A family of 3D lanthanide organic frameworks with tunable luminescence and slow magnetic relaxation†
Qipeng Liab and
Shaowu Du*a
aState Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, Fujian 350002, P. R. China. E-mail: swdu@fjirsm.ac.cn; Fax: +86 591 83709470
bUniversity of Chinese Academy of Sciences, Beijing 100039, P. R. China
First published on 7th January 2015
Abstract
A family of 3D lanthanide organic frameworks with 5-(pyridin-4-yl)isophthalic acid (H2PIP), formulated as [Ln2(PIP)3(H2O)4]·2DMF·3H2O (Ln = Sm 1, Eu 2, Gd 3, Tb 4, Dy 5 and Ho 6) have been synthesized and structurally characterized. They are isomorphous and feature a 3D 6-connected pcu topology. The tunable luminescence of 2 and 4 is investigated by changing the temperature and the variation of excitation wavelength. In addition, the slow magnetic relaxation behavior is observed in 5.
Introduction
The design and construction of lanthanide organic frameworks have attracted great interest not only due to their fascinating structures but also because of their potential applications as luminescent and magnetic materials.1 To date, a large number of lanthanide organic frameworks have been synthesized and their magnetic properties including single-molecule magnets (SMMs) and single-chain magnets (SCMs) have been extensively studied.2 Such lanthanide organic frameworks can be constructed by combining the lanthanide ions with various organic linkers under solvothermal conditions. Particularly, the Dy(III) ion has been widely used in the synthesis of molecule- based magnetic framework materials due to its large number of unpaired electrons and the presence of significant uniaxial anisotropy.3 In addition, the luminescent intensity and emission chromaticity of lanthanide organic frameworks can be tuned by varying the temperature and excitation wavelengths.4On the other hand, the selection of organic ligands is very crucial for the synthesis of lanthanide organic frameworks. For example, the multi-carboxylic acid ligands are widely used in the construction of lanthanide organic frameworks owing to their various coordination modes and high affinity of lanthanide ions for the oxygen atom.5 So far, a great deal of lanthanide organic frameworks with 1,3-benzenedicarboxylic acid (1,3-H2BDC) and pyridine-3,5-dicarboxylic acid (3,5-H2PDC) ligands have been synthesized.4a,6 In comparison, just a few examples of lanthanide organic frameworks constructed by 5-(4-pyridyl)isophthalic acid (H2PIP), which has one additional aromatic ring compared to H2BDC and 3,5-H2PDC ligands have been reported up to now.7
In our present work, we successfully construct a family of 3D lanthanide organic frameworks based on the H2PIP ligand, formulated as [Ln2(PIP)3(H2O)4]·2DMF·3H2O (Ln = Sm 1, Eu 2, Gd 3, Tb 4, Dy 5 and Ho 6), which are isomorphous and both display a 3D 6-connected pcu topology. Our results showed that the luminescence of 2 and 4 could be tuned by changing the temperature and the excitation wavelength. Furthermore, the slow magnetic relaxation behavior is observed in 5.
Experimental
Materials and methods
All the starting materials and solvents were available commercially sources and used as purchased without further purification. Thermogravimetric experiments were performed using a TGA/NETZSCH STA-449C instrument heated from 30–1000 °C (heating rate of 10 °C min−1, nitrogen stream). IR spectra using KBr pellets were recorded on a Spectrum-One FT-IR spectrophotometer. The powder X-ray diffraction (XRD) patterns were recorded on crushed single crystals in the 2θ range 5–50° using Cu-Kα radiation. Element analyses for C, H and N were measured with an Elemental Vairo ELIII analyzer. Fluorescence spectra for the solid samples were performed on an Edinburgh Analytical instrument FLS920. The magnetic susceptibility data were collected on Quantum Design MPMS (SQUID)-XL magnetometer and PPMS-9T system.Synthesis of [Ln2(PIP)3(H2O)4]·2DMF·3H2O (Ln = Sm 1, Eu 2, Gd 3, Tb 4, Dy 5 and Ho 6)
H2PIP (0.5 mmol) and Sm(NO3)3·6H2O (0.25 mmol) were placed in a 20 mL of Teflon-lined stainless steel vessel with 10 mL of DMF–H2O (v/v = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The mixtures were heated to 120 °C in 4 h and kept at this temperature for three days, and then cooled to room temperature during another two days. Pink crystals of 1 were obtained in 60% yield based on Sm(NO3)3·6H2O. Elemental anal. calcd. for 1 Sm2C45H50N5O22 (%): C, 41.15; N, 5.33; H, 3.84. Found (%): C, 41.40; H, 3.87; N, 5.23. IR (KBr, cm−1): 3360 s, 3053 vw, 2928 vw, 1872 vw, 1660 s, 1550 w, 1429 m, 1300 w, 1099.5 vw, 1005 m, 936.2 m, 878 w, 827.0 s, 781.6 w, 715 w, 634.2 m.
