Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Rationalizing polymorphism with local correlation-based methods: a case study of pnictogen molecular crystals†
Ahmet
Altun
 a,
Eduardo
Schiavo
a,
Eduardo
Schiavo
 a,
Michael
Mehring
a,
Michael
Mehring
 b,
Stephan
Schulz
b,
Stephan
Schulz
 c,
Giovanni
Bistoni
c,
Giovanni
Bistoni
 *d and
Alexander A.
Auer
*d and
Alexander A.
Auer
 *a
*a
aMax-Planck-Institut für Kohlenforschung, Kaiser Wilhelm Platz 1, D-45470 Mülheim an der Ruhr, Germany. E-mail: alexander.auer@kofo.mpg.de
bFakultät für Naturwissenschaften, Institut für Chemie, Professur Koordinationschemie, Technische Universität Chemnitz, Straße der Nationen 62, D-09107 Chemnitz, Germany
cInstitute of Inorganic Chemistry and Center for Nanointegration Duisburg-Essen (CENIDE), University of Duisburg-Essen, Universitätsstraße 5-7, D-45117 Essen, Germany
dDepartment of Chemistry, Biology and Biotechnology University of Perugia, Via Elce di Sotto, 8, 06123 Perugia, Italy. E-mail: giovanni.bistoni@unipg.it
First published on 31st October 2024
Abstract
A computational workflow is proposed to quantify and rationalize the relative stability of different structures of molecular crystals using cluster models and quantum chemical methods. The Hartree–Fock plus London Dispersion (HFLD) scheme is used to estimate the lattice energy of molecular crystals in various structural arrangements. The fragment-pairwise Local Energy Decomposition (fp-LED) scheme is then employed to quantify the key intermolecular interactions responsible for the relative stability of different crystal structures. The fp-LED scheme provides also in-depth chemical insights by decomposing each interaction into energy components such as dispersion, electrostatics, and exchange. Notably, this analysis requires only a single interaction energy computation per structure on a suitable cluster model. As a case study, two polymorphs of each of the following are considered: naphthyl-substituted dipnictanes (with As, Sb, and Bi as the pnictogen atom) and tris(thiophen-2-yl)bismuthane. The approach outlined offers high accuracy as well as valuable insights for developing design principles to engineer crystal structures with tailored properties, opening up new avenues in the study of molecular aggregates, potentially impacting diverse fields in materials science and beyond.
Introduction
Accurate quantification and analysis of intermolecular interactions in molecular crystals are crucial for comprehending the underlying effects that affect the formation of polymorphs, their stability, and phase transitions. This knowledge is essential for predicting and controlling crystal structures, and for designing materials with tailored properties. In pharmaceuticals, different polymorphs can significantly impact the solubility of a drug as well as its stability and bioavailability.1 Additionally, in fields like molecular electronics and drug design, the arrangement of molecules within a crystal structure influences the performance and properties of the material.2,3To develop structure–property relationships of molecular systems, typically their constituting isolated structural motifs are analyzed. However, conclusions drawn from chemical intuition-based structural analyses can be misleading.4–6 The interaction strength and nature of a given structural motif can vary across different chemical environments. For instance, what appears to be a classical “π–π interaction” might actually be dominated by dispersion forces.7 Similarly, pnictogen–π interactions primarily stem from dispersion, accompanied by a tunable donor–acceptor contribution.8–10 Hence, computing energetic contributions is essential for properly assessing the nature of the chemical interaction.11,12 Unfortunately, this task is far from trivial. In particular, for molecular organic crystals, nearly all experimentally-known polymorphs are separated by less than 2.5 kcal mol−1,6 necessitating the use of accurate quantum chemical methods. However, these methods become computationally very demanding, if not entirely unfeasible, as the system size grows. Additionally, beyond employing accurate methods, a scheme that accurately quantifies the different physical components of the interaction (electrostatic, exchange, dispersion, etc.) is also essential.
A feasible approach to gain insights into the strength of intermolecular interactions in a molecular crystal is to investigate isolated dimers while neglecting other environmental effects.13 For example, density functional theory (DFT) dimer analyses reproduce trends in the interactions between naphthyl-substituted dipnictanes, allowing to explain polymorphism.14,15 However, it should be kept in mind that for systems like closely packed crystal structures with noticeable charge transfer or polarization effects this approach may fail drastically. For example, for an accurate estimation of the lattice energy of a benzene crystal, it is necessary to compute terms in the many-body expansion (MBE) up to at least the four-body level.16
Efforts toward describing large molecular systems with increasing accuracy and efficiency constitute a growing active research field. MBE-based methods, such as the method of increments that truncate correlation energy,17 have significantly extended the limits of computational methods. When combined with embedding/periodic schemes and/or local correlation methods, these approaches enable investigation of not only small models but also a wide range of larger structures, including rare-gas crystals, covalent semiconductors, ionic insulators, diamond, metallic and half-metallic solids, fullerenes, graphite, and polymers.17–21 Especially periodic local versions of MP2 are a big step forward in accurately investigating large crystal systems.22–24 When periodic boundary conditions are implemented using the projector-augmented wave method for Coupled Cluster methods, even better accuracies are achieved.25 However, this approach still suffers from the steep scaling of Coupled Cluster methods with system size, restricting their use to solid models such as LiH, noble gases, boron nitride sheets, and the diamond and graphite phases of carbon.26–28 An alternative approach involves using local variants of CCSD(T), which achieve linear or low-order scaling with system size.29–31 In particular, the popular Domain-based Local Pair Natural Orbital CCSD(T) method [DLPNO-CCSD(T)]32–40 allows for the computation of energies and properties of systems as large as entire proteins.41
To rationalize molecular crystal structures in terms of the underlying intermolecular interactions, in addition to having an accurate and efficient quantum chemical method, a scheme that facilitates the interpretation of the energies is also required. Indeed, Symmetry Adapted Perturbation Theory (SAPT)42–48 and Energy Decomposition Analysis (EDA)49–51 methods breakdown interaction energies into physically meaningful components, such as electrostatics, induction, exchange-repulsion, and London dispersion. However, these schemes were primarily developed for the interaction of pairs of fragments. The extension of SAPT to multi-fragment systems marks an active era of research, distinguished by exciting developments.52–61
On the other hand, the Local Energy Decomposition (LED)62–65 scheme breaks down the energy of DLPNO-based local correlation methods into several physically meaningful terms for both closed–shell and open–shell molecular systems composed of any given number of fragments. The LED energy terms correlate well with the corresponding SAPT terms in the weak-interaction regime.66,67 As LED is instrumental not only in weak- but also in strong-interaction regimes, it has found applications in diverse fields.8–10,62–76 It is worth emphasizing here that, unlike other fragmentation methods that estimate the energy of the entire system by a series of computations on its isolated fragments and fragment combinations, LED requires only one supermolecular interaction energy computation. In addition, when used in conjunction with its recently introduced fragment pairwise extension fp-LED,75 this scheme provides a decomposition of the binding energy (e.g., lattice energies, protein–ligand interactions, etc.) into purely fragment-pairwise contributions.
