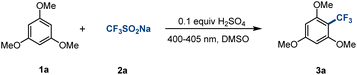An in situ generated proton initiated aromatic fluoroalkylation via electron donor–acceptor complex photoactivation†
Panyi
Huang
a,
Chun
Lv
a,
Haijing
Song
a,
Chenjing
Wang
a,
Junze
Du
a,
Jianjun
Li
 ab,
Bin
Sun
*ab and
Can
Jin
ab,
Bin
Sun
*ab and
Can
Jin
 *ab
*ab
aCollege of Pharmaceutical Sciences, Collaborative Innovation Center of Yangtze River Delta Region Green Pharmaceuticals, Zhejiang University of Technology, Hangzhou 310014, China. E-mail: jincan@zjut.edu.cn; sunbin@zjut.edu.cn
bKey Laboratory for Green Pharmaceutical Technologies and Related Equipment of Ministry of Education and Key Laboratory of Pharmaceutical Engineering of Zhejiang Province, Zhejiang University of Technology, Hangzhou 310014, PR China
First published on 14th May 2024
Abstract
The photoactivation of electron donor–acceptor complexes to obtain fluoroalkyl arenes provides an economical and environment-friendly means of directly installing fluoroalkyl groups (–CF2H, –CF3, –C4F9, –C6F13, etc.) in late-stage drug discovery. However, recent progress in the field has been constrained to the use of electron-rich arenes as electron donors due to their inherent electronic properties. In comparison, the role reversal strategy of using commercial fluoroalkyl reagents as donors and various aromatic substrates as acceptors remains an unexplored frontier. Herein, we proposed a photoactivation of electron donor–acceptor (EDA) complexes between fluoroalkyl sulfonates (donors) and in situ-generated protonated aromatic hydrocarbons (catalytic acceptors) to achieve the C–H fluoroalkylation and tandem cyclization/fluoroalkylation of more than 80 (hetero)arenes. The process relies on available reagents, avoiding the need for metals, exogenous photocatalysts, and additives.
Introduction
The distinctive effect of introducing fluorine containing groups towards lipophilicity, metabolic stability, and bioavailability enables organofluorine chemistry to play a critical role in drug development.1 Direct C(sp2)–H fluoroalkylation using abundant (hetero)aromatic resources can eliminate the requirement for pre-functionalization in traditional methods, thus offering atom-economical approaches.2 The crucial advance was made by Baran and MacMillan who pioneered the direct fluoroalkylation of (hetero)arenes using conventional oxidation of fluoroalkyl precursors by peroxide and photocatalysis to generate CF3 and CF2H shell-opening intermediates.3 However, a large portion of the trifluoromethyl radicals are consumed by side reactions, resulting in the use of super-stoichiometric fluoroalkyl reagents and peroxides to achieve desirable yields. To avoid harsh reaction conditions, the application of metal catalysts (Cu, Pt, Pd, Co, Ni),4 organic dyes (rose bengal),5 homogeneous photoconjugates of transition metals (Ru-, Ir-, and Ni-),6 and non-homogeneous semiconductor photocatalysts (g-C3N4, TiO2, CdSe)7 has been advanced for the radical aryl C–H fluoroalkylation (Scheme 1a). These procedures eliminate the necessity for using substantial quantities of peroxide through the utilization of 5–10 mol% catalyst. However, the requirement for less abundant ruthenium- and platinum-based metals, the problem of metal residues, and the cost of catalyst procurement have stimulated the search for economical, safe, and environmentally friendly fluoroalkylation strategies.The utilization of visible light to trigger the desired radical reactivity has transformed many traditional reaction pathways by virtue of its high efficiency and gentleness.8 Unfortunately, both (hetero)aromatic and conventional fluoroalkyl precursors are colorless presenting absorption curves in the ultraviolet region, which cannot independently absorb low-energy photons to trigger high-energy shell-opening intermediates in the absence of external photocatalysts. To compensate for this deficiency, recent photocatalytic protocols using electron-deficient fluoroalkyl molecules (acceptors) and additional amines (sacrificial donors) or electron-rich aromatic molecules (donors) to form colored electron donor–acceptor (EDA) complexes with smaller HOMO–LUMO gaps that can absorb lower energy radiation in the visible region compared to the individual components have recently been developed (Scheme 1b).9 Stephenson and coworkers have achieved (hetero)arene trifluoromethylation through the formation of EDA complexes between the pyridine N-oxide/trifluoroacetic anhydride adduct and arenes.10 Later, the Stephenson group converted the donor from an aromatic substrate to a catalytic amount of an exogenous donor (2-methoxynaphthalene), providing optimized trifluoromethylation conditions.11 A similar EDA pathway was recently reported by the Li group who designed various perfluoroalkyl precursors containing aryl ketones as a recognition element of the EDA receptor.12 The above EDA strategies rely on modification of fluoroalkyl precursors via preinstalling recognition fragments (acetylated pyridine N-oxide, aryl ketone, etc.). Neutral and electron-rich aromatic hydrocarbons have a high ionization potential, leading them to consistently act as donors.13 If an EDA complex can be developed employing available fluoroalkyl reagents as donors and arenes as acceptors, it would enable a trifluoromethylation pathway with role reversal.