1). The mixtures were heated to 120 °C in 4 h and kept at this temperature for three days, and then cooled to room temperature during another two days. Pink crystals of 1 were obtained in 60% yield based on Sm(NO3)3·6H2O. Elemental anal. calcd. for 1 Sm2C45H50N5O22 (%): C, 41.15; N, 5.33; H, 3.84. Found (%): C, 41.40; H, 3.87; N, 5.23. IR (KBr, cm−1): 3360 s, 3053 vw, 2928 vw, 1872 vw, 1660 s, 1550 w, 1429 m, 1300 w, 1099.5 vw, 1005 m, 936.2 m, 878 w, 827.0 s, 781.6 w, 715 w, 634.2 m.
Colorless crystals of 2–5 and pink crystals of 6 were obtained in moderate yields (49–66%) by a similar method as described for 1 except that the corresponding Ln(NO3)3·6H2O salts were used instead of Sm(NO3)3·6H2O. Elemental anal. calcd. for 2 Eu2C45H50N5O22 (%): C, 41.05; H, 3.83; N, 5.32. Found (%): C, 41.40; H, 3.78; N, 5.43. IR (KBr, cm−1): 3382.3 s, 3065.3 w, 2928.6 w, 1815.2 w, 1663.3 vw, 1500.9w, 1440.2 vw, 1298.7 w, 1255.7 w, 1099.5 w, 1068.9 vw, 1007.4 w, 932.1 vw, 876.7 m, 826.8 s, 781.7 s, 716.5 m, 666.5 m, 643.9 s. Elemental anal. calcd. for 3 Gd2C45H50N5O22 (%): C, 40.72; N, 5.28; H, 3.80. Found (%): C, 40.38; H, 3.87; N, 5.25. IR (KBr, cm−1): 3360 s, 3060 vw, 2914 vw, 1869 vw, 1665 s, 1552 w, 1442 m, 1305 w, 1100.8 vw, 1007 m, 937 m 879 w, 827 s, 785 w, 717 w, 636 m. Elemental anal. calcd. for 4 Tb2C45H50N5O22 (%): C, 40.62; H, 3.79; N, 5.26. Found (%): C, 40.40; H, 3.87; N, 5.23. IR (KBr, cm−1): 3383.8 s, 3065.7 vw, 2928.6 vw, 1944.1 w, 1869.2 vw, 1666.5 w, 1552.3 vw, 1507.9 w, 1440.6 w, 1299.5 m, 1225.6 m, 1174.9 w, 1099.8 m, 1007.7 m, 936.6 w, 877.7 w, 781.6 s, 716.4 s, 667.2 m, 635.3 s, 571.8 m, 513.3 w. Elemental anal. calcd. for 5 Dy2C45H50N5O22 (%): C, 40.12; N, 3.68; H, 5.47. Found (%): C, 40.40; H, 3.77; N, 5.23. IR (KBr, cm−1): 3381 s, 3065 vw, 2928 vw, 1869 vw, 1658 s, 1552 w, 1439 m, 1300 w, 1099.8 vw, 1007 m, 936.6 m 877.7 w, 827.0 s, 781.6 w, 716.4 w, 635.3 m. Elemental anal. calcd. for 6 Ho2C45H50N5O22 (%): C, 40.23; N, 5.22; H, 3.75. Found (%): C, 40.12; H, 3.79; N, 5.25. IR (KBr, cm−1): 3392 s, 3058 vw, 2910 vw, 1860 vw, 1658 s, 1545 w, 1445 m, 1300 w, 1099 vw, 1007 m, 938 m 877.8 w, 827 s, 783 w, 716.8 w, 635.6 m.