Hence, LED constitutes a basis for the development of new methods that are accurate and efficient. This led to the semi-empirical “Natural Orbital Tied Constructed Hamiltonian” (NOTCH) method77 and the non-empirical “Hartree–Fock plus London Dispersion (HFLD)” method.78,79 HFLD is a variant of the DLPNO-CCSD(T) method for accurately and efficiently quantifying and analyzing non-covalent interaction (NCI) energies of both closed–hell and open–shell systems. It not only extends the applicability limits of its already efficient parent method significantly but also provides consistently an accuracy between those of CCSD and CCSD(T).78,79 Therefore, HFLD has found widespread applications in large-scale computations on protein,78 DNA,80 and crystal81 structures and on the solute–solvent interactions.79
In the present study, the HFLD method is used in conjunction with the fp-LED scheme to investigate the factors affecting the structure and stability of two forms of naphthyl-substituted dipnictane (Pn2Naph2, Pn = As, Sb, and Bi)14,15 crystal structures and of tris(thiophen-2-yl)bismuthane (Bi(C4H3S)3)82 crystal structures in the context of polymorphism. With this scheme, a quantification and analysis of all pairwise interactions of the central monomer with the neighboring monomers is provided within one single interaction energy calculation. It is worth noting here that some of these crystal structures were investigated previously using DFT-based dimer calculations which are much more laborious to carry out and yield less accurate results.15,82
In previous studies,14,15,82,83 we investigated London dispersion forces in various molecular systems containing group 15 elements as dispersion energy donors, with the aim of assessing their potential in crystal engineering and catalysis. Among the compounds studied, the Bi(C4H3S)3 is an excellent showcase to test our novel theoretical approach for the following reasons: (i) it shows enantiotropic phase transition near the room temperature (ca. 250 K) with low transition energy; (ii) the polymorphism relies on a subtle and reversible change from a dispersion interaction with the π-system and a sulfur atom of one ligand. Last but not least, both polymorphs were successfully characterized by X-ray single-crystal structure analysis.82 In the case of Pn2Naph2, the crystal structures showed different structural arrangements despite being isovalent.14,15,83
Thus, these two examples were chosen to illustrate how the proposed scheme can be used to rationalize rather small differences in the crystal structures of related compounds and how it can be used to identify and quantify the interactions that give rise to structural diversity observed in polymorphs.
Structures
Structure of dinaphthyl dipnictane clusters
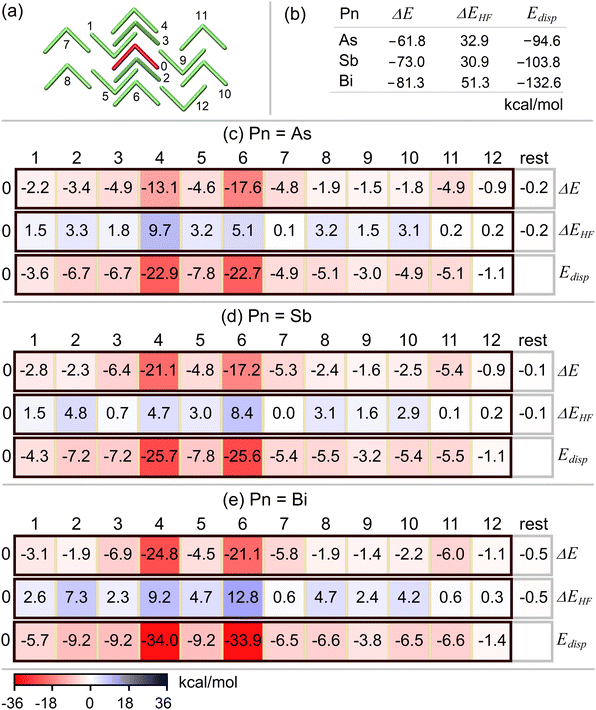
As2Naph2 and Bi2Naph2 crystal structures were taken from the literature.14,15 Despite being isovalent, the observed packing for these two systems differs. We label a molecular arrangement similar to that experimentally found for As2Naph2 as “A1”, while we label a molecular arrangement similar to the one observed for Bi2Naph2 as “A2”. In this study, we examine the key intermolecular interactions in Pn2Naph2 crystal structures with different pnictogen atoms (Pn = As, Sb, Bi) in both A1 and A2 forms to understand the origin of the experimentally observed structural variations.Initially, the experimentally determined crystal structures for As2Naph2 and Bi2Naph2 were optimized using periodic boundary conditions at the PBE-D3(BJ) level of theory. Then, 14-molecule (see Fig. 1) and 13-molecule (see Fig. 2) clusters were cut out from the resulting optimized A1 and A2 structures, respectively. These clusters include all monomers directly interacting with the central monomer. To assess the effect of the Pn atom on the stability of the crystal structure, the Pn sites were substituted with Sb and Bi in the A1 cluster (originally containing As), while they were substituted with As and Sb in the A2 cluster (originally containing Bi), without any further geometry optimization.
Structure of tris(thiophen-2-yl)bismuthane clusters
Bi(C4H3S)3 crystallizes in the space group of R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) at 269 K (labeled as B1 in this study) and in P
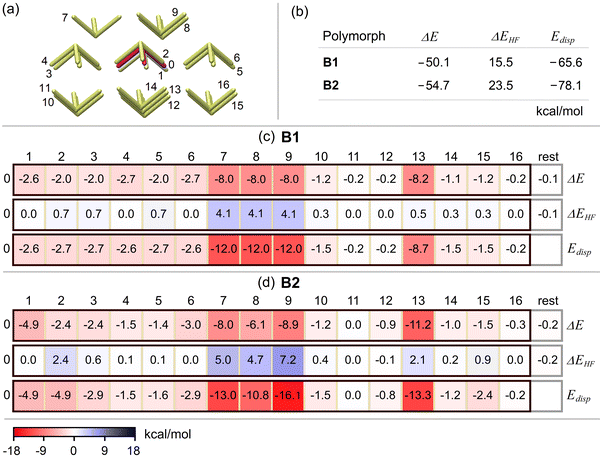
at 269 K (labeled as B1 in this study) and in P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) at 245 K (labeled as B2 in this study). These crystal structures show an enantiotropic phase transition at 250 K with a transition energy of 1.4 kJ mol−1.82 Although both clusters look very similar, B2 packs more closely than B1.82 17-Molecule clusters were cut out from the supramolecular arrangements as determined by single crystal X-ray structure analyses82 of both B1 and B2 polymorphs without any geometry optimization. The resulting clusters that include all monomers directly interacting with the central monomer are shown in Fig. 3 together with the labeling of their monomers.