The commercial Langlois reagent (CF3SO2Na, an inexpensive stable solid) was considered as a potential electron-donor molecule because of the existence of an appropriate leaving group (SO2) that can trigger irreversible cleavage events to form radical fragments at a rate competing with the back electron transfer (BET).14 The Mulliken theory predicts that the charge transfer (CT) bands of the complexes will be redshifted with increasing electron donation or electron acceptance.15 Therefore, a suitable reactive label needs to be sought that can enhance the electron affinity of the arenes to become acceptors. As a conceptual blueprint, the interaction of arenes (Ar) with acids to afford [Ar–H+] adducts provided inspiration.16 Owing to the high electron acceptance of protonated aromatics with reduced LUMO orbital energy,17 we envisage proton adducts as catalytic acceptors to acquire EDA complexes with CF3SO2Na through electrostatic recognition, then SEAr processes regenerate the protons to complete the cycle (Scheme 1c). Herein, we first propose an EDA complex between in situ-generated protonated aromatic hydrocarbons (catalytic acceptors) and fluoroalkyl sulfonates (donors) to achieve the fluoroalkylation of a series of (hetero)arenes. This approach avoids the need for super-stoichiometric oxidants, metals, and electron sacrificial agents, and can effectively initiate fluoroalkyl radicals through the catalytic in situ-generated protons (Scheme 1d).
Results and discussion
Considering the above factors, we made a preliminary attempt to react 1,3,5-trimethoxybenzene (1a) with CF3SO2Na (2a) after the addition of 0.1 equiv. H2SO4 under 400–405 nm irradiation, and the desired CF3-substituted product 3a was obtained in 76% yield (Table 1, entry 1). Encouraged by this result, a variety of acids were investigated for this transformation process (Table 1, entries 2–5). The experiment showed that CF3SO3H (pKa = 0.3 in DSMO) promoted the reaction best and CH3COOH (pKa = 12.6 in DMSO) worst with the same irradiation time, which was positively correlated with the ability of the acid to provide protons. When adding 2 equiv. CF3SO3H, the reaction rate is decreased as compared to 1 equiv. CF3SO3H (Table 1, entries 6 and 7), which may be due to the reduction of concentration of free aromatic hydrocarbons in the system. Surprisingly, the target product 3a was obtained in 78% yield by prolonging the reaction time without the addition of a Brønsted acid (Table 1, entry 8). The pH value, high-resolution mass spectrometry, and 19F NMR showed that 2a was transformed into CF3SO3H at a slow rate under continuous 400–405 nm irradiation in air, which leads to in situ generation of protons (see the ESI, Fig. S2–4, for details†). Considering the cost of the reagents and the environmental impact, entry 8 sacrificing some of the reaction rate but representing greener conditions, was determined to be the optimal conditions. The control experiments suggested that light was necessary for this transformation (Table 1, entry 9).| Entry | Variation from standard conditionsa | Yieldb of 3a (%) |