Crystal structure determination
Single-crystal X-ray diffraction data were collected on a Rigaku Diffractometer with a Mercury CCD area detector (Mo Kα; λ = 0.71073 Å) at room temperature. Empirical absorption corrections were applied to the data using the Crystal Clear program.8 The structures were solved by the direct method and refined by the full-matrix least-squares on F2 using the SHELXTL-97 program.9 Metal atoms in each compounds were located from the E-maps and other non-hydrogen atoms were located in successive difference Fourier syntheses. All non-hydrogen atoms were refined anisotropically and the hydrogen atoms were positioned geometrically. The hydrogen atoms of coordinated water and lattice water molecules were not added. The PIP2− ligand and coordinated water molecule in 1 are both positional disordered and have been treated as two equal parts. Some lattice water molecules in 1 are also disordered.10 The crystal data of 1–6 all existed severe disordered problems, thus only the refinement of crystal data of 1 and 5 were provided. Crystallographic data and other pertinent information for 1 and 5 are summarized in Table 1. Selected bond lengths and angles for 1 and 5 are listed in Table S1 and S2.†| Compounds | 1 | 5 |
|---|---|---|
| a R = ∑(‖Fo| − |Fc‖)/∑|Fo|, wR = {∑w[(Fo2 − Fc2)2]/∑w[(Fo2)2]}1/2. | ||
| CCDC | 1010482 | 958686 |
| Formula | C45H50N5O22Sm2 | C45H50N5O22Dy2 |
| Mr | 1313.60 | 1337.90 |
| Space group | P21/c | P21/c |
| a (Å) | 13.472(6) | 13.405(3) |
| b (Å) | 14.814(7) | 14.705(4) |
| c (Å) | 13.413(8) | 13.388(3) |
| α (deg) | 90 | 90 |
| β (deg) | 99.14(3) | 99.34(4) |
| γ (deg) | 90 | 90 |
| V (Å3) | 2642.9(2) | 2603.9(1) |
| Z | 2 | 2 |
| Dc (g cm−3) | 1.630 | 1.686 |
| M (mm−1) | 2.28 | 2.93 |
| F(000) | 1278 | 1326 |
| GOF | 1.09 | 1.05 |
| R1a | 0.041 | 0.032 |
| wR2a | 0.099 | 0.075 |
Results and discussion
Synthesis and description of crystal structures
A family of 3D lanthanide organic frameworks with H2PIP ligand, namely [Ln2(PIP)3(H2O)4]·2DMF·3H2O (Ln = Sm 1, Eu 2, Gd 3, Tb 4, Dy 5 and Ho 6) have been successfully prepared by the hydrothermal reaction of Ln(NO3)3·6H2O and H2PIP in the mixed-solvent of DMF–H2O (v/v = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Compounds 1–6 are isomorphous, which is confirmed by XRD, TGA, IR, elemental analysis and lattice parameters. Herein, only the structure of 1 is discussed in detail.
1). Compounds 1–6 are isomorphous, which is confirmed by XRD, TGA, IR, elemental analysis and lattice parameters. Herein, only the structure of 1 is discussed in detail.
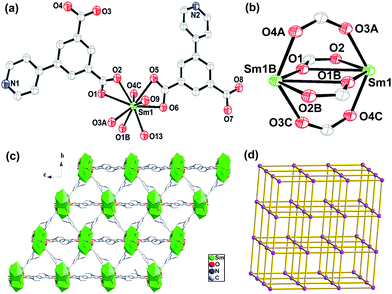
Compound 1 crystallizes in the monoclinic space group P21/c and its asymmetric unit contains one Sm(III) ion, one and a half PIP2− ligands, two coordinated water molecules, one guest DMF and one and a half lattice water molecules. The Sm(III) center is nine-coordinated by seven carboxylate O atoms from five different PIP2− ligands and two O atoms from two different coordinated water molecules (Fig. 1a). The PIP2− ligands in 1 display (κ1-κ1)-(κ2-μ2)-μ4 and (κ2)-(κ2)-μ2 coordination modes, respectively (Scheme S1†).