at 245 K (labeled as B2 in this study). These crystal structures show an enantiotropic phase transition at 250 K with a transition energy of 1.4 kJ mol−1.82 Although both clusters look very similar, B2 packs more closely than B1.82 17-Molecule clusters were cut out from the supramolecular arrangements as determined by single crystal X-ray structure analyses82 of both B1 and B2 polymorphs without any geometry optimization. The resulting clusters that include all monomers directly interacting with the central monomer are shown in Fig. 3 together with the labeling of their monomers.
Computational scheme and details
Combined HFLD/fp-LED scheme for interaction energies
Labeling the energy of the entire cluster as E, the energy of the central monomer as E0, and the energy of the rest of the cluster (the “environment”) as Eenv, the interaction energy ΔE of the central monomer with the environment becomes within the supermolecular approach:| ΔE = E − (E0 + Eenv) | (1) |
It is important to note that, in the framework of MBE for computing the lattice energy, ΔE is expressed as the sum of the contributions from all isolated dimers, trimers, tetramers, and so on. When this expansion is truncated at the dimer level for highly symmetric molecular crystals, half of ΔE corresponds to the electronic component of the lattice energy.84 This estimate of lattice energy can be systematically refined by incorporating higher-body terms using well-established scaling protocols.84 In our approach, rather than summing the contributions from individual fragment combinations, we decompose ΔE computed using the supermolecular approach into pairwise contributions, which inherently include many-body effects. For the crystals considered in this study, by symmetry, ΔE represents twice the electronic component of the lattice energy.
In eqn (1), the entire system and the environment are both composed of multiple monomers, and thus require performing LED analyses on top of their standard electronic structure calculations to obtain pairwise contributions. Using the notation introduced in ref. 75 for the standard LED decomposition of interaction energies between subsystems of many fragments, we obtain:
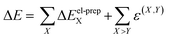 | (2) |
The summations run over the individual fragments (the monomers) labeled as X, Y = 0, 1, 2,…, (N − 1), where N is the number of fragments in the cluster. The electronic preparation term ΔEel-prepX corresponds to the change in the energy of monomer X upon the interaction between the central monomer and the environment. It is shown by the diagonal elements (i.e., X = Y) enclosed by violet boxes in Fig. 4(a).
 | ||
| Fig. 4 A1 form of As2Naph2: HFLD/LED interaction energy map of the central monomer 0 with those at its environment (1–13), using (a) standard LED and (b) fp-LED. For simplicity, interaction terms with absolute values smaller than 0.2 kcal mol−1 are not annotated on the heat maps. See ESI† for fully annotated heat maps. | ||
The interaction between fragment X and fragment Y (where X ≠ Y) is denoted as ε(X,Y), or equivalently as ε(Y,X). To avoid double counting of ε(X,Y) and ε(Y,X), the sum over X and Y was constrained in the second term of eqn (2) with the X > Y condition. In the heat maps, we place X labels on the horizontal axis, while placing Y labels on the vertical axis. This choice corresponds to upper-diagonal heat maps as given in Fig. 4.
The pairwise ε(X,Y) terms (where X ≠ Y) are of two kinds: genuine interaction terms between the central monomer and each fragment in the environment, ε(X,0) (elements enclosed by black boxes in the first row of Fig. 4(a)), and “inductive” interaction terms, ε(X,Y≠0). The inductive terms (elements enclosed by green boxes in Fig. 4(a)) represent the changes in fragment-pairwise interaction energies within the environment that occur upon the formation of the entire cluster. These terms arise from the cooperativity effects of various noncovalent interactions and thus vanish in the absence of many-body effects.
In eqn (2), ΔEel-prepX incorporates the dominant repulsive contributions for the interaction of fragment X with all the remaining fragments in the system. The fp-LED75 scheme allows to disentangle the contributions from pairs of fragments within ΔEel-prepX terms as
| ΔEel-prepXY = ω(X,Y)XΔEel-prepX + ω(X,Y)YΔEel-prepY | (3) |
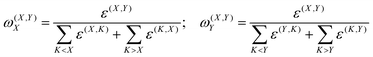 | (4) |
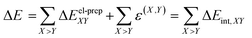 | (5) |
The standard LED map based on eqn (2) contains positive and negative values of large magnitude (Fig. 4(a)). In contrast, the fp-LED map based on eqn (5) (Fig. 4(b)) directly provides the strength of all interactions of the fragment pairs within the structure, with interaction energy values comparable to those of the isolated fragment pairs. However, unlike the isolated dimer computations, these values also include the effect of the chemical environment, thus provide an exact decomposition of the interaction energy into dimer contributions.
Eqn (3) was formulated based on the perfect correlation between ε(X,Y) and the sum of ΔEel-prepX and ΔEel-prepY for noncovalently interacting isolated X⋯Y dimers.75 In other words, the strength of pairwise interaction is proportional to the increase in the energy of the fragments upon their interaction. This proportionality suggests that, by design, the ε(X,Y) and ΔEint,XY terms in standard and fp-LED typically follow the same trends. This is also reflected in the LED data for all clusters considered in this study (see the ESI†). Hence, despite the fact that interpreting standard LED results is less intuitive due to very large ε(X,Y) and ΔEel-prepX values, which often have opposite signs, standard LED is still useful in trend studies.
In the HFLD scheme,78–80 the ΔEint,XY terms contain electronic preparation, electrostatic, and exchange components computed at the HF level as well as the dispersion component from Coupled Cluster. In this study, for the sake of simplicity, ΔEint,XY terms are simply decomposed into non-dispersive and dispersive components, which we denote as ΔEHF,XY and Edisp,XY. Decomposed nondispersive pairwise terms (electronic preparation, electrostatic, and exchange) are all provided in the ESI.† In the HFLD scheme, inductive Edisp,XY dispersion terms (where Y ≠ 0 in the present study) are neglected, as they are practically zero for noncovalent interactions.80 This significantly enhances the efficiency of the method without sacrificing accuracy. Therefore, only genuine Edisp,XY dispersion terms (where Y = 0 in the present study) are computed within the HFLD scheme.
