Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Oxidation-derived anticancer potential of sumanene–ferrocene conjugates†
Artur
Kasprzak
 *a,
Agnieszka
Zuchowska
*a,
Agnieszka
Zuchowska
 a,
Pawel
Romanczuk
a,
Pawel
Romanczuk
 a,
Agata
Kowalczyk
a,
Agata
Kowalczyk
 b,
Ireneusz P.
Grudzinski
b,
Ireneusz P.
Grudzinski
 c,
Anna
Malkowska
c,
Anna
Malkowska
 c,
Anna M.
Nowicka
c,
Anna M.
Nowicka
 b and
Hidehiro
Sakurai
b and
Hidehiro
Sakurai
 de
de
aFaculty of Chemistry, Warsaw University of Technology, Noakowskiego Str. 3, 00-664 Warsaw, Poland. E-mail: artur.kasprzak@pw.edu.pl
bFaculty of Chemistry, University of Warsaw, Pasteura Str. 1, 02-093 Warsaw, Poland
cFaculty of Pharmacy, Medical University of Warsaw, Banacha Str. 1, 02-097 Warsaw, Poland
dDivision of Applied Chemistry, Graduate School of Engineering, Osaka University, 2-1 Yamadaoka, Suita, 565-0871 Osaka, Japan
eInnovative Catalysis Science Division, Institute for Open and Transdisciplinary Research Initiatives (ICS-OTRI), Osaka University, Suita, Osaka 565-0871, Japan
First published on 30th November 2023
Abstract
An effective synthetic protocol towards the oxidation of sumanene–ferrocene conjugates bearing one to four ferrocene moieties has been established. The oxidation protocol was based on the transformation of FeII from ferrocene to FeIII-containing ferrocenium cations by means of the treatment of the title organometallic buckybowls with a mild oxidant. Successful isolation of these ferrocenium-tethered sumanene derivatives 5–7 gave rise to the biological evaluation of the first, buckybowl-based anticancer agents, as elucidated by in vitro assays with human breast adenocarcinoma cells (MDA-MB-231) and embryotoxicity trials in zebrafish embryos supported with in silico toxicology studies. The designed ferrocenium-tethered sumanene derivatives featured attractive properties in terms of their use in cancer treatments in humans. The tetra-ferrocenium sumanene derivative 7 featured especially beneficial biological features, elucidated by low (<40% for 10 μM) viabilities of MDA-MB-231 cancer cells together with a 1.4–1.7-fold higher viability of normal cells (human mammary fibroblasts, HMF) for respective concentrations. Compound 7 featured significant cytotoxicity against cancer cells thanks to the presence of sumanene and ferrocenium moieties; the latter motif also provided the selectivity of anticancer action. The biological properties of 7 were also improved in comparison with those of native building blocks, which suggested the effects of the presence of the sumanene skeleton towards the anticancer action of this molecule. Ferrocenium-tethered sumanene derivatives exhibited potential towards the generation of reactive oxygen species (ROS), responsible for biological damage to the cancer cells, with the most efficient generation of the tetra-ferrocenium sumanene derivative 7. Derivative 7 also did not show any embryotoxicity in zebrafish embryos at the tested concentrations, which supports its potential as an effective and cancer-specific anticancer agent. In silico computational analysis also showed no chromosomal aberrations and no mutation with AMES tests for the compound 7 tested with and without microsomal rat liver fractions, which supports its further use as a potent drug candidate in detailed anticancer studies.
Introduction
Sumanene (1; see Fig. 1a) is a bowl-shaped buckybowl compound that was first synthesized in 2003.1 The possibilities of (i) functionalization of sumanene at the benzylic and aromatic positions,2–5 as well as (ii) the generation of sumanenyl carbanions6,7 were demonstrated. As the result, various organometallic complexes comprising sumanene were synthesized, and these derivatives can be grouped into two general classes. The first class of compounds employed sumanene itself as a ligand, with organometallic complexes containing ruthenium,8,9 iron,9–11 or zirconium12 as representative examples. The second class of organometallic complexes comprising sumanene are the derivatives in which the structural moiety attached to the sumanene skeleton plays a role of a ligand. This approach for molecule design was recently employed for the synthesis of ruthenium complexes containing sumanene molecules functionalized with a pincer ligand13 or sumanene-containing organic capsules.14 Ferrocene (Fc)-containing sumanene derivatives can be regarded as an interesting class of sumanene organometallic congeners, not only in the field of structural chemistry buckybowls, but also towards their applied sciences. For example, recent studies revealed that such conjugates can be applied in cesium cation selective voltammetric sensors.15–18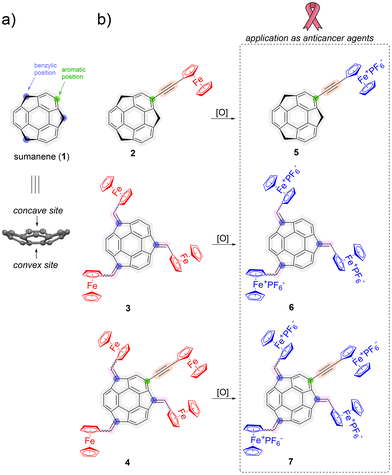 | ||
| Fig. 1 (a) Structure of sumanene (1); (b) sumanene derivatives 2–7 investigated herein, together with the graphical representation of the aims of this work. | ||
Fc-tethered compounds have been widely explored over the years in different areas of applications. This is because Fc is an air-stable and easy to modify molecule with various prospective applications.19–21 Cancer research is one of the most important applications of Fc containing systems.19,20,22–25 Interestingly, the derivatives comprising ferrocenium cations (Fc+) were reported to be attractive and effective anticancer agents. First reports are dated back to 1984, when Köpf-Maier and co-workers reported the anticancer action of Fc+ salts.26 While Fc did not feature significant toxicity towards cancer cells, Fc+ inhibited cancer cell growth, which was further supported by other studies.27,28 Detailed studies on this topic revealed that Fc+ features the improved properties of generating reactive oxygen species, such as OH˙, which in consequence lead to the formation of radical metabolites responsible for biological damage to the cells, e.g., oxidative DNA damage.27–30 Despite these beneficial biological properties of Fc+ containing systems, the reports on their use in cancer treatments are limited, mostly because of difficulties in the synthesis of such a class of compounds.