|---|---|---|
| a Standard conditions: 1a (0.3 mmol), 2a (1.5 equiv.), acid (0.1 equiv.), DMSO (2 mL), air, rt, 5 h under 400–405 nm irradiation. b Isolated yield. c 3 h under 400–405 nm irradiation. d 10 h under 400–405 nm irradiation. | ||
| 1 | None | 76 |
| 2 | CF3SO3H as a proton source | 79 |
| 3 | HCl as a proton source | 74 |
| 4 | H3PO4 as a proton source | 70 |
| 5 | CH3CO2H as a proton source | 67 |
| 6c | 1 equiv. CF3SO3H as a proton source | 76 |
| 7c | 2 equiv. CF3SO3H as a proton source | 67 |
| 8 | No acid | 49, 78d |
| 9 | In the dark | 0 |
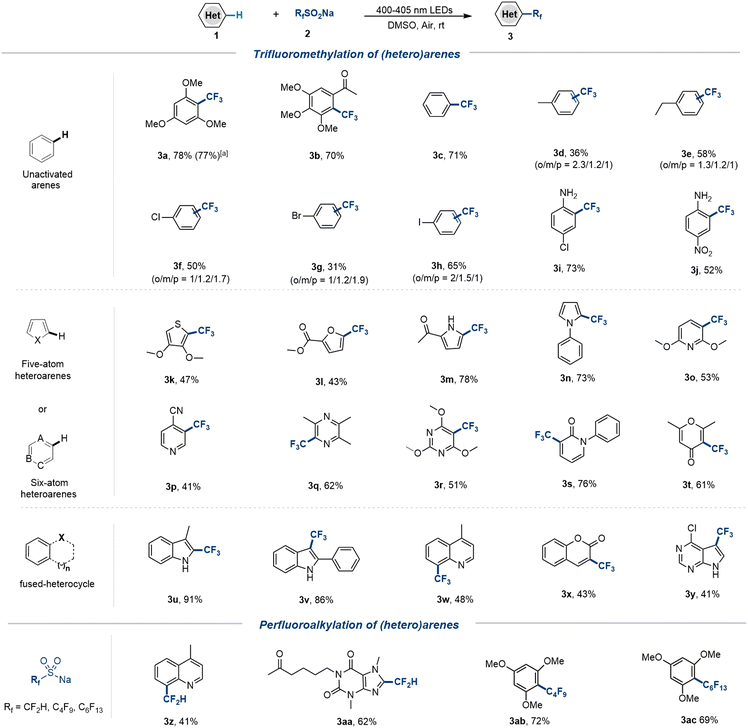
In order to further assess the scope of the protocol, C–H trifluoromethylation of other (hetero)arenes was investigated under the optimized conditions (Scheme 2). Electron-rich and neutral arenes were converted to valuable trifluoromethylated derivatives in high yields (3a–3c). Notably, alkyl aromatics, halohydrocarbons, and arylamines that are not tolerated with many reaction conditions (such as oxidants, transition metals, chemically equivalent acids, and ultraviolet light) can yield chemicals needed for industrial production (3d–3i). In addition, the electron-withdrawing groups NO2 and CN are tolerated although in a lower yield (3j, 3p). Thiophene, furan, pyrrole, pyridine, pyrazine, pyrimidine, pyridone, pyrone, and other five- and six-membered heterocycles as drug pharmacophores are compatible with the trifluoromethylation strategy (3k–3t). The indoles, quinoline, coumarin, and 7-diazapurine with various potential reaction sites displayed high regioselectivity for C–H trifluoromethylation at the most electron-rich positions (3u–3y). In addition, the application of other fluoroalkyl sulfonates to 1,3,5-trimethoxybenzene, quinoline and pentoxifylline was successful in the incorporation of perfluoroalkyl and CF2H groups that commonly act as bioequivalents of hydroxyls and amides (3z–3ac). Gratifyingly, several drugs, bioactive molecules, and hormones, including dimethyluracil 1ad (RNA base analogue), tryptophol 1ae (metabolite), heteroauxin 1af (endogenous phytohormone), flavone 1ag (vitamin P), theophylline 1ah (respiratory drug), allopurinol 1ai (uric acid lowering drug), pentoxifylline 1aj (PDE inhibitor), dibenzo-18-crown-6 1ak (phase-transfer catalyst), and estrone 1al (steroid hormone) are capable of late-stage functionalization through a simple trifluoromethylation strategy (Scheme 3). In addition, the antiviral drug trifluridine 3am can be prepared in one step by simply mixing 2′-deoxyuridine 1am with CF3SO2Na without adding any additives needed in the conventional production process. The structurally complex peptides in the presence of Gly (1an), Phe (1ao), Asp (1ap), and Lys (1aq) units afforded trifluoromethylated peptides at the C2 position of the Trp residue with retention of chirality, providing a mild and efficient tool for late-stage modifications in protein engineering.