It is also worth mentioning that the “inductive” ε(X,Y≠0) terms, depicted in green in Fig. 4(b), are very small, summing to less than 1 kcal mol−1 in magnitude. Therefore, in the following, we will only display the first row of the LED maps, plus a cell labeled “rest” at the end of the maps that corresponds to the sum of all of these cooperative ε(X,Y≠0) terms. Full LED maps, as shown in Fig. 4, are provided in the ESI.†
Finally, it is important to emphasize that the pairwise terms just discussed are not derived from isolated dimer calculations but are obtained through a single supermolecular interaction energy calculation, and thus account for many-body effects. The supermolecular approach involves electronic structure calculations only on the central molecule and two molecular clusters: the environment surrounding the central molecule and the entire system. Note that the HFLD scheme78,79 for noncovalent interactions already efficiently handles molecular clusters of ∼1000 atoms with triple-ζ quality basis sets (∼14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 basis functions).74,75,80
000 basis functions).74,75,80
Computational details
The experimentally determined crystal structures for As2Naph214 and Bi2Naph215 were initially optimized in the solid-state with periodic boundary conditions using the PWSCF (v.6.7) module of the QuantumESPRESSO package.85,86 The PBE87 functional was used by incorporating the D388 dispersion correction with Becke–Johnson damping (BJ).89 Ultrasoft Vanderbilt-type pseudo potentials were utilized.90 A kinetic-energy cutoff of 40 Ry and a 7 × 4 × 2 Monkhorst–Pack k-point grid91 was employed.All the multi-fragment HFLD/LED computations were performed with the ORCA program package (version 5.0)92–95 at the cluster geometries described in the Structures section. The RIJCOSX approach96,97 was used for the reference HF part. The def2-TZVP(-f) basis set was used together with its matching auxiliary basis sets in the HF and correlation parts.98 NormalPNO* settings78 of DLPNO Coupled Cluster methods and default frozen core settings99 were applied for the correlation energy calculations. The Foster-Boys scheme100 was used for localizing both occupied orbitals and PNOs.
Since in the HFLD scheme the electronic preparation is computed at the HF level, the ω(X,Y)X and ω(X,Y)Y terms in eqn (3) were also computed at the HF level (neglecting the dispersion contribution), as suggested in the theory paper of fp-LED.75 LED Analysis Wizard (LEDAW) program package101 was used in computing interaction energy matrices from ORCA outputs and plotting the corresponding heat maps.
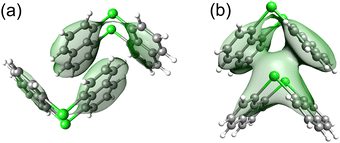
Dispersion Interaction Density (DID)64,102 plots were computed to assess the spatial origin of the dispersion interaction between the central monomer and environmental monomers by using the above-described HFLD/LED settings. For the ease of visualization, DID plots are shown herein for the monomer pairs that most significantly contribute to the stability of the crystal structures separately rather than on the entire cluster.
Results and discussion
In this section, on two forms of Pn2Naph2 (A1 and A2) and Bi(C4H3S)3 (B1 and B2) crystals we assess (i) the energetic contribution of each dimeric motif on the entire cluster formation, (ii) the effect of the type of Pn atom on the individual noncovalent interactions and on the overall stability of a given cluster; (iii) the individual interactions responsible for the difference in the binding energies, and thus, lattice energies of the two forms.Dinaphthyl dipnictane clusters
Pn2Naph2 molecules form crystal structures with quite diverse intermolecular interaction patterns.14,15,83 As2Naph214 (see the A1 form in Fig. 1) exhibits an intermolecular network of π⋯π, CH⋯π, As⋯As interactions,15 whereas Bi2Naph2 (see the A2 form in Fig. 2) features two Bi⋯π contacts per Bi atom rather than π⋯π contacts in the solid state.15 Through a very large series of DFT-D dimer computations,15 the A2 model with Pn = Bi was shown to benefit from the enhanced Pn⋯π intermolecular dispersion interaction. In order to demonstrate the advantage of combined HFLD/fp-LED scheme that allows quantifying and rationalizing all intermolecular interactions in crystal structures with one supermolecular interaction energy calculation, in this study we revisit these A1 and A2 models together with a series of hypothetical structures obtained by just replacing the Pn atom by their lighter or heavier homologs (see the Structures section).The HFLD/fp-LED pairwise interaction energy map and its nondispersive HF and dispersive interaction energy sub-maps are shown for the A1 form (Pn = As) and its Sb- and Bi-substituted variants in Fig. 5 and for the A2 form (Pn = Bi) and its As- and Sb-substituted variants in Fig. 6. The total HFLD interaction energy ΔE as well as its total nondispersive HF interaction energy ΔEHF and the total dispersion energy Edisp components are provided in panel b of these figures while their pairwise components are provided in panel c–e.
For the A1 form, with increasing size of the Pn atom, overall repulsive non-dispersive and overall attractive dispersion energies both increase roughly at the same rate. This trend is due to the π⋯π interaction between one of the naphthyl moieties of the central monomer 0 and monomer 8 (and, to a smaller extent, monomer 6). The other pairwise interaction energies remain roughly the same with the change of the Pn atom. Consequently, the sum of all interaction energies does not exhibit a strong dependence on the type of the Pn atom for the A1 form.
The primary stabilizing pairwise interaction in the A1 form emerges from the π⋯π (appearing once in the pair) and CH⋯π (appearing once in the pair) interactions between one of the naphthyl moieties of the central monomer 0 and the monomer 4, with a secondary contribution from the same type of interaction between central monomer 0 and monomer 6 (see Fig. 7(a) for the structural motif of these monomer pairs), consistent with previous DFT-D results.15 These interactions are mainly of dispersion nature and the associated DID plot is shown in Fig. 7(a). The combined contribution of these two pairs constitutes ca. 50% of the total interaction energy. Although individual contributions from other pairs are much smaller, their collective effect is significant, accounting for the remaining half of the crystal structure's stability.
Transition from A1 to A2 involves a decrease in both the nondispersive HF and dispersive interaction components, with exception of the dispersion component in the case that Pn = Bi. Therefore, the enhanced stability of the A2 form with Pn = Bi compared with A1 stems from a dual effect: a reduction in the nondispersive component (ca. 50%) and an increase in the dispersive interaction (ca. 50%).