In addition, two equivalent Sm(III) ions are bridged by four carboxylates via bridging bidentate and tridentate arrangements to generate a {Sm2(CO2)4} unit with the Sm⋯Sm distance of 4.117 Å (Fig. 1b). Such dinuclear units act as secondary building units (SBUs) which are expanded into a 1D chain through carboxylates. These chains are further extended into a 2D layer network and 3D frameworks via the carboxylates of PIP2− ligands (Fig. 1c). From the topological point of view, the PIP2− ligands can be defined as a connector and the dinuclear {Sm2(CO2)4} units can be simplified as a six-connected node. Therefore 1 can be abstracted into a 6-connected network with the Schläfli symbol of {4^12.6^3}, which is a typical pcu topological net (Fig. 1d).11
XRD patterns and thermal properties
The powder XRD of 1–6 are performed at room temperature to characterize their purity (Fig. S1†). All the diffraction peak positions on the curves correspond well with the simulated XRD patterns, indicating the phase purity of the as-synthesized samples. The thermo-gravimetric analysis (TGA) experiments of 1–6 are conducted in the temperature range of 30–1000 °C under a flow of nitrogen with the heating rate of 10 °C min−1. The TGA curves of 1–6 are very similar and 1 has a weight loss of 20.55% from 40 to 400 °C, which is assigned to the release of three guest water and two DMF and four coordinated water molecules (calcd. 20.34%). Then the framework begins to decompose upon further heating (Fig. S2†).Tunable luminescence
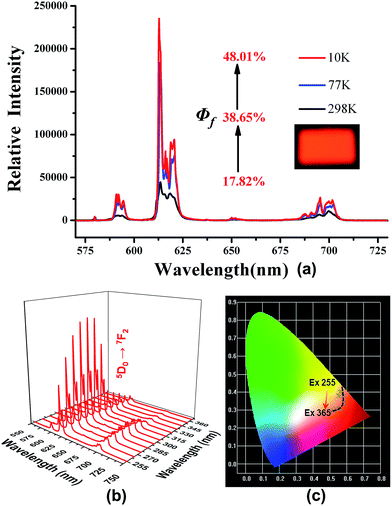
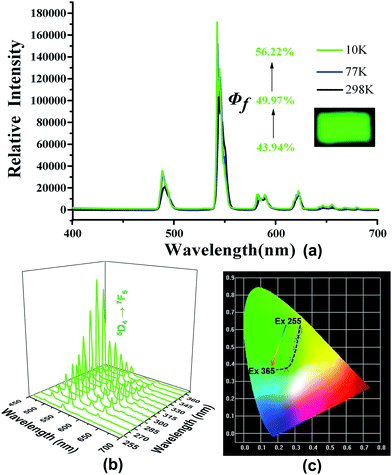
The free H2PIP ligand presented an emission with the band peaking around 459 nm upon excitation at 394 nm (Fig. S3†). Upon excitation at 280 nm, the characteristic emissions of 2 are observed in the range of 570–720 nm. The typical emission peaks are associated with the 4f → 4f transitions of the 5D0 excited state to the low-lying 7FJ (J = 0–4) levels. Note that the 5D0 → 7F2 transition of Eu(III) centered at 614 nm for 2 (Fig. S4†).4 When it's excited at 310 nm, the emission spectrum of 4 displays green luminescence with the typical emission peaks at 488, 543, 582 and 621 nm, which are assigned to 5D4 → 7FJ (J = 6–3) transitions (Fig. S5†).The quantum yields were measured at 298, 77 and 10 K are 17.82, 38.6 and 48.01% for 2 (at λex = 280 nm) and 43.94, 49.97 and 56.22% for 4 (at λex = 310 nm), respectively (Fig. 2a and 3a). These results reveal that the quantum yields of 2 and 4 increase gradually with dropping temperature, which may be due to the reduction of molecular thermal vibration at low temperatures.12,7b The corresponding lifetime at 298 K for 2 is ca. 0.3911 ms, while that for 4 is ca. 0.8372 ms (Table S3†). The luminescence decay curves of 2 and 4 are determined by monitoring 5D0 → 7F2 line excited at 280 nm and 5D4 → 7F5 line excited at 310 nm, respectively.
The emission intensity of 5D0 → 7F2 in 2 gradually increases with the excitation wavelength varied from 255 to 285 nm, but gradually decreases with the excitation wavelength varied from 295 to 365 nm (Fig. 2b). As a result, the emission of 2 varies from light-yellow to red and then to orange, as illustrated by the CIE-1931 chromaticity diagram (Fig. 2c). A similar situation occurs with the emission intensity of 5D4 → 7F5 in 4. As the excitation wavelength varies from 255 to 365 nm, the emission of 4 changes from yellow-green to yellowish green, and then to green (Fig. 3b and c).