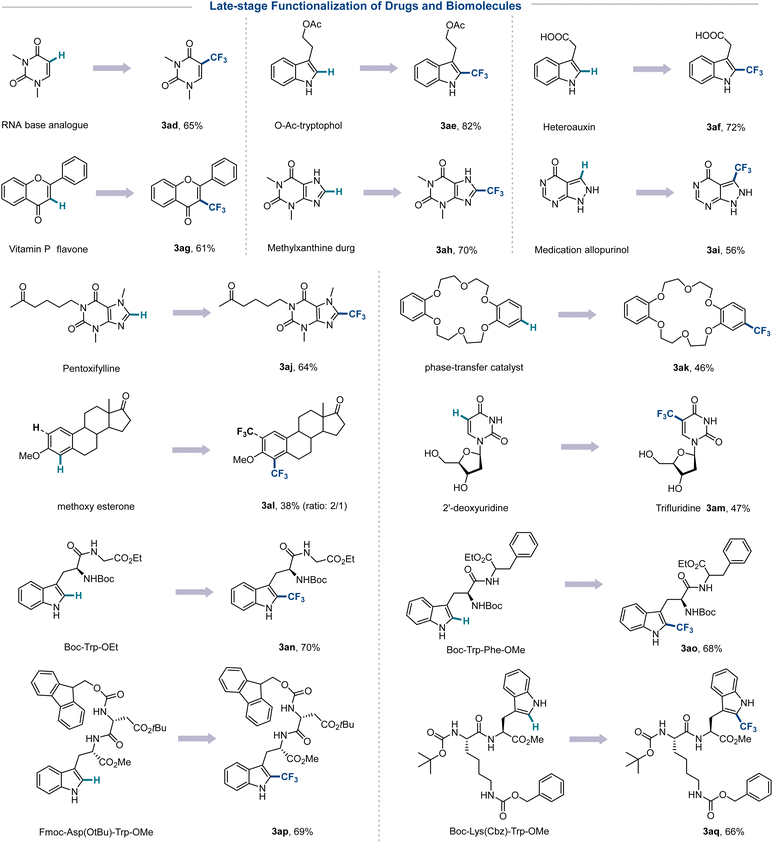 | ||
| Scheme 3 Trifluoroalkylation of drugs and biomolecules. Reaction conditions: 1 (0.30 mmol, 1.0 equiv.), 2 (0.45 mmol, 1.5 equiv.), DMSO (2.0 mL), 6–24 h, under 400–405 nm irradiation in air. | ||
Besides arene C(sp2)–H direct radical fluoroalkylation, we sought to extend the strategy to C(sp2)–H fluoroalkylation/tandem cyclization for accessing fluoroalkyl-substituted polycyclic backbones with pharmaceutical potential (Scheme 4).18 The study was initiated with benzimidazoles containing unactivated alkenes without further optimization of the previously determined reaction conditions (5a). Regardless of electronic bias, substituted benzimidazoles, containing methyl, ester, halogen, cyano, and trifluoromethyl substituents on the benzene ring, can be employed to access the polycyclic benzimidazoles 5b–5h in 70%–83% yield. Benzimidazoles disubstituted and monosubstituted at different positions can be smoothly transformed into the corresponding products (5i–5m). Moreover, pyridine, naphthalene, imidazole skeletons, and diverse branched olefins are adaptable to the current strategy (5n–5r). Inspiringly, the mild tandem cyclization strategy is also applicable to the synthesis of trifluoromethylated polycyclic indoles, dihydroisoquinolones, isoquinolinones, indolines, tricyclic oxindoles, phenanthridines, polycyclic 1,4-naphthoquinones, and chromanones (5s–5ab). Finally, C(sp2)–H perfluoroalkylation cyclization provides the target products 5ac–5ak in 68%–86% yield.