Unlike A1, the overall stability of A2 significantly increases with the increase in the size of the Pn atom, governed primarily by the increase in the dispersion interaction. This is actually consistent with the larger dispersion energy donation capacity of the larger Pn atoms.
The interaction energy of A2 is only marginally larger than that of A1 when considering Pn = As, yet it demonstrates significantly greater stability with Pn = Sb and Bi. This implies the potential for synthesizing the A2 form also with Pn = As and Sb under diverse experimental conditions. Notably, in the case of Pn = Bi, consistent with the experimental isolation of the Bi structure in the A2 form, the stability of the A2 crystal structure surpasses that of A1 by approximately 20 kcal mol−1.
The pairs that align precisely on top of each other, i.e., the 0–4 and 0–6 monomer pairs, provide the largest pairwise contribution to the stability of the A2 form (see Fig. 7(b) for their structural motif), consistent with the results of the previous DFT-D study.15 These pairs exhibit Pn⋯π interactions between the Pn atom of one monomer and both naphthyl moieties of the other monomer. This interaction is of mainly dispersive character and increases with the size of the Pn atom (see Fig. 7(b) for the associated DID plot). These two fragment pairs collectively contribute 50%, 52%, and 57% to the total interaction energy with Pn = As, Sb, and Bi, respectively. While the contributions from other individual fragment pairs are much smaller, their cumulative impact becomes notably significant.
As a closing remark of this section, it is worth emphasizing that the formation of both polymorphs, A1 and A2, is primarily driven by dispersive forces irrespective of the type of Pn atom as their nondispersive HF contributions are repulsive. Hence, using the approach described above, it is possible to quantify the relative stability of a given crystal structure, to identify individual structure determining interactions and explain their contribution using the concepts of intermolecular interactions.
Tris(thiophen-2-yl)bismuthane clusters
Two interconvertible polymorphs of Bi(C4H3S)3 were previously crystallized at distinct temperatures.82 We label here the high temperature polymorph as B1 while labeling the low temperature polymorph as B2 (see Fig. 3 for their structures and monomer labeling).The polymorphs show very small but obviously decisive structural changes in the arrangement of the molecules in the unit cell. Based on our previous DFT-D and MP2 studies on small model systems,82 the Bi⋯π and Bi⋯S interactions are primarily dispersive in nature. Therefore, the formation of the Bi(C4H3S)3 structure should also be governed primarily by dispersion forces. Furthermore, gas-phase geometry optimizations on the Bi(C4H3S)3⋯(C4H3S)n system (n = 1 and 3) yielded only one minimum.82 This suggests that the polymorphism of the Bi(C4H3S)3 system stems from the interplay between different intermolecular interactions, which induce several structural rearrangements and give rise to polymorphism.
In order to shed light into the origin of the polymorphism, we performed HFLD/fp-LED computations on the large Bi(C4H3S)3 clusters of B1 and B2 (see Fig. 3). The resulting HFLD/fp-LED pairwise interaction energy map and its nondispersive HF and dispersive interaction energy sub-maps are shown in Fig. 8 together with the total HFLD interaction energy ΔE, its total nondispersive HF interaction energy ΔEHF, and total dispersion energy Edisp components.
The B2 cluster is 4.6 kcal mol−1 more stable than the B1 cluster. The overall nondispersive HF interaction components of both B1 and B2 are repulsive and 3–4 times smaller in absolute value than their overall attractive dispersion components. Therefore, the formation of these structures is largely driven by dispersion interaction between Bi(C4H3S)3 molecules, consistent with the results on small model systems.82
The largest contribution to the stability of these polymorphs arises from the interaction of the central monomer 0 with monomers 13, 9, 7, 8, and 1, listed in descending interaction strength on B2. These pairs collectively account for 69% and 72% of the total interaction energy of B1 and B2. Although the cumulative impact of the remaining 11 pairs to the stability of the clusters is still significant, their individual contributions do not vary much between B1 and B2. Therefore, in the following we focus on the most strongly interacting five monomer pairs listed above.
To assess the relation between the structure and the stability of the monomer pairs, in Fig. 9(a) we show the superimposed structures (with respect to the central monomer) of the most stable five monomer pairs in the B1 and B2 polymorphs. As the stabilizing component of the interaction is largely intermolecular dispersion, we also provide the DID plots of these pairs on the B1 and B1 clusters in Fig. 9(b) and (c), respectively.
Transitioning from B1 to B2, the distance between the monomers of the strongest pair (0–13) is significantly shortened, including the intermolecular C4H3S⋯C4H3S ring and the Bi⋯Bi pnictogen separations. This introduces a slight increase in the repulsive component of the interaction while notably enhancing dispersive CH⋯π interactions between the rings (see Fig. 8 for the energy contributions and Fig. 9 for the associated DID plots). These structural modifications account for more than half (3.0 kcal mol−1) of the larger stability of B2 compared to B1.
Pairs 0–7, 0–8, and 0–9, involving intermolecular π⋯π, Bi⋯π, Bi⋯S, π⋯S, S⋯S interactions (see Fig. 9), experience noticeably large repulsion, which accounts for 79% and 72% (see Fig. 8) of the overall repulsive contribution to the interaction energy of B1 and B2, respectively. Despite this, the magnitude of the dispersion interaction in these pairs is akin to that computed for the strongest pair (0–13) of B2. Consequently, due to the counteracting effects of increased and decreased attractive and repulsive components, these pairs demonstrate individual interaction strengths similar to those in the 0–13 pair of B1 discussed above. While transitioning from B1 to B2, as the structural variations in these pairs are relatively minor, they do not noticeably contribute to the relative stability of B1 and B2, despite some variations in their individual interaction strengths and their components.
The interaction strength of the 0–1 pair is significantly smaller than the other four pairs discussed above. During the transition from B1 to B2, the intermolecular distance between its monomers is noticeably shortened. This enhances intermolecular dispersive CH⋯π and S⋯S interactions (see Fig. 9) between the monomers by 2.3 kcal mol−1 (see Fig. 8), accounting for the other half of the larger stability of B2.
To sum up, a combined HFLD/fp-LED analysis allows to quantify and analyze the energetic contributions to the stability of different polymorphs in Bi(C4H3S)3 crystal structures. The analysis demonstrates that the larger stability of the polymorph B2 over B1 arises from the increase of the (predominantly dispersion) interaction of central monomer 0 with monomers 13 and 1, facilitated by their relatively shorter intermolecular contacts.