Magnetic properties
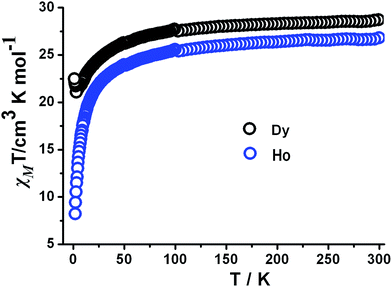
Temperature-dependent magnetic susceptibility measurements of 5 and 6 have been carried out in an applied magnetic field of 1000 Oe in the temperature range 300–2 K. The χMT value of 5 at 300 K is 28.73 cm3 K mol−1, which is very close to the value of 28.33 cm3 K mol−1 for two uncoupled Dy(III) ions (S = 5/2, L = 5, 6H15/2 and g = 4/3).13 The χMT product gradually decreases with the temperature to reach a minimum of 21.09 cm3 K mol−1 at 3.91 K, which is mainly ascribed to the progressive depopulation of excited Stark sublevels (Fig. 4). At low temperatures, the χMT product increases up to 22.50 cm3 K mol−1 at 2 K, which obviously suggests the presence of weak ferromagnetic interactions between Dy(III) ions as observed in other dinuclear Dy(III) systems.14 In addition, the temperature dependent χM−1 value above 10 K obeys the Curie–Weiss law with C = 28.7 cm3 K mol−1 and θ = −4.67 K (Fig. S6†).At room temperature, the χMT value is 24.46 cm3 K mol−1 for 6, which is lower than 28.15 cm3 K mol−1 for two independent Ho(III) (5I8 and g = 5/4).15 Below 50 K, the χMT value decreases smoothly to reach a minimal value of 8.30 cm3 K mol−1 at 2 K (Fig. 4). This feature indicates anti-ferromagnetic interactions between Ho(III) ions. This tendency can be explained by the spin orbital coupling of Ho-MOFs, which leads to the splitting of 4fn configuration into 7F6 states and finally into Stark components under the ligand field perturbation.15 Above 10 K, the magnetic data was fitted by Curie–Weiss equation to give a Curie constant C = 27.30 cm3 mol−1 and Weiss temperature θ = −6.10 K (Fig. S7†). The negative θ value indicates the presence of anti-ferromagnetic interactions between the Ho(III) ions.
In the M vs. H/T plot of 5, the M values show a rapid increase in the magnetization at low field and the maximum value is 14.96Nβ at 2 K and 7 T (Fig. S8†), which is lower than 20Nβ for two Dy(III) ions. It is likely that the anisotropy and crystal-field effect at the Dy(III) ion eliminate the 16-fold degeneracy of the 6H15/2 ground state.16 The absence of saturation on the M vs. H data at 2 K suggests the presence of significant anisotropy and low-lying excited states.
In order to understand the dynamics of magnetization, the ac susceptibility measurements of 5 were carried out in a zero-applied dc field with 3 Oe ac field oscillating at the indicated frequencies (311–1511 Hz) under the temperature range of 2–8 K (Fig. 5). Both in-phase (χ′T) and out-of-phase (χ′′) signals display frequency dependence below 8 K, which suggest the slow magnetization (M) relaxation behavior.17
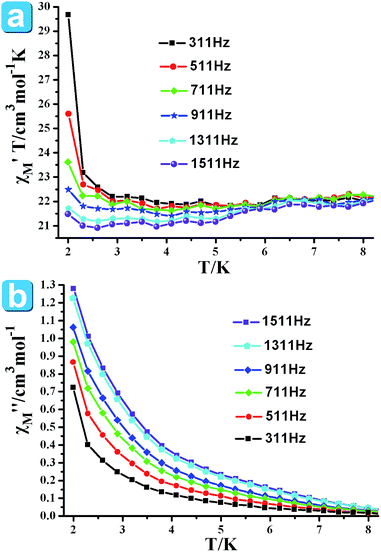 | ||
| Fig. 5 Ac susceptibility measured in zero dc fields and plotted as χ′T vs. T (a) and χ′′ vs. T (b) for 5. | ||
Conclusions
A family of new 3D lanthanide organic frameworks constructed by H2PIP have been synthesized and structurally characterized, which are isomorphous and each features a 3D 6-connected pcu topology. It has been observed that the luminescent quantum yields of 2 and 4 increase with the decreasing of temperature and their emissions can be tuned by altering the excitation wavelength. In addition, the slow magnetic relaxation behavior is observed in 5.Acknowledgements
This work was supported by the National Basic Research Program of China (973 Program, 2012CB821702), the National Natural Science Foundation of China (21233009 and 21173221) and the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences.Notes and references
- (a) M. D. Allendorf, C. A. Bauer, R. K. Bhakta and R. J. T. Houka, Chem. Soc. Rev., 2009, 38, 1330 RSC; (b) A. J. Tasiopoulos, A. Vinslava, W. Wermsdorfer, K. A. Abboub and G. Christou, Angew. Chem., Int. Ed., 2004, 43, 2117 CrossRef CAS PubMed; (c) Y. J. Cui, Y. F. Yue, G. D. Qian and B. L. Chen, Chem. Rev., 2012, 112, 1126 CrossRef CAS PubMed; (d) M. Kurmoo, Chem. Soc. Rev., 2009, 38, 1353 RSC; (e) H. L. Sun, Z. M. Wang and S. Gao, Coord. Chem. Rev., 2010, 254, 1081 CrossRef CAS PubMed; (f) J. Xie, H. M. Shu, Z. X. Han, S. S. Shen, F. Yuan, M. L. Yang, F. X. Dong and G. L. Xue, ChemPlusChem, 2014, 79, 985 CrossRef CAS.
- (a) M. T. Gamer, Y. Lan, P. W. Roesky, A. K. Powell and R. Cle'rac, Inorg. Chem., 2008, 47, 6581 CrossRef CAS PubMed; (b) H. S. Ke, G. F. Xu, Y. N. Guo, P. Gamez, C. M. Beavers, S. J. Teat and J. K. Tang, Chem. Commun., 2010, 46, 6057 RSC; (c) B. Hussain, D. Savard, T. J. Burchell, W. Wernsdorfer and M. Murugesu, Chem. Commun., 2009, 1100 RSC; (d) J. D. Rinehart and J. R. Long, Chem. Sci., 2011, 2, 2078 RSC; (e) T. A. Hudson, K. J. Berry, B. J. Moubaraki, K. S. Murray and R. Robson, Inorg. Chem., 2006, 45, 549 CrossRef PubMed; (f) A. S. R. Chesman, D. R. Turner, B. Moubaraki, K. S. Murray, G. B. Deacon and S. R. Batten, Chem.–Eur. J., 2009, 15, 5203 CrossRef CAS PubMed.
- (a) L. Bogani, C. Sangregorio, R. Sessoli and D. Gatteschi, Angew. Chem., Int. Ed., 2005, 44, 5817 CrossRef CAS PubMed; (b) K. Bernot, L. Bogani, A. Caneschi, D. Gatteschi and R. Sessoli, J. Am. Chem. Soc., 2006, 128, 7947 CrossRef CAS PubMed; (c) J. Rocha, L. D. Carlos, F. A. Paz and D. Ananias, Chem. Soc. Rev., 2011, 40, 926 RSC; (d) H. S. Ke, P. Gamez, L. Zhao, G. F. Xu, S. F. Xue and J. K. Tang, Inorg. Chem., 2010, 49, 7549 CrossRef CAS PubMed; (e) R. Sessoli and A. K. Powell, Coord. Chem. Rev., 2009, 253, 2328 CrossRef CAS PubMed; (f) D. N. Woodruff, R. E. P. Winpenny and R. A. Layfield, Chem. Rev., 2013, 113, 5110 CrossRef CAS PubMed; (g) Y. N. Guo, L. Ungur, G. E. Granroth, A. K. Powell, C. J. Wu, S. E. Nagler, J. K. Tang, L. F. Chibotaru and D. M. Cui, Sci. Rep., 2014, 4, 5471 Search PubMed; (h) S. Y. Lin, W. Wernsdorfer, L. Ungur, A. K. Powell, Y. N. Guo, J. K. Tang, L. Zhao, L. F. Chibotaru and H. J. Zhang, Angew. Chem., Int. Ed., 2012, 51, 12767 CrossRef CAS PubMed; (i) P. Zhang, Y. N. Guo and J. K. Tang, Coord. Chem. Rev., 2013, 257, 1728 CrossRef CAS PubMed.
