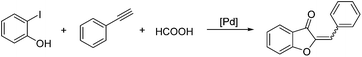Selective palladium-catalyzed carbonylative synthesis of aurones with formic acid as the CO source†
Xinxin
Qi
a,
Rui
Li
a and
Xiao-Feng
Wu
*ab
aDepartment of Chemistry, Zhejiang Sci-Tech University, Xiasha Campus, Hangzhou 310018, People's Republic of China. E-mail: xiao-feng.wu@catalysis.de
bLeibniz-Institut für Katalyse e.V. an der Universit Rostock, Albert-Einstein-Strasse 29a, 18059 Rostock, Germany
First published on 27th June 2016
Abstract
A general and practical strategy has been developed to prepare aurone derivatives. In the presence of the palladium catalyst, 2-iodophenol and terminal alkynes were reacted by using formic acid as the CO source with acetic anhydride as the additive. A variety of aurones were obtained in moderate to good yields. Notably, this is first report on carbonylative synthesis of aurones with formic acid as the CO source.
Flavonoids are a major class of plant natural products, exhibiting a broad variety of biological activities.1 As a subclass of the flavonoid family, aurones2 displaying a wide range of significant biological activities and have been utilized as antifungal agents,3 anticancer agents,4 and antioxidants.5 Therefore, numerous approaches for aurone synthesis have been reported. The most popular strategy is the condensation of benzofuran3(2H)-ones with benzaldehydes.6 This aldol-type reaction needs to prepare benzofuran3(2H)-ones from 2-phenoxyacetic acids under very harsh reaction conditions with modest yields. Other traditional synthetic methods include Wheeler aurone synthesis from chalcones dihalides,7 oxidative cyclization of 2′-hydroxychalcones,8 and ring closure of o-hydroxyaryl phenylethynyl ketones.9 However, these methods always result in a mixture of aurones and flavones. Thus, an selective and practical synthetic protocol for aurones synthesis is still in great demand.
Recently, palladium-catalyzed carbonylation reaction has drawn much attention for their widely application in both academy and industry.10 In these reactions, CO is used as the cheapest C1 source to prepare the carbonyl-containing compounds. Carbonylative procedures have also been applied in aurones synthesis, however problems such as the selectivity between aurones and flavones and limited substrates testing were not solved.11 Additionally, gaseous CO which is toxic, odorless, flammable and difficult to handle have to be applied. Under this background, various CO surrogates were developed and applied.12 In this regard, we wish to describe here a selective palladium-catalyzed carbonylation reaction of 2-iodophenol and terminal alkynes using formic acid as the CO precursor to provide aurone derivatives.13,14 The reaction proceeds in a selective manner and give the desired aurones in good yields. Notably, this is first report on carbonylative synthesis of aurones with formic acid as the CO source.
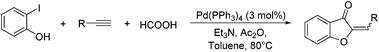
Initially, 2-iodophenol and phenyl acetylene was chosen as the model substrates to screen the reaction conditions (Table 1). Fortunately, the aurone product was obtained in 27% yield using Pd(OAc)2 as catalyst, PPh3 as ligand, Et3N as base in toluene at 80 °C (entry 1). Other bases such pyridine, DBU, tBuONa, and NaHCO3 were also studied, and Et3N still give the best results. We then tested different solvents (entries 6–8), toluene proven to be the optimal solvent here. Furthermore, a variety of mono- and bidentate ligands were investigated (entries 9–14), DPPF, DPPPE, and Xphos resulted in similar yields compared with PPh3, 40% and 49% yields were observed by using P(o-tolyl)3 and BuPAd2 (entries 13–14). The yield decreased in the absence of phosphine ligand (entry 15). To our delight, the highest yield can be obtained when the reaction was performed with preformed Pd(PPh3)4 as the catalyst (entry 16).