 | ||
| Scheme 4 Substrate scope of fluoroalkylation/cyclization. 4 (0.30 mmol, 1.0 equiv.), 2 (0.45 mmol, 1.5 equiv.), DMSO (2.0 mL), 12 h, under 400–405 nm irradiation in air. | ||
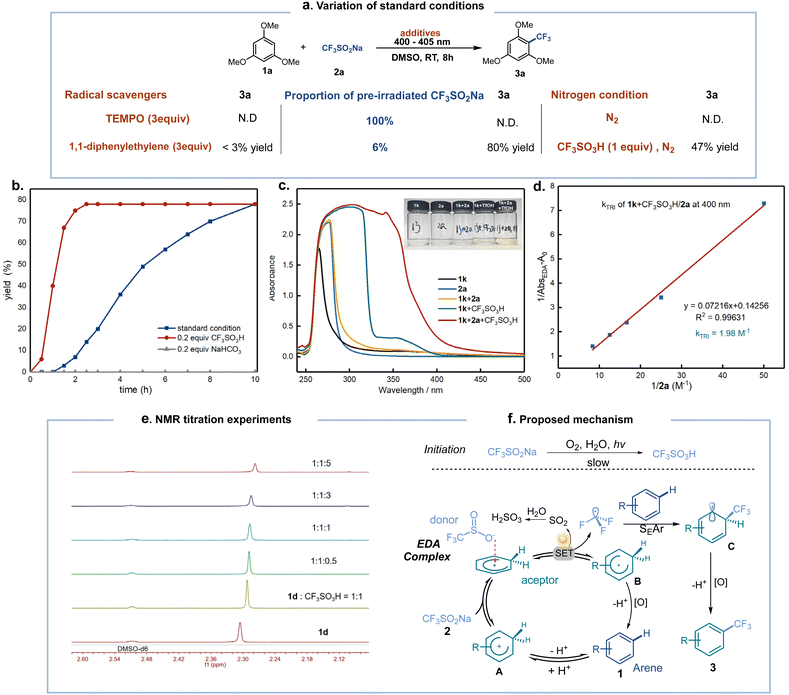
To explore the mechanism of aromatic trifluoromethylation, we have performed control experiments as shown in Scheme 5 and the ESI.† Firstly, the transformation was completely restrained upon the addition of TEMPO or 1,1-diphenylethylene radical scavengers under the standard conditions, and the captured intermediates were detected by ESI-MS or 19F NMR, which indicated the formation of trifluoromethyl radicals (Scheme 5a). When the template reaction was performed with CF3SO2Na which was irradiated for 48 h the desired product could not be obtained. However, if the proportion of CF3SO2Na pre-irradiated was decreased to 6%, the reaction could proceed smoothly to deliver 3a in 80% yield in 6 h, which is consistent with the conclusion that CF3SO3H is formed from 2a when irradiated in air. Subsequently, the desired product 3a was not observed when the template reaction was irradiated under a nitrogen atmosphere. However, the standard protocol gave 3a in 47% yield under a nitrogen atmosphere with the addition of 1 equiv. CF3SO3H. These findings support the key role of oxygen in promoting the in situ formation of protons from 2a. Next, adding CF3SO3H showed a faster rate mainly due to eliminating the induction period for the in situ formation of protonated aromatics. In contrast, the addition of NaHCO3 strongly inhibited the reaction, and no product was provided even after extending the reaction time to 24 h (Scheme 5b). To clarify the effect of protons in the process, the individual and mixed fractions were analyzed by UV/vis absorption spectroscopy (Scheme 5c). The thiophene (1k) or CF3SO3Na (2a) only displayed absorption curves in the UV region, while the redshift of the mixed solution of 1k and CF3SO3H demonstrates the possible formation of protonated aromatic hydrocarbons (green line). The absorption band tail of the mixture of 1k, 2a, and CF3SO3H extends to the visible region (red line) compared with the mixture of 1k and 2a (yellow line), indicating that the acid promotes the formation of an EDA complex between the aromatic hydrocarbons and the fluoroalkyl sulfonate. The same phenomenon was obtained by performing UV/visible absorption experiments on benzene (1c) (for details, see the ESI, Fig. S9†). Moreover, the existence of the EDA complex was also verified using Benesi–Hildebrand experiments with an association constant of 1.98 M−1 in DMSO (Scheme 5d). NMR titration experiments were carried out and the 1H NMR signal was gradually shifted downfield as 2a was increased in the presence of CF3SO3H and toluene (1d) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (Scheme 5e). The quantum yield (Φ = 0.14) determined by chemical actinometry and on/off experiments preclude a chain radical mechanism, as shown in Fig. S16.† Overall, these studies revealed the presence of protonated aromatic hydrocarbons generated in situ and that they play a crucial role in the formation of EDA complexes.