Due to the origin of buckybowls from carbon allotropes, whose use in cancer nanotechnology has been widely studied over the years,31–33 prospective biological applications of buckybowls can be considered. However, to the best of our knowledge, the reported studies on applications of buckybowls in medicinal chemistry are extremely sparse. The reports only include corannulene derivatives as cholera toxin inhibitors34 and click chemistry derived corannulene glycoconjugates as cancer-specific molecules.35 The potential biological behaviors of sumanene derivatives only include recent (2021–2023) theoretical studies36,37 on the prospective use of sumanene molecules in drug delivery. Interestingly, in these computational works, the bowl-to-bowl inversion property of sumanene molecules was considered as the driving force towards the design of effective drug delivery systems. Therefore, inspired by the promising anticancer effects of Fc+-tethered compounds as well as highly limited reports on the biological applications of functionalized buckybowls, herein, in pursuit of gaining new knowledge on the synthesis, properties and applications of sumanene derivatives, we report for the first time the synthesis and the assessment of the anticancer potential of sumanene-derivatives 5–7 bearing various numbers of Fc+ units (from one to four, see Fig. 1b). FeIII-containing derivatives 5–7 were obtained via the treatment of ferrocene (FeII) containing compounds 2–4 with a mild oxidant. Considering that the most common cancer worldwide is breast cancer,38,39 cytotoxicity profiles were estimated in vitro against human breast adenocarcinoma cells (MDA-MB-231) and human mammary fibroblasts (HMF) as normal cells. As a result of this work, a promising class of metallocene-tethered buckybowl-based anticancer agents was established.
Results and discussion
Synthesis and characterization of sumanene–ferrocene conjugates 2–4
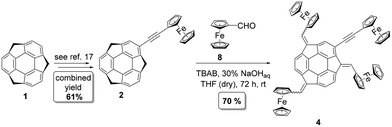
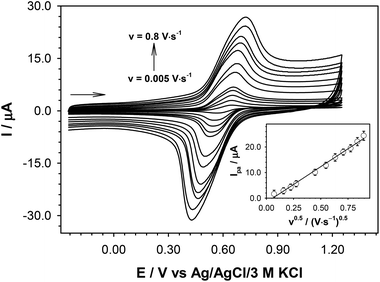
A Materials and methods section, as well as full experimental details, can be found in ESI, Subsection S1.2† (synthetic procedures are also listed below in the Experimental section). 2-(Ferrocenylethynyl)sumanene (2)17 and tris(ferrocenylmethidene)sumanene (3)16 (Fig. 1b) were synthesized according to the literature protocols. The synthesis of the tetraferrocenylated sumanene derivative 4 (Fig. 2) was based on the condensation-type reaction16 and employed formylferrocene (8) and compound 217 as the starting materials. Notably, the reported procedures for the syntheses of tris-substituted sumanene derivatives employing the condensation-type reaction16,18,40–42 to date only employed native sumanene as the starting material. Our successful experiment with 4 demonstrated that it is possible to subject a sumanene derivative functionalized at the aromatic carbon to this process. The formation of compound 4 was confirmed by NMR spectroscopy, as well as high-resolution mass spectrometry (HRMS). Copies of the NMR spectra and the respective discussion on 4 can be found in ESI, Section S2,† whilst the HRMS spectra are presented in ESI, Section S3.†A profile of the UV-vis spectrum of 4 was different from that for the parent 2 and was similar to the respective spectrum of 3, with the three major absorption maxima (λmax) between 245 and 535 nm (the UV-Vis and emission spectra of 4 are presented in ESI, Section S4†). Emission spectroscopy studies revealed that the compound 4 is a red-light emitter (λem = 640 nm). The slightly lower emission intensity of 4 in comparison with that of 3 was ascribed to the fact that Fc is an emission (excited states) quencher.43,44 Cyclic voltammetry experiments with 4 revealed one pair of current signals related to the reversible Fc redox process (Fig. 3). This suggested that the Fc units in 4 might not be electronically communicated. Since the intensity of the Fc electrooxidation signal varied linearly versus the square root of the scan rate, it can be concluded that the rate-limiting step in the electrode reaction was the diffusion of the depolarizer to the electrode surface.45
Taking into account that sumanene–ferrocene conjugates have been recently reported to feature caesium cation (Cs+) selective trapping properties,15–18 which were based on the non-covalent cation–π interactions between Cs+ and the concave site of the sumanene bowl,7,15,46 the prospective use of compound 4 as the Cs+ recognition material was investigated using emission spectroscopy and voltammetry. The detailed studies on this topic are discussed in ESI, Sections S5 (spectroscopy) and S6 (electrochemistry).† In brief, despite being structurally sophisticated, the tetrasubstituted sumanene derivative 4 displayed a property of selective interaction with Cs+. The estimated limit of detection value for the constructed voltammetric sensor (45 nM) was not as satisfactory as for the monoferrocenyl sumanenes (6–9 nM) and compound 3 (20 nM; see the comparison data in ESI, Section S6†). This trend could be ascribed to the steric factors towards trapping Cs+ by two sumanene bowls within 4.