- (a) H. B. Zhang, L. J. Zhou, J. Wei, Z. H. Li, P. Lin and S. W. Du, J. Mater. Chem., 2012, 22, 21210 RSC; (b) H. B. Zhang, X. C. Shan, Z. J. Ma, L. J. Zhou, M. J. Zhang, P. Lin, S. M. Hu, E. Ma, R. F. Li and S. W. Du, J. Mater. Chem. C, 2014, 2, 1367 RSC; (c) L. Wang, E. Yang, H. B. Zhang and J. Zhang, ChemPlusChem., 2014, 79, 1080 CrossRef CAS.
- (a) L. P. Zhang, Y. H. Wan and L. P. Jin, Polyhedron, 2003, 22, 981 CrossRef CAS; (b) L. Pan, E. B. Woodlock, X. T. Wang and C. Zheng, Inorg. Chem., 2000, 39, 4174 CrossRef CAS; (c) J. G. Wang, C. C. Huang, X. H. Huang and D. S. Liu, Cryst. Growth Des., 2008, 8, 795 CrossRef CAS; (d) Q. P. Li, C. B. Tian, H. B. Zhang, J. J. Qian and S. W. Du, CrystEngComm, 2014, 16, 9208 RSC; (e) T. Devic, C. Serre, N. Audebrand, J. Marrot and G. Férey, J. Am. Chem. Soc., 2005, 127, 12788 CrossRef CAS PubMed; (f) Q. P. Li and J. J. Qian, RSC Adv., 2014, 4, 32391 RSC; (g) S. Muniappan, S. Lipstman, S. George and I. Goldberg, Inorg. Chem., 2007, 46, 5544 CrossRef CAS PubMed; (h) Q. P. Li, X. Jiang and S. W. Du, RSC Adv., 2015, 5, 1785 RSC; (i) R. Wang, and Z. Zheng, Rare Earth Complexes with Carboxylic Acids, Polyaminopolycarboxylic Acids, and Amino Acids, in Rare Earth Coordination Chemistry: Fundamentals and Applications, ed. C. Huang, John Wiley & Sons, Ltd, Chichester, UK, 2010, p. 91, DOI:10.1002/9780470824870.ch3.
- (a) Y. H. Wan, L. P. Zhang, L. P. Jin, S. Gao and S. Z. Lu, Inorg. Chem., 2003, 42, 4985 CrossRef CAS PubMed; (b) Y. H. Wan, L. P. Zhang and L. P. Jin, J. Mol. Struct., 2003, 658 Search PubMed; (c) B. L. Chen, L. B. Wang and G. D. Qian, Angew. Chem., Int. Ed., 2009, 48, 500 CrossRef CAS PubMed; (d) Y. H. Zhao, Z. M. Su, Y. M. Fu, K. Z. Shao, P. Li, Y. Wang, X. R. Hao, D. X. Zhu and S. D. Liu, Polyhedron, 2008, 27, 583 CrossRef CAS PubMed; (e) J. L. Yi, Z. Y. Fu and S. J. Liao, J. Coord. Chem., 2009, 14, 2290 CrossRef; (f) Y. Y. Lv, Y. Qi, L. X. Sun, F. Luo, Y. X. Che and J. M. Zheng, Eur. J. Inorg. Chem., 2010, 5592 CrossRef CAS; (g) Y. T. Liu, Y. Q. Du, X. Wu, Z. P. Zheng, X. M. Lin, L. C. Zhu and Y. P. Cai, CrystEngComm, 2014, 16, 6797 RSC.
- (a) J. Huang, H. M. Li, J. Y. Zhang and C. Y. Su, Inorg. Chim. Acta, 2012, 388, 16 CrossRef CAS PubMed; (b) X. T. Rao, T. Song, B. L. Chen and G. D. Qian, J. Am. Chem. Soc., 2013, 135, 15559 CrossRef CAS PubMed; (c) Q. F. Zhang, F. L. Hu, S. N. Wang and J. M. Dou, Aust. J. Chem., 2012, 65, 524 CrossRef CAS; (d) Y. G. Sun, J. Li, K. L. Li, Z. H. Xu, F. Ding, B. Y. Ren, S. J. Wang, L. X. You, G. Xiong and P. F. Smet, CrystEngComm, 2014, 16, 1777 RSC; (e) Q. P. Li and S. W. Du, RSC Adv., 2014, 4, 30963 RSC; (f) T. Song, X. T. Rao, Y. J. Cui, Y. Yang and G. D. Qian, J. Alloys Compd., 2013, 555, 22 CrossRef CAS PubMed.