1 ratio (Scheme 5e). The quantum yield (Φ = 0.14) determined by chemical actinometry and on/off experiments preclude a chain radical mechanism, as shown in Fig. S16.† Overall, these studies revealed the presence of protonated aromatic hydrocarbons generated in situ and that they play a crucial role in the formation of EDA complexes.
Based on the above experiments, a proposed mechanism is depicted in Scheme 5f. The reaction is initiated by the formation of CF3SO3H with 2a and H2O under 400–405 nm light irradiation. Aromatic hydrocarbons then undergo protonation to form EDA complexes with 2a. The charge transfer event within the complex leads to the generation of trifluoromethyl radicals with the release of sulfur dioxide under 400–405 nm irradiation. In the presence of free aromatic hydrocarbons, the former trifluoromethyl radical can be efficiently captured to create a radical electrophilic adduct C. Subsequently, the oxidation and deprotonation of the adducts provides the trifluoromethyl-substituted arene and regenerates the proton to activate another aromatic substrate.
Conclusions
In conclusion, EDA complexes of in situ-generated protonated aromatic hydrocarbons with fluoroalkyl sulfonates are proposed to achieve C–H fluoroalkylation and tandem cyclization/fluoroalkylation of heteroaromatic hydrocarbons. Differing from the previous approach of aromatics as donors and fluoroalkyl reagents as acceptors, a simple activation program enabled a strategy for role reversal. This versatile activation mode of EDA complexes was presented for the first time. Various (hetero)arenes are used to construct fluorinated motifs in a green and efficient way without exogenous photocatalysts and additives. The practicability of this protocol is also emphasized by the late-stage functionalization of bioactive molecules.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 22078299 and 22108250) and Zhejiang Provincial Natural Science Foundation of China (LY21B060005, LQ20B060007). We are also grateful to the College of Pharmaceutical Sciences, Zhejiang University of Technology and Collaborative Innovation Center of Yangtze River Delta Region Green Pharmaceuticals for the financial help.References
- (a) C. Alonso, E. M. D. Marigorta, G. Rubiales and F. Palacios, Chem. Rev., 2015, 115, 1847 CrossRef CAS PubMed; (b) C. Le, T. Q. Chen, T. Liang, P. Zhang and D. W. C. MacMillan, Science, 2019, 360, 1010 CrossRef PubMed; (c) F. Ye, F. Berger, H. Jia, J. Ford, A. Wortman, J. Bçrgel, C. Genicot and T. Ritter, Angew., Chem. Int. Ed., 2019, 58, 14615 CrossRef CAS PubMed; (d) W.-Q. Hu, S. Pan, X.-H. Xu, D. A. Vicic and F.-L. Qing, Angew. Chem., Int. Ed., 2020, 59, 16076 CrossRef CAS PubMed; (e) J. Yang, S. Zhu, F. Wang, F.-L. Qing and L. Chu, Angew. Chem., Int. Ed., 2021, 60, 4300 CrossRef CAS PubMed; (f) Z.-J. Shen, C. Zhu, X. Zhang, C. Yang, M. Rueping, L. Guo and W. Xia, Angew. Chem., Int. Ed., 2023 DOI:10.1002/ange.202217244.
- (a) A. G. O'Brien, A. Maruyama, Y. Inokuma, M. Fujita, P. S. Baran and D. G. Blackmond, Angew. Chem., Int. Ed., 2014, 53, 11868 CrossRef PubMed; (b) Y. Pang, J. W. Lee, K. Kubota and H. Ito, Angew. Chem., Int. Ed., 2020, 59, 22570 CrossRef CAS PubMed; (c) H. Jia, A. P. Häring, F. Berger, L. Zhang and T. Ritter, J. Am. Chem. Soc., 2021, 143, 7623 CrossRef CAS PubMed; (d) H. Xiao, Z. Zhang, Y. Fang, L. Zhu and C. Li, Chem. Soc. Rev., 2021, 50, 6308 RSC; (e) J. Shen, J. Xu, L. He, C. Liang and W. Li, Chin. Chem. Lett., 2022, 33, 1227 CrossRef CAS.