Oxidation of sumanene–ferrocene conjugates 2–4 to the respective products 5–7 bearing ferrocenium cations
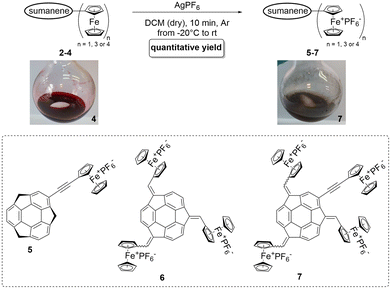
Voltammetric analyses revealed that compounds 2,17316 and 4 are electroactive, and this electroactivity results from the presence of Fc units in these molecules. Taking into account the demonstrated redox activity of Fc-containing systems,19,47–49 we hypothesized that it is possible to oxidize the Fc units in the structures of compounds 2–4 into the respective Fc+, leading to the creation of buckybowls bearing Fc+ moieties, namely compounds 5–7 (Fig. 4). The commonly used oxidants for the oxidation of FeII to FcIII (oxidation of Fc to Fc+) include, but are not limited, to nitrosonium and nitronium salts,50 hypochlorite salts,51 benzoquinone and its derivatives,52–54 or iron(III) salts.49,52,55,56To oxidize sumanene–Fc conjugates 2–4 to the Fc+-containing compounds 5–7, silver(I) hexafluorophosphate (AgPF6) was selected as an easy-to-handle oxidant, and the properties of oxidizing Fc to ferrocenium hexafluorophosphate (FcPF6) in dichloromethane (DCM) solutions were demonstrated.57 The reaction scheme for synthesizing compounds 5–7 is presented in Fig. 4. Full experimental details can be found in ESI, Sections S1–S4.† In brief, the dropwise addition of a DCM solution of AgPF6 at −20 °C to a solution of 2–4 resulted in a significant colour change, as well as the formation of a brownish precipitate, as visualized in Fig. 4 for compound 7.58 Such behaviours were reported as typical of oxidizing Fc and its derivatives.48,49,53,57,59 The syntheses of compounds 5–7 were chromatography-free, since the products were isolated by filtration and washing with ethanol (EtOH) and DCM. The reaction yields of 5–7 were quantitative. The oxidation of 2–4 to 5–7 resulted in significant changes in the solubility profiles. While Fc-containing starting materials 2–4 were well-soluble in many commonly used organic solvents, including chloroalkanes (DCM and CHCl3), aromatics (PhMe and PhCl), and tetrahydrofuran (THF) and insoluble in dimethylsulfoxide (DMSO), Fc+-containing products 5–7 were soluble in acetone and DMSO.
The formation of compounds 5–7 was confirmed by NMR spectroscopy, HRMS and elemental analysis. Copies of the NMR spectra of 5–7 and the respective discussion can be found in ESI, Section S2,† whilst the HRMS spectra of 5–7 are presented in Section S3.† In brief, the NMR spectra of 5–7 measured in deuterated acetone ((CD3)2CO) featured broad signals with chemical shift (δH) values higher than 15 ppm. This feature was ascribed to the presence of paramagnetic Fc+ units in the structures of compounds 5–7, which is consistent with the literature data for the simple Fc+-containing molecules.48,57,60 The signal coming from the presence of PF6− units in 5–7 (δF from −72.96 ppm to −77.70 ppm) was also clearly detected by 19F NMR experiments.
The change in the oxidation state of iron (from FeII to FeIII) also resulted in the changes in the UV-vis spectrum of Fc+-containing 5–7 in comparison with Fc-tethered stating materials 2–4, which is in a good agreement with the absorption spectral studies on the oxidation of simple Fc and Fc+ containing systems (see UV-Vis spectra of 5–7 in ESI, Subsection S4.1†).48,53,61,62 The UV-vis analyses in acetone–water solvent mixtures revealed that among the tested Fc+-containing sumanene derivatives 5–7, the water solubility of compound 7 (tetracation) is the most satisfactory (see spectra and discussion in ESI, Subsection S4.2†).