- CrystalClear, version 1.36, Molecular Structure Corp and Rigaku Corp., The Woodlands, TX, and Tokyo, Japan, 2000 Search PubMed.
- G. M. Sheldrick, SHELXS 97, Program for Crystal Structure Solution, University of Göttingen, Göttingen, Germany, 1997 Search PubMed.
- Q. P. Li, J. J. Qian, C. B. Tian, P. Lin, Z. Z. He, N. Wang, J. N. Shen, H. B. Zhang, T. Chu, Y. Yang, L. P. Xue and S. W. Du, Dalton Trans., 2014, 43, 3238 RSC.
- (a) J. R. Li, Y. Tao, Q. Yu and X. H. Bu, Chem. Commun., 2007, 1527 RSC; (b) J. Y. Zou, W. Shi, J. Y. Zhang, Y. F. He, H. L. Gao, J. Z. Cui and P. Cheng, CrystEngComm, 2014, 16, 7133 RSC.
- (a) Z. H. Weng, D. C. Liu, Z. L. Chen, H. H. Zou, S. N. Qin and F. P. Liang, Cryst. Growth Des., 2009, 9, 4163 CrossRef CAS; (b) Y. J. Cui, H. Xu, Y. F. Yue, Z. Y. Guo, J. C. Yu, Z. X. Chen, J. K. Gao, Y. Yu, G. D. Qian and B. L. Chen, J. Am. Chem. Soc., 2012, 134, 3979 CrossRef CAS PubMed.
- Y. Ma, G. F. Xu, X. Yang, L. C. Li, J. K. Tang, S. P. Yan, P. Cheng and D. Z. Liao, Chem. Commun., 2010, 46, 8264 RSC.
- P. H. Lin, T. Burchell, R. Cle'ac and M. Murugesu, Angew. Chem., Int. Ed., 2008, 47, 8848 CrossRef CAS PubMed.
- H. B. Zhang, C. B. Tian, S. T. Wu, J. D. Lin, Z. H. Li and S. W. Du, J. Mol. Struct., 2011, 355 CrossRef CAS PubMed.
- (a) G. F. Xu, Q. L. Wang, P. Gamez, J. K. Tang, S. P. Yan, P. Cheng and D. Z. Liao, Chem. Commun., 2010, 46, 1506 RSC; (b) J. Tang, I. Hewitt, N. T. Madhu, G. Chastanet, W. Wernsdorfer, C. E. Anson, C. Benelli, R. Sessoli and A. K. Powell, Angew. Chem., Int. Ed., 2006, 45, 1729 CrossRef CAS PubMed.
- (a) Y. L. Hou, G. Xiong, B. Shen, B. Zhao, Z. Chen and J. Z. Cui, Dalton Trans., 2013, 42, 3587 RSC; (b) Z. R. Jhu, C. I. Yang and G. H. Lee, CrystEngComm, 2013, 15, 2456 RSC; (c) X. L. Liu, X. Wang, T. Gao, Y. Xu, X. Shen and D. R. Zhu, CrystEngComm, 2014, 16, 2779 RSC; (d) Y. L. Hou, R. R. Cheng, G. Xiong, J. Z. Cui and B. Zhao, Dalton Trans., 2014, 43, 1814 RSC; (e) X. H. Wei, L. Y. Yang, S. Y. Liao, M. Zhang, J. L. Tian, R. Y. Du and X. Liu, Dalton Trans., 2014, 43, 5793 RSC; (f) Y. Peng, C. B. Tian, Y. H. Lan, N. Magnani, Q. P. Li, H. B. Zhang, A. K. Powell and S. W. Du, Eur. J. Inorg. Chem., 2013, 5534 CrossRef CAS; (g) Y. N. Guo, G. F. Xu, Y. Guo and J. K. Tang, Dalton Trans., 2011, 40, 9953 RSC; (h) L. Ungur, S. Y. Lin, J. K. Tang and L. F. Chibotaru, Chem. Soc. Rev., 2014, 43, 6894 RSC; (i) P. Zhang, L. Zhang, C. Wang, S. F. Xue, S. Y. Lin and J. K. Tang, J. Am. Chem. Soc., 2014, 136, 4484 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Additional figures, TGA and XRD pattern. CCDC 1010482 for 1 and 958686 for 5. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra14939d |
| This journal is © The Royal Society of Chemistry 2015 |