| Entry | Catalyst | Ligand | Base | Solvent | Yieldb (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 2-iodophenol (1.0 mmol), phenyl acetylene (2.0 mmol), catalyst (3 mol%), ligand (6 mol%), base (5 equiv.), HCOOH (2.0 mmol), acetic anhydride (2.0 mmol), solvent (2 mL), 14 h. b GC yield, with dodecane as the internal standard. c Ligand (4 mol%). | |||||
| 1 | Pd(OAc)2 | PPh3 | Et3N | Toluene | 27 |
| 2 | Pd(OAc)2 | PPh3 | Pyridine | Toluene | 25 |
| 3 | Pd(OAc)2 | PPh3 | DBU | Toluene | 6 |
| 4 | Pd(OAc)2 | PPh3 | tBuONa | Toluene | 6 |
| 5 | Pd(OAc)2 | PPh3 | NaHCO3 | Toluene | 24 |
| 6 | Pd(OAc)2 | PPh3 | Et3N | THF | 9 |
| 7 | Pd(OAc)2 | PPh3 | Et3N | DMF | 20 |
| 8 | Pd(OAc)2 | PPh3 | Et3N | CH3CN | 25 |
| 9c | Pd(OAc)2 | DPPF | Et3N | Toluene | 24 |
| 10c | Pd(OAc)2 | DPPPE | Et3N | Toluene | 20 |
| 11c | Pd(OAc)2 | Xantphos | Et3N | Toluene | 18 |
| 12 | Pd(OAc)2 | XPhos | Et3N | Toluene | 27 |
| 13 | Pd(OAc)2 | P(o-tolyl)3 | Et3N | Toluene | 40 |
| 14 | Pd(OAc)2 | BuPAd2 | Et3N | Toluene | 49 |
| 15 | Pd(OAc)2 | — | Et3N | Toluene | 17 |
| 16 | Pd(PPh3)4 | — | Et3N | Toluene | 85 |
| 17 | PdCl2(PPh3)2 | — | Et3N | Toluene | 54 |
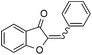
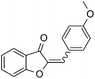
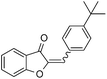
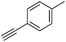
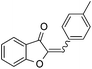
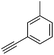
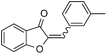
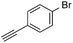
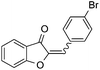
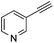
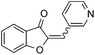
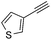
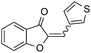
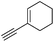
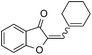
With the best reaction conditions in hand, we went on our study with a series of terminal alkynes (Table 2). First, various aromatic alkynes were examined, substrates with electron-donating groups including methoxy, tert-butyl, and methyl gave the corresponding products in very good yields (entries 2–4) those substrates with methyl group substituted at ortho-position gave higher yield than meta- and para- substitution (entry 4, 5 vs. 6). Electron-withdrawing group such as trifluoromethyl provided the desired products in 73% yields (entry 7). Subsequent examination of halide groups showed that fluoro and chloro substitutions resulted in better yields than bromo group (entry 8, 9 vs. 10). Moreover, heteroaryl groups involved thiophene, and pyridine moieties worked well to afford the desired aurone products in good yields (entries 11–12). Furthermore, alkyl alkynes were also tolerated well to provide the target products in good yields (entries 13–15).
| Entry | Alkyne | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: 2-iodophenol (1.0 mmol), terminal acetylene (2.0 mmol), Pd(PPh3)4 (3 mol%), Et3N (5 equiv.), HCOOH (2.0 mmol), acetic anhydride (2.0 mmol), toluene (2 mL), 12–18 h. b Isolated yields. | |||
| 1 |
|
|
82 |
| 2 |
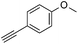
|
|
82 |
| 3 |
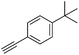
|
|
68 |
| 4 |
|
|
75 |
| 5 |
|
|
72 |
| 6 |
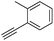
|
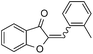
|
81 |
| 7 |
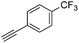
|
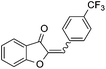
|
73 |
| 8 |
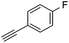
|

|
80 |
| 9 |
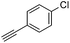
|
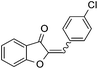
|
75 |
| 10 |
|
|
58 |
| 11 |
|
|
55 |
| 12 |
|
|
68 |
| 13 |
|
|
54 |
| 14 |
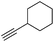
|
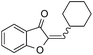
|
62 |
| 15 |
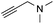
|
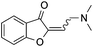
|
51 |
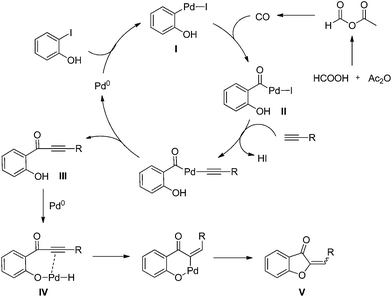
Based on the results of aurone synthesis from 2-iodophenol and terminal alkynes, a plausible reaction mechanism was proposed in Scheme 1. Initially, Pd(0) underwent oxidative addition with 2-iodophenol to form arylpalladium species I, and aroylpalladium iodide complex II was formed by the insertion of CO, which generated from formic acid and acetate anhydride. Then alkynes attack and elimination to afford alkynyl ketone intermediate III. Then, Pd(0) inserted to the O–H bond of phenol to provide complex IV, followed by insertion and reductive elimination to give the final aurones V products. However, the insertion of the alkyne into intermediate II followed by cyclopalladation and reductive elimination cannot be excluded.
Conclusions
In conclusion, a palladium-catalyzed carbonylative reaction of 2-iodophenol and terminal alkynes has been developed. In this strategy, instead of toxic CO gas, formic acid was utilized as the CO source with acetic anhydride as the activator. Aurones were prepared in good yields with good selectivity and functional group tolerance.Acknowledgements
The authors thank the financial supports from NSFC (21472174) and Zhejiang Natural Science Fund for Distinguished Young Scholars (LR16B020002). X.-F. Wu appreciates the general support from Professor Matthias Beller in LIKAT.Notes and references
- (a) I. Vints and S. Rozen, J. Org. Chem., 2014, 79, 7261 CrossRef CAS; (b) Y. B. Su, C. Hong, Q. Li, W. Luo and C. J. Wang, Chin. J. Org. Chem., 2014, 34, 600 CrossRef CAS; (c) D. H. Lou, G. Q. Wang, L. Xia, L. Chen and Q. A. Wang, Chin. J. Org. Chem., 2013, 33, 535 CrossRef CAS; (d) A. Detsi, M. Majdalani, C. A. Kontogiorgis, D. Hadjipavlou-Litina and P. Kefalas, Bioorg. Med. Chem., 2009, 17, 8073 CrossRef CAS; (e) N. C. Veitch and R. J. Grayer, Nat. Prod. Rep., 2008, 25, 555 RSC; (f) T. Iwashina, J. Plant Res., 2000, 113, 287 CrossRef CAS; (g) O. M. Andersen and K. R. Markham, Flavanoids: Chemistry, Biochemistry and Applications, CRC Press, Boca Raton, 2006 Search PubMed.
- (a) A. Boumendjel, Curr. Med. Chem., 2003, 10, 2621 CrossRef CAS; (b) E. Ono, M. Fukuchi-Mizutani, N. Nakamura, Y. Fukui, K. Yonekura-Sakakibara, M. Yamaguchi, T. Nakayama, T. Tanaka, T. Kusumi and Y. Tanaka, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 11075 CrossRef CAS.
- C. J. W. Brooks and D. G. Watson, Nat. Prod. Rep., 1985, 427 RSC.
- (a) N. J. Lawrence, D. Rennison, A. T. McGown and J. A. Hadfield, Bioorg. Med. Chem. Lett., 2003, 13, 3759 CrossRef CAS; (b) H. Cheng, L. Zhang, Y. Liu, S. Chen, H. Cheng, X. Lu, Z. Zheng and G.-C. Zhou, Eur. J. Med. Chem., 2010, 45, 5950 CrossRef CAS.
- (a) S. Venkateswarlu, G. K. Panchagnula and G. V. Subbaraju, Biosci., Biotechnol., Biochem., 2004, 68, 2183 CrossRef CAS; (b) A. Detsi, M. Majdalani, C. A. Kontogiorgis, D. Hadjipavlou-Litina and P. Kefalas, Bioorg. Med. Chem., 2009, 17, 8073 CrossRef CAS.
- (a) H. Cheng, L. Zhang, Y. Liu, S. Chen, H. Cheng, X. Lu, Z. Zheng and G.-C. Zhou, Eur. J. Med. Chem., 2010, 45, 5950 CrossRef CAS; (b) S. Okombi, D. Rival, S. Bonnet, A.-M. Mariotte, E. Perrier and A. Boumendjel, J. Med. Chem., 2006, 49, 329 CrossRef CAS; (c) R. S. Varma and M. Varma, Tetrahedron Lett., 1992, 33, 5937 CrossRef CAS; (d) I. Hawkins and S. T. Handy, Tetrahedron, 2013, 69, 9200 CrossRef CAS; (e) S. Venkateswarlu, G. K. Panchagnula and G. V. Subbaraju, Biosci., Biotechnol., Biochem., 2004, 68, 2183 CrossRef CAS.
- (a) J. A. Donnelly, M. J. Fox and T. C. Sharma, Tetrahedron, 1979, 35, 875 CrossRef CAS; (b) G. Bose, E. Mondal, A. T. Khan and M. J. Bordoloi, Tetrahedron Lett., 2001, 42, 8907 CrossRef CAS.
- (a) A. Lévai and A. L. Tökés, Synth. Commun., 1982, 12, 701 CrossRef; (b) K. Imafuku, M. Honda and J. F. W. McOmie, Synthesis, 1987, 199 CrossRef CAS; (c) H. Sekizaki, Bull. Chem. Soc. Jpn., 1988, 61, 1407 CrossRef CAS; (d) K. Thakkar and M. Cushman, J. Org. Chem., 1995, 60, 6499 CrossRef CAS; (e) A. K. Patel, N. H. Patel, M. A. Patel and D. I. Brahmbhatt, J. Heterocycl. Chem., 2012, 49, 504 CrossRef CAS; (f) J. H. Yang, Y. M. Xie, S. B. Feng, W. B. Zuo, B. B. Chen, X. Q. Ma and Y. Feng, Chin. J. Org. Chem., 2013, 33, 2155 CrossRef CAS; (g) S. Moussouni, A. Detsi, M. Majdalani, D. P. Makris and P. Kefalas, Tetrahedron Lett., 2010, 51, 4076 CrossRef CAS; (h) S. Venkateswarlu, G. K. Panchagnula, A. L. Gottumukkala and G. V. Subbaraju, Tetrahedron, 2007, 63, 6909 CrossRef CAS.
- (a) H. Garcia, S. Iborra, J. Primo and M. A. Miranda, J. Org. Chem., 1986, 51, 4432 CrossRef CAS; (b) C. M. Brennan, C. D. Johnson and P. D. McDonnell, J. Chem. Soc., Perkin Trans. 2, 1989, 957 RSC; (c) Z.-W. An, M. Catellani and G. P. Chiusoli, J. Organomet. Chem., 1990, 397, 371 CrossRef CAS; (d) T.-T. Jong and S.-J. Leu, J. Chem. Soc., Perkin Trans. 1, 1990, 423 RSC.
- For reviews on palladium-catalyzed carbonylations, see: (a) A. Brennführer, H. Neumann and M. Beller, Angew. Chem., Int. Ed., 2009, 48, 4114 CrossRef; (b) R. Skoda-Foldes and L. Kollör, Curr. Org. Chem., 2002, 6, 1097 CrossRef CAS; (c) R. Grigg and S. P. Mutton, Tetrahedron, 2010, 66, 5515 CrossRef CAS; (d) X.-F. Wu, H. Neumann and M. Beller, Chem. Soc. Rev., 2011, 40, 4986 RSC; (e) X.-F. Wu, H. Neumann and M. Beller, Chem. Rev., 2013, 113, 1 CrossRef CAS; (f) B. Gabriele, R. Mancuso and G. Salerno, Eur. J. Org. Chem., 2012, 6825–6839 CrossRef CAS.
- (a) Z. An, M. Catellani and G. P. Chiusoli, J. Organomet. Chem., 1989, 371, C51 CrossRef CAS; (b) J. Liu, M. Liu, Y. Yue, N. Zhang, Y. Zhang and K. Zhuo, Tetrahedron Lett., 2013, 54, 1802 CrossRef CAS; (c) P. G. Ciattini, E. Morera, G. Ortar and S. S. Rossi, Tetrahedron, 1991, 47, 6449 CrossRef CAS; (d) M. Catellani, G. P. Chiusoli, M. C. Fagnola and G. Solari, Tetrahedron Lett., 1994, 35, 5923 CrossRef CAS.
- For reviews on CO surrogates, see: (a) T. Morimoto and K. Kakiuchi, Angew. Chem., Int. Ed., 2004, 43, 5580 CrossRef CAS; (b) L. Wu, Q. Liu, R. Jackstell and M. Beller, Angew. Chem., Int. Ed., 2014, 53, 6310 CrossRef CAS PubMed; (c) P. Gautam and B. M. Bhanage, Catal. Sci. Technol., 2015, 5, 4663 RSC; (d) B. Sam, B. Breit and M. J. Krische, Angew. Chem., Int. Ed., 2015, 54, 3267 CrossRef CAS; (e) S. D. Friis, A. T. Lindhardt and T. Skrydstrup, Acc. Chem. Res., 2016, 49, 594 CrossRef CAS; (f) L. R. Odell, F. Russo and M. Larhed, Synlett, 2012, 685 CrossRef CAS; (g) H. Konishi and K. Manabe, Synlett, 2014, 25, 1971 CrossRef CAS.
- (a) S. Cacchi, G. Fabrizi and A. Goggiamani, Org. Lett., 2003, 5, 4269 CrossRef CAS; (b) S. Cacchi, G. Fabrizi and A. Goggiamani, J. Comb. Chem., 2004, 6, 692 CrossRef CAS; (c) S. Korsager, R. H. Taaning and T. Skrydstrup, J. Am. Chem. Soc., 2013, 135, 2891 CrossRef CAS; (d) J. Hou, J. H. Xie and Q.-L. Zhou, Angew. Chem., Int. Ed., 2015, 54, 6302 CrossRef CAS.
- (a) X. Qi, L.-B. Jiang, C.-L. Li, R. Li and X.-F. Wu, Chem.–Asian J., 2015, 10, 1870 CrossRef CAS; (b) X. Qi, L.-B. Jiang, H. P. Li and X. F. Wu, Chem.–Eur. J., 2015, 21, 17650 CrossRef CAS; (c) L. B. Jiang, R. Li, H. P. Li, X. Qi and X. F. Wu, ChemCatChem, 2016, 8, 1788–1791 CrossRef CAS; (d) X. Qi, C. L. Li and X. F. Wu, Chem.–Eur. J., 2016, 22, 5835 CrossRef CAS; (e) X. Qi, C.-L. Li, L.-B. Jiang, W.-Q. Zhang and X.-F. Wu, Catal. Sci. Technol., 2016, 6, 3099 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental detail. See DOI: 10.1039/c6ra13615j |
| This journal is © The Royal Society of Chemistry 2016 |