Application of compounds 5–7 as anticancer agents
Full details on biological assays can be found in ESI, Subsection S1† (key-procedure details are also listed below in the Experimental section).We anticipated that due to the presence of Fc+ units in molecules 5–7, these compounds might feature attractive cytotoxicity against cancer cells. Considering that the most common cancer worldwide is breast cancer,38,39 cytotoxicity profiles were estimated in vitro against human breast adenocarcinoma cells (MDA-MB-231) as the representative breast cancer cell line, together with the respective studies with human mammary fibroblasts (HMF) as normal cells. The results of biological assays with Fc+-containing 5–7 are presented in Fig. 5. The cytotoxicity profiles of native sumanene (1), Fc, FcPF6, as well as patent Fc-containing mono-, tris- and tetra-ferrocenylated sumanene derivatives 2–4 are also presented for the reference. The biological assays were performed for three concentrations, namely 10, 50 and 100 μM. In general, for all tested compounds lower cell viabilities (after 24 hours) were observed for higher concentrations (50 and 100 μM). In the used model, the most essential differences in cell viabilities between the tested compounds were found for the lowest concentration, i.e., 10 μM; thus, cell viabilities for this concentration were selected for most of the discussion on differences in the cytotoxicity profiles of the tested compounds.
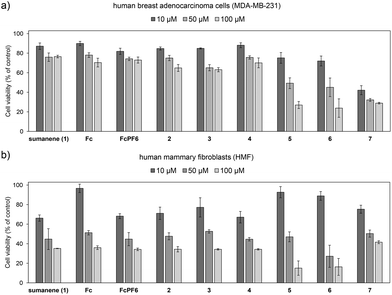 | ||
| Fig. 5 Cell viabilities (after 24 hours) after treatment with sumanene (1), Fc, FcPF6 and compounds 2–7: (a) MDA-MB-231 (breast cancer) cells and (b) human mammary fibroblasts (HMF, normal cells). | ||
In the used model, native Fc featured a slight cytotoxic effect towards MDA-MB-231 cancer cells (cell viability: ca. 94% for 10 μM), whilst the cell viability of HMF normal cells was similar to that of the control (ca. 97% for 10 μM). FcPF6 featured improved cytotoxicity against MDA-MB-231 cancer cells (cell viability: ca. 75% for 10 μM) in comparison with Fc. HMF normal cell viability after the treatment with FcPF6 equalled ca. 68% for 10 μM. Sumanene (1) featured slightly lower cytotoxicity against MDA-MB-231 cancer cells (cell viability ca. 84% for 10 μM) in comparison with FcPF6, whilst cytotoxicity against HMF normal cells was similar to that of FcPF6. It means that FcPF6 featured better cancer-specific cytotoxic action than sumanene (1). In the case of ferrocene-containing sumanene derivatives 2–4, cell viabilities of MDA-MB-231 cancer cells were at the level of ca. 82–85% for 10 μM, which means that these molecules featured cytotoxicity profiles similar to sumanene (1) and improved cytotoxicity in comparison with native Fc. The cell viability of HMF normal cells after the treatment with 2–4 ranged from ca. 67 to ca. 77% for 10 μM.
Significant differences between the cytotoxicity profiles of Fc+-containing sumanene derivatives 5–7versus Fc, sumanene (1) and parent Fc derivatives 2–4 were observed. The cytotoxicity profiles of compounds 5–7 towards MD-MB-231 cells were improved in comparison with those of not only the parent compounds 2–4, but also native sumanene (1). This suggested that the presence of Fc+ units attached to the sumanene skeleton resulted in the improved anticancer potential of these molecules, associated with the oxidation state of iron. Compounds 5–7 featured higher cytotoxicity towards MDA-MB-231 cancer cells (ca. 23–70%) than other tested compounds, also including FcPF6. This was especially observed for higher concentrations (ca. 32–49% for 50 μM and ca. 23–26% for 100 μM). Additionally, the satisfactory cancer-cell specific cytotoxic action of 5–7 was observed for the concentration of 10 μM, since at this concentration the viabilities of HMF normal cells were ca. 1.3–1.8-fold higher in comparison with the viability of MDA-MB-231 cancer cells.
Notably, the cytotoxicity profile for the Fc+-containing sumanene derivative 7 was the most promising among all compounds tested. For the 10 μM concentration of compounds 5 and 6, MDA-MB-231 cancer cell viability equaled ca. 72–77% and HMF normal cell viabilities were satisfactory (ca. 88%); however, compounds 5 and 6 were found to be relatively toxic for both cells at higher concentrations (50–100 μM). Compound 7 featured a significant cytotoxic effect towards MDA-MB-231 cancer cells for all tested concentrations (cell viability < 42%). Importantly, for the most beneficial concentration of 10 μM, MDA-MB-231 cancer cell viability after the treatment with 7 was ca. 40% and HMF normal cell viability (ca. 75%) was higher than those for FcPF6, sumanene (1) and parent compound 4. It can be concluded that compound 7 featured significant cytotoxicity against cancer cells due to the presence of sumanene and Fc+ moieties. The Fc+ motif also provided the selectivity of the anticancer action of the compound 7. The biological properties of 7 were also improved in comparison with that of native building blocks, which suggested the effects of the presence of the sumanene skeleton on the anticancer action of this molecule. Similar beneficial cytotoxicity profiles of 7 could be also observed for higher concentrations. In other words, for the respective concentrations of compound 7, the viability of HMF normal cells was 1.4–1.7-fold higher in comparison with that of MDA-MB-231 cancer cells. In consequence, compound 7 not only featured the best cytotoxicity against cancer cells among all compounds tested, but also the best selectivity of cytotoxic action. Taking into account the above-noted improved cancer-specific action of FcPF6 in comparison with that of native sumanene (1), one might state that these beneficial cytotoxicity profiles of the sumanene buckybowl derivative 7 might be related to the presence of the highest number of ferrocenium residues in this molecule in comparison with compounds 5 and 6, providing the most satisfactory cancer-specific features among the ferrocenium-tethered sumanene derivatives tested.
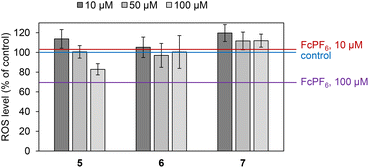
The level of ROS with the tested Fc+-containing sumanene derivatives 5–7 was evaluated for MDA-MB-231 (breast cancer) cells to investigate the potential of these derivatives towards the generation of ROS. The results of this assay are presented in Fig. 6. In the used model, compounds 5–7 featured the property of generating ROS, elucidated by the higher level of ROS in comparison with the control for most tested concentrations. This suggested that the cytotoxic action of 5–7 was associated with the generation of ROS leading to the formation of radical metabolites responsible for biological damage to the cancer cells. The ROS levels of 5–7 were also higher in comparison with that of FcPF6, as graphically presented in Fig. 6. The most satisfactory ROS level for all compounds was observed for the concentration of 10 μM. Notably, while compounds 5 and 6 were able to generate ROS only at this concentration, compound 7 was not only able to effectively generate ROS at all tested concentrations (10, 50 and 100 μM), but also the ROS levels of this derivative were the highest among the compounds tested. This further supported the most satisfactory biological action of compound 7 among the investigated Fc+-containing sumanene derivatives concluded from in vitro assays with MDA-MB-231 cells (Fig. 6).
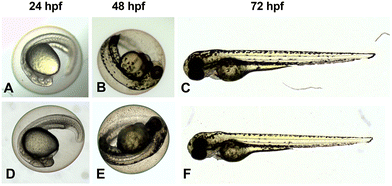
To finally elucidate the biological potential of 7, as the most promising anticancer agent among the sumanene–ferrocenium conjugates tested, we intended to evaluate its embryotoxicity. The use of zebrafish embryos is one of the most attractive, leading toxicology models in this field, and it constitutes a suitable alternative for the use of small laboratory animals, such as rats or mice, due to short analysis times, short life cycle and transparency of embryos, and, importantly, zebrafish share approximately seventy percent protein-coding genes with humans.63–65 Thus, zebrafish (Danio rerio) teratogenicity assays are commonly used for the prediction of the embryotoxic effects of chemicals, including new drug candidates. The images of zebrafish (Danio rerio) embryos treated with and without the compound 7 are presented in Fig. 7. To our delight, there was no observed mortality, change in hatching, or morphological alteration between zebrafish embryos exposed to compound 7 for all concentrations (1–50 μg mL−1) tested and control embryos (E3 with 0.1% DMSO). We did not also observe any sublethal morphological malformations such as yolk or heart oedema, abnormal body shape, reduced yolk, impaired fin development, or eye malformation in all examined groups (Fig. 7). It means that the compound 7 showed no toxicity at the tested concentrations in zebrafish embryos.
Taking into account the growing importance of in silico modeling methods66,67 on novel drug candidates related to the reduction of the usage of animal models before human trials,68,69 ADME–Tox properties for compounds 4–7 were also assessed and the details are presented in ESI, Section S7.† In brief, in silico computational analysis showed no chromosomal aberrations and no mutation with AMES tests for the compound 7 tested with and without microsomal rat liver fractions, which supports its further use as a potent drug candidate in detailed anticancer studies.
Conclusions
In conclusion, the efficient and easy-to-perform method for the oxidation of ferrocene units in ferrocene–sumanene conjugates 2–4 into the derivatives 5–7 comprising ferrocenium cations (Fc+) was achieved under mild conditions. Biological assays revealed that the target FeIII-containing buckybowls represent an attractive class of anticancer agents towards killing breast cancer, as supported by in vitro cell viability assays with human breast adenocarcinoma cells. Among the compounds tested, the tetracationic sumanene derivative 7 featured the most satisfactory biological features. Compound 7 featured the best cytotoxic action towards cancer cells, with the cell viabilities of MDA-MB-231 cancer cells lower than 40% for all tested concentrations, together with the best selectivity towards killing cancer cells, with cell viabilities of HMF normal cells at the level of 75% (for the concentration of 10 μM). Reactive oxygen species (ROS) generation assay with MDA-MB-231 cancer cells revealed the potential of compounds 5–7 towards the generation of ROS responsible for biological damage to the cancer cells, with the most efficient effects for the tetra-ferrocenium sumanene derivative 7. Zebrafish (Danio rerio) teratogenicity assays with 7 also revealed no embryotoxic effects of this molecule, which is also in silico-predicted to have no clastogenic and mutagenic activities, supporting its potential as an anticancer drug candidate. Notably, while sumanene itself was found to be cytotoxic to cancer cells, it also featured significant cytotoxicity against normal cells. In fact, the attachment of ferrocenium residues to the sumanene backbone (compound 7 obtained) improved the biological action of native sumanene, in terms of cytotoxic action selective towards cancer cells. The cytotoxicity of ferrocenium hexafluorophosphate (FcPF6) itself against cancer cells was also not as satisfactory as that of compound 7. Such conclusions thus revealed an effect of the presence of the sumanene backbone in 7 towards providing a good cytotoxicity profile of 7, also in terms of the improved selectivity of the cytotoxic action of 7 in comparison with that of FcPF6. We believe that this work demonstrates the application potential of specifically designed sumanene derivatives in medicinal chemistry and opens new possibilities in this field of applied science of buckybowls.Experimental section
Synthesis of 4
2-(Ferrocenylethynyl)sumanene (2; 21.6 mg, 0.046 mmol, 1 eq.) was placed in a reaction test tube. Tetrabutylammonium bromide (TBAB; 8.0 mg, 0.024 mmol, 0.5 eq.) was added, followed by the addition of dry THF (0.5 mL) and degassed NaOHaq (30%; 4 mL). The reaction mixture was stirred for 5 min at room temperature. Solid formylferrocene (8; 80.0 mg, 0.368 mmol, 8 eq.) was added in one portion, and the reaction mixture was stirred for 72 hours at room temperature. Distilled water (10 mL) was added, and the crude product was extracted with CH2Cl2 (3 × 25 mL). Organic layers were combined and washed with saturated NH4Cl (10 mL), water (10 mL), and brine (10 mL). After drying with MgSO4 followed by filtration, volatiles were distilled off on a rotary evaporator. Finally, the product was purified using a PTLC (SiO2, 50% CH2Cl2/hexane) to provide the target compound 4 as a deep-red solid (34.1 mg, 70%).1H NMR (THF-d8, 500 MHz, ppm), δH 8.22–8.19 (m, 1H), 8.05–8.02 (m, 0.5H), 7.89 (s, 0.5H), 7.76–7.64 (m, 2.5H), 7.47–7.46 (m, 0.5H), 7.37–7.26 (m, 3H), 5.27–5.08 (m, 3H), 4.83–4.48 (m, 12H), 4.40–4.22 (m, 21H); 13C{1H} NMR (THF-d8, 125 MHz, ppm), δC 147.5, 147.3, 146.3, 146.2, 145.2, 145.0, 144.0, 142.2, 138.6, 138.4, 137.7, 131.6, 131.5, 131.3, 129.4, 129.2, 129.0, 128.8, 128.3, 126.1, 125.2, 125.1, 125.0, 124.8, 124.6, 124.5, 122.4, 121.7 × 2, 121.5, 121.3, 121.1, 93.4, 93.2, 82.0, 81.9, 81.8, 72.8, 72.7, 72.5, 72.3, 72.0, 71.7, 71.6, 71.4, 71.3, 71.1, 70.8, 70.6, 70.2; HRMS (ESI) m/z [M]+ calcd for C66H44Fe4 1060.0835, found 1060.0840; Rf (50% CH2Cl2/hexane) = 0.37.
General procedure for the synthesis of 5–7
Compounds 2–4 (0.02 mmol, 1 eq.) were placed in a reaction flask. The content of the flask was evacuated and purged with argon. Dry DCM (5 mL) was added, and the content of the flask was cooled to −20 °C. A solution of silver(I) hexafluorophosphate (AgPF6; 0.06 mmol, 4 eq.) in dry DCM (3 mL) was slowly added at −20 °C. The addition of AgPF6 resulted in a color change, as well as the formation of a brownish precipitate. The reaction mixture was stirred for 10 min at room temperature, filtered off and washed with DCM and EtOH. Finally, after drying under high vacuum for several hours, compounds 5–7 were obtained.Biological tests
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 72 including coagulation, lack of somite formation, no heartbeat, or non-detachment of the tail. The embryos were analyzed under an Olympus CKX53, and images were captured using an Olympus EP50 camera. A tricaine (0.3%) solution was applied at the end of the experiment for euthanasia.
72 including coagulation, lack of somite formation, no heartbeat, or non-detachment of the tail. The embryos were analyzed under an Olympus CKX53, and images were captured using an Olympus EP50 camera. A tricaine (0.3%) solution was applied at the end of the experiment for euthanasia.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The financial support from the National Science Centre, Poland, OPUS grant no. 2021/43/B/ST4/00114 (A. K.), Warsaw University of Technology (WUT; A. K.), as well as JSPS KAKENHI (grant no. JP19H00912 and JP21H05233; H. S.) is acknowledged. The IIMCB Zebrafish Core Facility for service and fish material is also acknowledged.References
- H. Sakurai, T. Daiko and T. Hirao, Science, 2003, 301, 1878 CrossRef CAS.
- M. Saito, H. Shinokubo and H. Sakurai, Mater. Chem. Front., 2018, 2, 635–661 RSC.
- T. Amaya and T. Hirao, Chem. Rec., 2015, 15, 310–321 CrossRef CAS.
- T. Amaya and T. Hirao, Chem. Commun., 2011, 47, 10524 RSC.
- S. Alvi and R. Ali, Beilstein J. Org. Chem., 2020, 16, 2212–2259 CrossRef.
- H. Sakurai, T. Daiko, H. Sakane, T. Amaya and T. Hirao, J. Am. Chem. Soc., 2005, 127, 11580–11581 CrossRef.
- S. N. Spisak, Z. Wei, A. Yu. Rogachev, T. Amaya, T. Hirao and M. A. Petrukhina, Angew. Chem., Int. Ed., 2017, 56, 2582–2587 CrossRef.
- T. Amaya, W. Wang, H. Sakane, T. Moriuchi and T. Hirao, Angew. Chem., Int. Ed., 2010, 49, 403–406 CrossRef.
- T. Amaya and T. Hirao, in Advances in Organometallic Chemistry and Catalysis, ed. A. J. L. Pombeiro, John Wiley & Sons, Inc., Hoboken, NJ, USA, 2013, pp. 473–483 Search PubMed.
- T. Amaya, H. Sakane and T. Hirao, Angew. Chem., 2007, 119, 8528–8531 CrossRef.
- H. Sakane, T. Amaya, T. Moriuchi and T. Hirao, Angew. Chem., Int. Ed., 2009, 48, 1640–1643 CrossRef CAS PubMed.
- T. Amaya, Y. Takahashi, T. Moriuchi and T. Hirao, J. Am. Chem. Soc., 2014, 136, 12794–12798 CrossRef CAS.
- J. Han, Y. Yakiyama, Y. Takeda and H. Sakurai, Inorg. Chem. Front., 2023, 10, 211–217 RSC.
- Y. Yakiyama, T. Hasegawa and H. Sakurai, J. Am. Chem. Soc., 2019, 141, 18099–18103 CrossRef CAS.
- A. Kasprzak and H. Sakurai, Dalton Trans., 2019, 48, 17147–17152 RSC.
- A. Kasprzak, A. Kowalczyk, A. Jagielska, B. Wagner, A. M. Nowicka and H. Sakurai, Dalton Trans., 2020, 49, 9965–9971 RSC.
- A. Kasprzak, A. Gajda-Walczak, A. Kowalczyk, B. Wagner, A. M. Nowicka, M. Nishimoto, M. Koszytkowska-Stawińska and H. Sakurai, J. Org. Chem., 2023, 88, 4199–4208 CrossRef CAS.
- J. S. Cyniak, Ł. Kocobolska, N. Bojdecka, A. Gajda-Walczak, A. Kowalczyk, B. Wagner, A. M. Nowicka, H. Sakurai and A. Kasprzak, Dalton Trans., 2023, 52, 3137–3147 RSC.
- D. Astruc, Eur. J. Inorg. Chem., 2017, 6–29 CrossRef CAS.
- F. A. Larik, A. Saeed, T. A. Fattah, U. Muqadar and P. A. Channar, Appl. Organomet. Chem., 2017, 31, e3664 CrossRef.
- P. Štěpnička, Dalton Trans., 2022, 51, 8085–8102 RSC.
- R. Wang, H. Chen, W. Yan, M. Zheng, T. Zhang and Y. Zhang, Eur. J. Med. Chem., 2020, 190, 112109 CrossRef CAS.
- C. Ornelas, New J. Chem., 2011, 35, 1973 RSC.
- L. V. Snegur, Inorganics, 2022, 10, 226 CrossRef CAS.
- M. Koszytkowska-Stawińska and W. Buchowicz, Dalton Trans., 2023, 52, 1501–1517 RSC.
- P. Köpf-Maier, H. Kopf and E. W. Neuse, J. Cancer Res. Clin. Oncol., 1984, 108, 336–340 CrossRef PubMed.
- D. Osella, M. Ferrali, P. Zanello, F. Laschi, M. Fontani, C. Nervi and G. Cavigiolio, Inorg. Chim. Acta, 2000, 306, 42–48 CrossRef.
- G. Tabbì, C. Cassino, G. Cavigiolio, D. Colangelo, A. Ghiglia, I. Viano and D. Osella, J. Med. Chem., 2002, 45, 5786–5796 CrossRef.
- M. F. R. Fouda, M. M. Abd-Elzaher, R. A. Abdelsamaia and A. A. Labib, Appl. Organomet. Chem., 2007, 21, 613–625 CrossRef.
- C. Y. A. Morantes, E. Meléndez, S. P. Singh and J. E. Ramírez-Vick, J. Cancer Sci. Ther., 2012, 4, 271–275 Search PubMed.
- Z. Chen, R. Mao and Y. Liu, Curr. Drug Metab., 2012, 13, 1035–1045 CrossRef PubMed.
- L. Tang, Q. Xiao, Y. Mei, S. He, Z. Zhang, R. Wang and W. Wang, J. Nanobiotechnol., 2021, 19, 423 CrossRef PubMed.
- J. Li, H. Zeng, Z. Zeng, Y. Zeng and T. Xie, ACS Biomater. Sci. Eng., 2021, 7, 5363–5396 CrossRef.
- M. Mattarella, J. Garcia-Hartjes, T. Wennekes, H. Zuilhof and J. S. Siegel, Org. Biomol. Chem., 2013, 11, 4333–4339 RSC.
- S. Liu, Z. Sun, M. Liang, W. Song, R. Zhang, Y. Shi, Y. Cui and Q. Gao, Adv. Sci., 2022, 9, 2105315 CrossRef.
- J. S. Al-Otaibi, Y. S. Mary, Y. S. Mary, A. Mondal, N. Acharjee and D. G. Churchill, Comput. Theor. Chem., 2022, 1215, 113811 CrossRef.
- (a) T. Reichert, M. Vučićević, P. Hillman, M. Bleicher, S. J. Armaković and S. Armaković, J. Mol. Liq., 2021, 342, 117526 CrossRef; (b) M. Nezamabadi, E. Balali and M. Qomi, Inorg. Chem. Commun., 2023, 155, 111098 CrossRef CAS.
- R. Hong and B. Xu, Cancer Commun., 2022, 42, 913–936 CrossRef.
- N. Harbeck, F. Penault-Llorca, J. Cortes, M. Gnant, N. Houssami, P. Poortmans, K. Ruddy, J. Tsang and F. Cardoso, Nat. Rev. Dis. Primers, 2019, 5, 66 CrossRef.
- T. Amaya, K. Mori, H.-L. Wu, S. Ishida, J. Nakamura, K. Murata and T. Hirao, Chem. Commun., 2007, 1902–1904 RSC.
- A. Kasprzak, A. Tobolska, H. Sakurai and W. Wróblewski, Dalton Trans., 2022, 51, 468–472 RSC.
- A. Kasprzak and H. Sakurai, Chem. Commun., 2021, 57, 343–346 RSC.
- S. Fery-Forgues and B. Delavaux-Nicot, J. Photochem. Photobiol., A, 2000, 132, 137–159 CrossRef CAS.
- J. Lehr, M. Tropiano, P. D. Beer, S. Faulkner and J. J. Davis, Chem. Commun., 2015, 51, 6515–6517 RSC.
- E. Laviron, L. Roullier and C. Degrand, J. Electroanal. Chem. Interfacial Electrochem., 1980, 112, 11–23 CrossRef CAS.
- D. Vijay, H. Sakurai, V. Subramanian and G. N. Sastry, Phys. Chem. Chem. Phys., 2012, 14, 3057 RSC.
- N. G. Connelly and W. E. Geiger, Chem. Rev., 1996, 96, 877–910 CrossRef CAS PubMed.
- A. J. Plajer, F. J. Rizzuto, L. K. S. Von Krbek, Y. Gisbert, V. Martínez-Agramunt and J. R. Nitschke, Chem. Sci., 2020, 11, 10399–10404 RSC.
- Š. Toma and R. Šebesta, Synthesis, 2015, 1683–1695 CrossRef.
- G. Noirbent, D. Brunel, T.-T. Bui, S. Péralta, P.-H. Aubert, D. Gigmes and F. Dumur, New J. Chem., 2021, 45, 13475–13498 RSC.
- M. Nakahata, Y. Takashima, H. Yamaguchi and A. Harada, Nat. Commun., 2011, 2, 511 CrossRef PubMed.
- G. Wilkinson, M. Rosenblum, M. C. Whiting and R. B. Woodward, J. Am. Chem. Soc., 1952, 74, 2125–2126 CrossRef.
- D. A. Khobragade, S. G. Mahamulkar, L. Pospíšil, I. Císařová, L. Rulíšek and U. Jahn, Chem. – Eur. J., 2012, 18, 12267–12277 CrossRef.
- H. B. Gray, D. N. Hendrickson and Y. S. Sohn, Inorg. Chem., 1971, 10, 1559–1563 CrossRef.
- A. Kasprzak, A. M. Nowicka, J. P. Sek, M. Fronczak, M. Bystrzejewski, M. Koszytkowska-Stawinska and M. Poplawska, Dalton Trans., 2018, 47, 30–34 RSC.
- J. C. Smart and B. L. Pinsky, J. Am. Chem. Soc., 1980, 102, 1009–1015 CrossRef.
- K. Venkatasubbaiah, I. Nowik, R. H. Herber and F. Jäkle, Chem. Commun., 2007, 2154–2156 RSC.
- Our initial attempts on oxidation of 2–4 in acetone using aqueous solution of iron(III) chloride, followed by the treatment with solid ammonium hexafluorophosphate were unsuccessful (NMR analyses only showed the regeneration of the starting materials, ca. 90%). We anticipated that this might be the result of the very poor solubility of sumanene–ferrocene conjugates 2–4 in acetone.
- H. Song, G. Kwon, C. Citek, S. Jeon, K. Kang and E. Lee, ACS Appl. Mater. Interfaces, 2021, 13, 46558–46565 CrossRef PubMed.
- F. S. T. Khan, A. L. Waldbusser, M. C. Carrasco, H. Pourhadi and S. Hematian, Dalton Trans., 2021, 50, 7433–7455 RSC.
- A. T. Liu, Y. Kunai, A. L. Cottrill, A. Kaplan, G. Zhang, H. Kim, R. S. Mollah, Y. L. Eatmon and M. S. Strano, Nat. Commun., 2021, 12, 3415 CrossRef PubMed.
- H.-G. Xu, S. Annamadov and A. Mokhir, J. Organomet. