: Selection of the Best Catalytic System and Nature of the Active Oxygen Species, J. Am. Chem. Soc., 1988, 1110, 8462–8470, DOI:10.1021/ja00233a023;
(d) A. Robert, B. Loock, M. Momenteau and B. Meunier, Catalase Modeling with Metalloporphyrin Complexes Having an Oxygen Ligand in a Proximal Position. Comparison with Complexes Containing a Proximal Nitrogen, Inorg. Chem., 1991, 30, 706–711, DOI:10.1021/ic00004a021;
(e) N. Suzuki, T. Higuchi, Y. Urano, K. Kikuchi, H. Uekusa, Y. Ohashi, T. Uchida, T. Kitagawa and T. Nagano, Novel Iron Porphyrin−Alkanethiolate Complex with Intramolecular NH⋯S Hydrogen Bond:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Synthesis, Spectroscopy, and Reactivity, J. Am. Chem. Soc., 1999, 121, 11571–11572, DOI:10.1021/ja992511v.
Synthesis, Spectroscopy, and Reactivity, J. Am. Chem. Soc., 1999, 121, 11571–11572, DOI:10.1021/ja992511v. - K. P. Bryliakov, I. V. Khavrutskii, E. P. Talsi and O. A. Kholdeeva, EPR Detection and Characterization of High-Valent Manganese Complexes in MnIII(Salen) Catalyzed Aerobic Olefin Epoxidation, React. Kinet. Catal. Lett., 2000, 71, 183–191, DOI:10.1023/A:1010323308321.
- O. Makrygenni, L. Vanmairis, S. Taourit, F. Launay, A. Shum Cheong Sing, A. Proust, H. Gérard and R. Villanneau, Selective Formation of Epoxylimonene Catalyzed by Phosphonyl/Arsonyl Derivatives of Trivacant Polyoxotungstates at Low Temperature, Eur. J. Inorg. Chem., 2020, 605–612, DOI:10.1002/ejic.201901152.
- Here is a non-exhaustive selection of works representative of the use of cis-{Mo(VI)O2} complexes with Schiff base for epoxidation reactions: (a) D. D. Agarwal and S. Shrivastava, Synthesis of some cis-dioxomolybdenum complexes and their use in the epoxidation of olefins, Polyhedron, 1988, 7, 2569–2573, DOI:10.1016/S0277-5387(00)83875-9; (b) C. Bibal, J. C. Daran, S. Deroover and R. Poli, Ionic Schiff base dioxidomolybdenum(VI) complexes as catalysts in ionic liquid media for cyclooctene epoxidation, Polyhedron, 2010, 29, 639–647, DOI:10.1016/j.poly.2009.09.001; (c) A. Rezaeifard, I. Sheikhshoaie, N. Monadi and H. Stoeckli-Evans, Synthesis, Crystal Structure, and Catalytic Properties of Novel Dioxidomolybdenum(VI) Complexes with Tridentate Schiff Base Ligands in the Biomimetic and Highly Selective Oxygenation of Alkenes and Sulfides, Eur. J. Inorg. Chem., 2010, 799–806, DOI:10.1002/ejic.200900814; (d) Y. Li, X. Fu, B. Gong, X. Zou, X. Tu and J. Chen, Synthesis of novel immobilized tridentate Schiff base dioxomolybdenum(VI) complexes as efficient and reusable catalysts for epoxidation of unfunctionalized olefins, J. Mol. Catal. A:Chem., 2010, 322, 55–62, DOI:10.1016/j.molcata.2010.02.015; (e) M. E. Judmaier, C. Holzer, M. Volpe and N. C. Moesch-Zanetti, Molybdenum(VI) Dioxo Complexes Employing Schiff Base Ligands with an Intramolecular Donor for Highly Selective Olefin Epoxidation, Inorg. Chem., 2012, 51, 9956–9966, DOI:10.1021/ic301464w; (f) Y. Sui, D. Liu, R. Hu and X. Que, Chiral Schiff base ligated dioxomolybdenum (VI) complexes and their asymmetric catalytic properties in the epoxidation of styrene, J. Chem. Res., 2012, 3, 584–586, DOI:10.3184/174751912X13457309578; (g) W. Wang, T. Guerrero, S. R. Merecias, H. Garcia-Ortega, R. Santillan, J.-C. Daran, N. Farfan, D. Agustin and R. Poli, Substituent effects on solvent-free epoxidation catalyzed by dioxomolybdenum(VI) complexes supported by ONO Schiff base ligands, Inorga. Chim. Acta, 2015, 431, 176–183, DOI:10.1016/j.ica.2014.12.022; (h) M. Cindric, G. Pavlovic, R. Katava and D. Agustin, Towards a global greener process: from solvent-less synthesis of molybdenum(VI) ONO Schiff base complexes to catalyzed olefin epoxidation under organic-solvent-free condition, New J. Chem., 2017, 41, 594–602, 10.1039/C6NJ03174A; (i) M. Bagherzadeh, S. Ataie, H. Mahmoudi and J. Janczak, Synthesis, structure characterization and study of a new molybdenum Schiff base complex as an epoxidation catalyst with very high turnover numbers, Inorg. Chem. Commun., 2017, 84, 63–67, DOI:10.1016/j.inoche.2017.07.025; (j) M. Karman and G. Romanowski, Cis-dioxidomolybdenum(VI) complexes with chiral tetradentate Schiff bases: Synthesis, spectroscopic characterization and catalytic activity in sulfoxidation and epoxidation, Inorg. Chim. Acta, 2020, 511, 119832, DOI:10.1016/j.ica.2020.119832.
- S. N. Rao, N. Kathale, N. N. Rao and K. N. Munshi, Catalytic air oxidation of olefins using molybdenum dioxo complexes with dissymmetric tridentate O,N,S-donor Schiff base ligands derived from o-hydroxyacetophenone and S-benzyldithiocarbazate or S-methyldithiocarbazate, Inorg. Chim. Acta, 2007, 360, 4010–4016, DOI:10.1016/j.ica.2007.05.035.
- G. Dhimba, A. Muller and K. Lammertsma, A Computational Study of the MoO2(acac)2 Catalyzed Epoxidation of Ethylene with Hydrogen Peroxide and t-Butyl Hydroperoxide, ChemCatChem, 2023, 15, e202201031, DOI:10.1002/cctc.202201031.
- M. Schley, S. Fritzsche, P. Lönnecke and E. Hey-Hawkins, Soluble monometallic salen complexes derived from O-functionalised diamines as metalloligands for the synthesis of heterobimetallic complexes, Dalton Trans., 2010, 39, 4090–4106, 10.1039/b920706f.
Footnote |
| † Deceased on 29th June 2021. |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2026 |