Conclusions
In this study, HFLD/fp-LED computations were conducted on large Pn2Naph2 and Bi(C4H3S)3 cluster models of crystal structures to estimate the binding energy of the central monomer in various possible polymorphs, which are related to lattice energies. These computations provide insights into the relative stability of the polymorphs. The proposed protocol provides an exact decomposition of approximated binding energies into contributions from pairs of monomers within the solid. This computational approach enabled us to rationalize the relative stability and polymorphism of the structures based on fundamental energetic considerations, rather than relying solely on the analysis of their geometry parameters, which are not necessarily correlated with stability.For the Pn2Naph2 polymorphs (A1 and A2), our present HFLD/fp-LED binding energy computations revealed that their most stable dimer structure accounts for half of the total stability of each polymorph, consistent with our earlier DFT-D results on isolated dimers.15 While the individual contributions of other pairs are much smaller, their cumulative effect accounts for the remaining stability. The enhanced stability of the A2 polymorph with Pn = Bi compared to the A1 polymorph is consistent with the experimental isolation of only A2 with Bi.15 This stabilization arises equally from a reduction in the nondispersive component and from an increase in the dispersive component. In the case of Pn = As, although only A1 has been experimentally isolated,14A1 and A2 are computed to be nearly isoenergetic. This suggests the potential for preparing A2 under different experimental conditions.
Having two polymorphs of Bi(C4H3S)3 (B1 and B2) at hand, we were able to gather insights into an enantiotropic phase transition having a low transition barrier. Our HFLD/fp-LED approach allowed us, for the first time, to decipher the subtle interplay of different intermolecular interactions on the stability of each polymorph and their relative strengths. In particular, we demonstrated that subtle changes in the relative orientation of two dimers in these polymorphs have a strong influence on their dispersion interactions, and are therefore almost entirely responsible for their relative stability.
In conclusion, the combined HFLD/fp-LED scheme is a powerful tool for analyzing structural motifs in molecular crystals by decomposing interaction energies, and thus lattice energies, into pairwise intermolecular interaction components. This approach offers valuable insights into the key factors that influence the formation and stability of molecular crystals and their polymorphs. Understanding how molecules aggregate into diverse structures is a significant advancement, especially considering that experiments often yield unexpected structures with slight variations in crystallization conditions. By rationalizing the effects of subtle changes in geometry and electronic structure on crystal stability through the HFLD/fp-LED scheme, material design strategies can be significantly enhanced. Given the pivotal role of crystal structure in various fields, from molecular electronics to drug design, this knowledge holds great potential for driving innovation across various industries.
Finally, it is worth noting that ongoing efforts in our theory lab to extend the LED scheme to covalent interactions are expected to significantly broaden the applicability of this approach to a wider range of molecular systems and interactions.
Author contributions
E. S. and A. A. carried out all the calculations. A. A. analyzed the results and wrote the original draft. A. A. A. and G. B. devised and coordinated the project, and prepared the final version of the manuscript with input from all authors. S. S. and M. M. were involved in the project design and writing and proof reading of the manuscript.Data availability
The XYZ coordinates and energetic data of all clusters, including heat maps, are available in the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors gratefully acknowledge the Priority Program “Control of Dispersion Interactions in Molecular Chemistry” (SPP 1807) of the Deutsche Forschungsgemeinschaft for financial support. A. A. gratefully acknowledges the Research Unit Program “Bioinspired Oxidation Catalysis with Iron Complexes” (FOR 5215) of the Deutsche Forschungsgemeinschaft for financial support. G. B. acknowledge financial support under the National Recovery and Resilience Plan (NRRP), Mission 4, Component 2, Investment 1.1, Call for tender no. 104 published on 2.2.2022 by the Italian Ministry of University and Research (MUR), funded by the European Union – NextGenerationEU – Project Title 2022WKTH9E – Modelling and design of organic conjugated redox materials for energy-saving applications: a bottom-up strategy. – CUP J53D23008810006 – Grant Assignment Decree No. 1064 adopted on 18.07.2022 by the Italian Ministry of University and Research (MUR). Open Access funding provided by the Max Planck Society.References
- J. Bernstein, Polymorphism in Molecular Crystals, Oxford University Press, Oxford, 2nd edn, 2020 Search PubMed.
- H. Chung and Y. Diao, J. Mater. Chem. C, 2016, 4, 3915–3933 RSC.
- M. Li, A. H. Balawi, P. J. Leenaers, L. Ning, G. H. L. Heintges, T. Marszalek, W. Pisula, M. M. Wienk, S. C. J. Meskers, Y. Yi, F. Laquai and R. A. J. Janssen, Nat. Commun., 2019, 10, 1–11 CrossRef PubMed.
- B. K. Saha, N. K. Nath and R. Thakuria, Chem. Rec., 2023, 23, e202200173 CrossRef PubMed.
- S. Varughese, J. Mater. Chem. C, 2014, 2, 3499–3516 RSC.
- G. J. O. Beran, Chem. Sci., 2023, 14, 13290–13312 RSC.
- S. Grimme, Angew. Chem., Int. Ed., 2008, 47, 3430–3434 CrossRef.
- E. Schiavo, K. Bhattacharyya, M. Mehring and A. A. Auer, Chem. – Eur. J., 2021, 27, 14520–14526 CrossRef.
- M. Krasowska, W. B. Schneider, M. Mehring and A. A. Auer, Chem. – Eur. J., 2018, 24, 10238–10245 CrossRef PubMed.
- M. Krasowska, A. M. Fritzsche, M. Mehring and A. A. Auer, ChemPhysChem, 2019, 20, 2539–2552 CrossRef PubMed.
- S. Rindfleisch, M. Krull, J. Uranga, T. Schmidt, F. Rabe von Pappenheim, L. L. Kirck, A. Balouri, T. Schneider, A. Chari, R. Kluger, G. Bourenkov, U. Diederichsen, R. A. Mata and K. Tittmann, Nat. Catal., 2022, 5, 332–341 CrossRef.
- X. Aniban, B. Hartwig, A. Wuttke and R. A. Mata, Phys. Chem. Chem. Phys., 2021, 23, 12093–12104 RSC.
- A. M. Preda, M. Krasowska, L. Wrobel, P. Kitschke, P. C. Andrews, J. G. MacLellan, L. Mertens, M. Korb, T. Rüffer, H. Lang, A. A. Auer and M. Mehring, Beilstein J. Org. Chem., 2018, 14, 2125–2145 CrossRef PubMed.
- K. Dzialkowski, A. Gehlhaar, C. Wölper, A. A. Auer and S. Schulz, Organometallics, 2019, 38, 2927–2942 CrossRef.
- A. Gehlhaar, E. Schiavo, C. Wölper, Y. Schulte, A. A. Auer and S. Schulz, Dalton Trans., 2022, 51, 5016–5023 RSC.
- J. Yang, W. Hu, D. Usvyat, D. Matthews, M. Schütz and G. K. L. Chan, Science, 2014, 345, 640–643 CrossRef.
- H. Stoll, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6700 CrossRef PubMed.
- E. Voloshina, N. Gaston and B. Paulus, J. Chem. Phys., 2007, 126, 134115 CrossRef.
- B. Paulus, Phys. Rep., 2006, 428, 1–52 CrossRef CAS.
- U. Birkenheuer, P. Fulde and H. Stoll, Theor. Chem. Acc., 2006, 116, 398–403 Search PubMed.
- H. Stoll, B. Paulus and P. Fulde, J. Chem. Phys., 2005, 123, 144108 CrossRef.
- L. Maschio, B. Civalleri, P. Ugliengo and A. Gavezzotti, J. Phys. Chem. A, 2011, 115, 11179–11186 CrossRef CAS PubMed.
- L. Maschio, D. Usvyat and B. Civalleri, CrystEngComm, 2010, 12, 2429–2435 RSC.
- C. Pisani, M. Schütz, S. Casassa, D. Usvyat, L. Maschio, M. Lorenz and A. Erba, Phys. Chem. Chem. Phys., 2012, 14, 7615–7628 RSC.
- I. Y. Zhang and A. Grüneis, Front. Mater., 2019, 6, 432749 Search PubMed.
- S. J. Nolan, M. J. Gillan, D. Alfè, N. L. Allan and F. R. Manby, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 80, 165109 CrossRef.
- T. Gruber, K. Liao, T. Tsatsoulis, F. Hummel and A. Grüneis, Phys. Rev. X, 2018, 8, 021043 Search PubMed.
- A. Grüneis, G. H. Booth, M. Marsman, J. Spencer, A. Alavi and G. Kresse, J. Chem. Theory Comput., 2011, 7, 2780–2785 CrossRef PubMed.
- Linear-Scaling Techniques in Computational Chemistry and Physics: Methods and Applications, ed. R. Zaleśny, M. G. Papadopoulos, P. G. Mezey and J. Leszczynski, Springer, 2011 Search PubMed.
- Q. Ma, M. Schwilk, C. Köppl and H.-J. Werner, J. Chem. Theory Comput., 2017, 13, 4871–4896 CrossRef PubMed.
- M. Nagy and P. R. Kállay, J. Chem. Theory Comput., 2019, 15, 5275–5298 CrossRef PubMed.
- F. Neese, A. Hansen and D. G. Liakos, J. Chem. Phys., 2009, 131, 064103 CrossRef PubMed.
- F. Neese, A. Hansen, F. Wennmohs and S. Grimme, Acc. Chem. Res., 2009, 42, 641–648 CrossRef.
- F. Neese, F. Wennmohs and A. Hansen, J. Chem. Phys., 2009, 130, 114108 CrossRef.
- A. Hansen, D. G. Liakos and F. Neese, J. Chem. Phys., 2011, 135, 214102 CrossRef.
- L. M. J. Huntington, A. Hansen, F. Neese and M. Nooijen, J. Chem. Phys., 2012, 136, 064101 CrossRef PubMed.
- C. Riplinger and F. Neese, J. Chem. Phys., 2013, 138, 034106 CrossRef.
- D. G. Liakos, A. Hansen and F. Neese, J. Chem. Theory Comput., 2011, 7, 76–87 CrossRef PubMed.
- C. Riplinger, P. Pinski, U. Becker, E. F. Valeev and F. Neese, J. Chem. Phys., 2016, 144, 024109 CrossRef.
- D. G. Liakos, M. Sparta, M. K. Kesharwani, J. M. L. Martin and F. Neese, J. Chem. Theory Comput., 2015, 11, 1525–1539 CrossRef PubMed.
- C. Riplinger, B. Sandhoefer, A. Hansen and F. Neese, J. Chem. Phys., 2013, 139, 134101 CrossRef PubMed.
- B. Jeziorski, R. Moszynski and K. Szalewicz, Chem. Rev., 1994, 94, 1887–1930 CrossRef.
- R. Podeszwa, R. Bukowski and K. Szalewicz, J. Chem. Theory Comput., 2006, 2, 400–412 CrossRef PubMed.
- A. Heßelmann, G. Jansen and M. Schütz, J. Chem. Phys., 2005, 122, 014103 CrossRef.
- R. Bukowski, R. Podeszwa and K. Szalewicz, Chem. Phys. Lett., 2005, 414, 111–116 CrossRef.
- A. Heßelmann, J. Chem. Theory Comput., 2018, 14, 1943–1959 CrossRef PubMed.
- B. Jeziorski, M. van Hemert and B. Jeziorski, Mol. Phys., 1976, 31, 713–729 CrossRef.
- T. M. Parker, L. A. Burns, R. M. Parrish, A. G. Ryno and C. D. Sherrill, J. Chem. Phys., 2014, 140, 094106 CrossRef.
- E. Pastorczak and C. Corminboeuf, J. Chem. Phys., 2017, 146, 120901 CrossRef.
- M. von Hopffgarten and G. Frenking, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 43–62 Search PubMed.
- M. J. S. Phipps, T. Fox, C. S. Tautermann and C.-K. Skylaris, Chem. Soc. Rev., 2015, 44, 3177–3211 RSC.
- R. M. Parrish, K. C. Thompson and T. J. Martínez, J. Chem. Theory Comput., 2018, 14, 1737–1753 CrossRef.
- R. M. Parrish, T. M. Parker and C. David Sherrill, J. Chem. Theory Comput., 2014, 10, 4417–4431 CrossRef PubMed.
- R. M. Parrish and C. D. Sherrill, J. Chem. Phys., 2014, 141, 44115 CrossRef PubMed.
- A. Heßelmann, Beilstein J. Org. Chem., 2018, 14, 979–991 CrossRef.
- J. Garcia, R. Podeszwa and K. Szalewicz, J. Chem. Phys., 2020, 152, 184109 CrossRef PubMed.
- D. Luu, C. Corminboeuf and K. Patkowski, J. Chem. Theory Comput., 2024, 20, 7884–7903 Search PubMed.
- D. Luu and K. Patkowski, J. Phys. Chem. A, 2023, 127, 356–377 CrossRef.
- O. R. Meitei and A. Heßelmann, J. Comput. Chem., 2017, 38, 2500–2508 CrossRef.
- E. Pastorczak, A. Prlj, J. F. Gonthier and C. Corminboeuf, J. Chem. Phys., 2015, 143, 224107 CrossRef PubMed.
- J. F. Gonthier and C. Corminboeuf, J. Chem. Phys., 2014, 140, 154107 CrossRef.
- W. B. Schneider, G. Bistoni, M. Sparta, M. Saitow, C. Riplinger, A. A. Auer and F. Neese, J. Chem. Theory Comput., 2016, 12, 4778–4792 CrossRef PubMed.
- A. Altun, M. Saitow, F. Neese and G. Bistoni, J. Chem. Theory Comput., 2019, 15, 1616–1632 CrossRef PubMed.
- A. Altun, F. Neese and G. Bistoni, J. Chem. Theory Comput., 2019, 15, 215–228 CrossRef.
- G. Bistoni, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2019, e1442 Search PubMed.
- A. Altun, F. Neese and G. Bistoni, Beilstein J. Org. Chem., 2018, 14, 919–929 CrossRef PubMed.
- A. Altun, R. Izsák and G. Bistoni, Int. J. Quantum Chem., 2021, 121, e26339 CrossRef.
- G. Bistoni, A. A. Auer and F. Neese, Chem. – Eur. J., 2017, 23, 865–873 CrossRef PubMed.
- Q. Lu, F. Neese and G. Bistoni, Phys. Chem. Chem. Phys., 2019, 2019, 11569–11577 RSC.
- L. P. Ho, A. Nasr, P. G. Jones, A. Altun, F. Neese, G. Bistoni and M. Tamm, Chem. – Eur. J., 2018, 24, 18922–18932 CrossRef.
- R. Ghafarian Shirazi, F. Neese, D. A. Pantazis and G. Bistoni, J. Phys. Chem. A, 2019, 123, 5081–5090 CrossRef.
- M. E. Beck, C. Riplinger, F. Neese and G. Bistoni, J. Comput. Chem., 2021, 42, 293–302 CrossRef PubMed.
- S. Ghosh, F. Neese, R. Izsák and G. Bistoni, J. Chem. Theory Comput., 2021, 17, 3348–3359 CrossRef PubMed.
- G. Bistoni, A. Altun, Z. Wang and F. Neese, Acc. Chem. Res., 2024, 57, 1411–1420 CrossRef PubMed.
- A. Altun, I. F. Leach, F. Neese and G. Bistoni, ChemRxiv, 2024, preprint DOI:10.26434/chemrxiv-2024-fg6lj.
- Q. Lu, F. Neese and G. Bistoni, Angew. Chem., Int. Ed., 2018, 57, 4760–4764 CrossRef PubMed.
- Z. Wang and F. Neese, J. Chem. Phys., 2023, 158, 184102 CrossRef PubMed.
- A. Altun, F. Neese and G. Bistoni, J. Chem. Theory Comput., 2019, 15, 5894–5907 CrossRef PubMed.
- A. Altun, F. Neese and G. Bistoni, J. Chem. Theory Comput., 2022, 18, 2292–2307 CrossRef PubMed.
- A. Altun, M. Garcia-Ratés, F. Neese and G. Bistoni, Chem. Sci., 2021, 12, 12785–12793 RSC.
- J. Schümann, L. Ochmann, J. Becker, A. Altun, I. Harden, G. Bistoni and P. Schreiner, J. Am. Chem. Soc., 2023, 145, 2093–2097 CrossRef PubMed.
- A. M. Preda, W. B. Schneider, D. Schaarschmidt, H. Lang, L. Mertens, A. A. Auer and M. Mehring, Dalton Trans., 2017, 46, 13492–13501 RSC.
- C. Ganesamoorthy, S. Heimann, S. Hölscher, R. Haack, C. Wölper, G. Jansen and S. Schulz, Dalton Trans., 2017, 46, 9227–9234 RSC.
- C. H. Borca, B. W. Bakr, L. A. Burns and C. D. Sherrill, J. Chem. Phys., 2019, 151, 144103 CrossRef PubMed.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. De Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. Buongiorno Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, M. Cococcioni, N. Colonna, I. Carnimeo, A. Dal Corso, S. De Gironcoli, P. Delugas, R. A. Distasio, A. Ferretti, A. Floris, G. Fratesi, G. Fugallo, R. Gebauer, U. Gerstmann, F. Giustino, T. Gorni, J. Jia, M. Kawamura, H. Y. Ko, A. Kokalj, E. Kücükbenli, M. Lazzeri, M. Marsili, N. Marzari, F. Mauri, N. L. Nguyen, H. V. Nguyen, A. Otero-De-La-Roza, L. Paulatto, S. Poncé, D. Rocca, R. Sabatini, B. Santra, M. Schlipf, A. P. Seitsonen, A. Smogunov, I. Timrov, T. Thonhauser, P. Umari, N. Vast, X. Wu and S. Baroni, J. Phys. Condens. Matter, 2017, 29, 465901 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- C. Zhang, J. Liu, Z. Ren, D. D. Koelling and B. N. Harmon, J. Phys. C: Solid State Phys., 1977, 10, 3107 CrossRef.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B: Condens. Matter Mater. Phys., 1976, 13, 5188–5192 CrossRef.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 73–78 CAS.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2018, 8, e1327 Search PubMed.
- F. Neese, F. Wennmohs, U. Becker and C. Riplinger, J. Chem. Phys., 2020, 152, 224108 CrossRef CAS PubMed.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- E. Rebolini, R. Izsák, S. S. Reine, T. Helgaker and T. B. Pedersen, J. Chem. Theory Comput., 2016, 12, 3514–3522 CrossRef CAS PubMed.
- S. Kossmann and F. Neese, Chem. Phys. Lett., 2009, 481, 240–243 CrossRef CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- G. Bistoni, C. Riplinger, Y. Minenkov, L. Cavallo, A. A. Auer and F. Neese, J. Chem. Theory Comput., 2017, 13, 3220–3227 CrossRef CAS PubMed.
- S. F. Boys, Rev. Mod. Phys., 1960, 32, 296–299 CrossRef CAS.
- A. Altun, LEDAW – LED Analysis Wizard for Automating Local Energy Decomposition Analysis Using ORCA Outputs, 2024, https://github.com/ahmetaltunfatih/LEDAW.
- A. Wuttke and R. A. Mata, J. Comput. Chem., 2017, 38, 15–23 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed energetics and optimized coordinates of the structures (XLSX). See DOI: https://doi.org/10.1039/d4cp03697b |
| This journal is © the Owner Societies 2024 |