- (a) Y. Fujiwara, J. A. Dixon, F. O'Hara, E. D. Funder, D. D. Dixon, R. A. Rodriguez, R. D. Baxter, B. Herlé, N. Sach, M. R. Collins, Y. Ishihara and P. S. Baran, Nature, 2012, 492, 95 CrossRef CAS PubMed; (b) Y. Fujiwara, J. A. Dixon, R. A. Rodriguez, R. D. Baxter, D. D. Dixon, M. R. Collins, D. G. Blackmond and P. S. Baran, J. Am. Chem. Soc., 2012, 134, 1494 CrossRef CAS PubMed; (c) D. A. Nagib and D. W. C. MacMillan, Nature, 2011, 480, 224–227 CrossRef CAS PubMed.
- (a) K. Natte, R. V. Jagadeesh, L. He, J. Rabeah, J. Chen, C. Taeschler, S. Ellinger, F. Zaragoza, H. Neumann, A. Brȕckner and M. Beller, Angew. Chem., Int. Ed., 2016, 55, 2782 CrossRef CAS PubMed; (b) S. Deolka, R. Govindarajan, E. Khaskin, R. R. Fayzullin, M. C. Roy and J. R. Khusnutdinova, Angew. Chem., Int. Ed., 2021, 60, 24620 CrossRef CAS PubMed; (c) H.-R. Zhang, C.-C. Feng, N. Chen and S.-L. Zhang, Angew. Chem., Int. Ed., 2023 DOI:10.1002/ange.202209029; (d) S. Deolka, R. Govindarajan, T. Gridneva, M. C. Roy, S. Vasylevskyi, P. K. Vardhanapu, J. R. Khusnutdinova and E. Khaskin, ACS Catal., 2023, 13, 13127 CrossRef CAS; (e) C. S. Kuehner, A. G. Hill, C. F. Harris, C. A. Owens, J. Bacsa and J. D. Soper, ACS Catal., 2023, 13, 13607 CrossRef CAS PubMed.
- (a) L. Cui, Y. Matusaki, N. Tada, T. Miura, B. Uno and A. Itoh, Adv. Synth. Catal., 2013, 355, 2203 CrossRef CAS; (b) S. P. Pitre, C. D. McTiernan, H. Ismaili and J. C. Scaiano, ACS Catal., 2014, 4, 2530 CrossRef CAS; (c) W. Zhang, X.-X. Xiang, J. Chen, C. Yang, Y.-L. Pan, J.-P. Cheng, Q. Meng and X. Li, Nat. Commun., 2020, 11, 638 CrossRef CAS PubMed.
- (a) J. W. Beatty, J. J. Douglas, K. P. Cole and C. R. J. Stephenson, Nat. Commun., 2015, 6, 7919 CrossRef PubMed; (b) B. Yang, D. Yu, X.-H. Xu and F.-L. Qing, ACS Catal., 2018, 8, 2839 CrossRef CAS; (c) L. Trump, A. Lemos, B. Lallemand, P. Pasau, J. Mercier, C. Lemaire, A. Luxen and C. Genicot, Angew. Chem., Int. Ed., 2019, 58, 13149 CrossRef CAS PubMed.
- (a) J. Lin, Z. Li, J. Kan, S. Huang, W. Su and Y. Li, Nat. Commun., 2017, 8, 14353 CrossRef PubMed; (b) I. Ghosh, J. Khamrai, A. Savateev, N. Shlapakov, M. Antonietti and B. König, Science, 2019, 365, 360 CrossRef CAS PubMed; (c) K. Muralirajan, R. Kancherla, J. A. Bau, M. R. Taksande, M. Qureshi, K. Takanabe and M. Rueping, ACS Catal., 2021, 11, 14772 CrossRef CAS.
- (a) Y. Zhao and W. Xia, Chem. Soc. Rev., 2018, 47, 2591 RSC; (b) M. Latrache and N. Hoffmann, Chem. Soc. Rev., 2021, 50, 7418 RSC; (c) A. R. Allen, E. A. Noten and C. R. J. Stephenson, Chem. Rev., 2022, 122, 2695 CrossRef CAS PubMed; (d) L. Chang, Q. An, L. Duan, K. Feng and Z. Zuo, Chem. Rev., 2022, 122, 2429 CrossRef CAS PubMed; (e) A. Y. Chan, B. Perry, N. B. Bissonnette, B. F. Buksh, G. A. Edwards, L. I. Frye, O. L. Garry, M. N. Lavagnino, B. X. Li, Y. Liang, E. Mao, A. Millet, J. V. Oakley, N. L. Reed, H. A. Sakai, C. P. Seath and D. W. C. MacMillan, Chem. Rev., 2022, 122, 1485 CrossRef CAS PubMed.
- (a) R. Wang, W. Guan, Z.-B. Han, F. Liang, T. Suga, X. Bi and H. Nishide, Org. Lett., 2017, 19, 2358 CrossRef CAS PubMed; (b) R. Wang, L. Wang, Q. Xu, B.-Y. Ren and F. Liang, Org. Lett., 2019, 21, 3072 CrossRef CAS PubMed; (c) X. Liang, Y. Li, Q. Xia, L. Cheng, J. Guo, P. Zhang, W. Zhang and Q. Wang, Green Chem., 2021, 23, 8865 RSC; (d) A. Dewanji, L. Dalsen, J. A. Rossi-Ashton, E. Gasson, G. E. M. Crisenza and D. J. Procter, Nat. Chem., 2023, 15, 43 CrossRef CAS PubMed; (e) H. Wang, J. Wu, A. Noble and V. K. Aggarwal, Angew. Chem., Int. Ed., 2022 DOI:10.1002/ange.202202061; (f) L. Dalsen, E. Brown, J. A. Rossi-Ashton and D. J. Procter, Angew. Chem., Int. Ed., 2023 DOI:10.1002/anie.202303104; (g) A. K. Wortman and C. R. J. Stephenson, Chem, 2023, 9, 1 CrossRef PubMed.
- J. W. Beatty, J. J. Douglas, R. Miller, R. C. McAtee, K. P. Cole and C. R. J. Stephenson, Chem, 2016, 1, 456 CAS.
- E. J. McClain, T. M. Monos, M. Mori, J. W. Beatty and C. R. J. Stephenson, ACS Catal., 2020, 10, 12636 CrossRef CAS.
- D. J. Castillo-Pazos, J. D. Lasso, E. Hamzehpoor, J. Ramos-Sánchez, J. M. Salgado, G. Cosa, D. F. Perepichka and C.-J. Li, Chem. Sci., 2023, 14, 3470 RSC.
- (a) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461 CrossRef CAS PubMed; (b) M. R. Lasky, T. K. Salvador, S. Mukhopadhyay, M. S. Remy, T. P. Vaid and M. S. Sanford, Angew. Chem., Int. Ed., 2022 DOI:10.1002/anie.202208741.
- (a) Q.-Y. Meng, N. Dçben and A. Studer, Angew. Chem., Int. Ed., 2020, 59, 19956 CrossRef CAS PubMed; (b) Y. Jiang, B. Li, N. Ma, S. Shu, Y. Chen, S. Yang, Z. Huang, D. Shi and Y. Zha, Angew. Chem., Int. Ed., 2021, 60, 19030 CrossRef CAS PubMed.
- R. S. Mulliken, J. Am. Chem. Soc., 1952, 74, 811 CrossRef CAS.
- (a) Z. Huang, M. Shanmugam, Z. Liu, A. Brookfield, E. L. Bennett, R. Guan, D. E. V. Herrera, J. A. Lopez-Sanchez, A. G. Slater, E. J. L. McInnes, X. Qi and J. Xiao, J. Am. Chem. Soc., 2022, 144, 6532 CrossRef CAS PubMed; (b) J. Wu, J. Chen, L. Wang, H. Zhu, R. Liu, G. Song, C. Feng and Y. Li, Green Chem., 2023, 25, 940 RSC.
- W. Liu, X. Yang, Z.-Z. Zhou and C.-J. Li, Chem, 2017, 2, 688 CAS.
- (a) J. Xuan and A. Studer, Chem. Soc. Rev., 2017, 46, 4329 RSC; (b) Y. Yuan, Y. Zheng, B. Xu, J. Liao, F. Bu, S. Wang, J.-G. Hu and A. Lei, ACS Catal., 2020, 10, 6676 CrossRef CAS; (c) S. Zheng, Á. Gutiérrez-Bonet and G. A. Molander, Chem, 2019, 5, 339 CrossRef CAS PubMed; (d) Q.-W. Gui, F. Teng, P. Yu, Y.-F. Wu, Z.-B. Nong, L.-X. Yang, X. Chen, T.-B. Yang and W.-M. He, Chin. J. Catal., 2023, 44, 111 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc00728j |
| This journal is © The Royal Society of Chemistry 2024 |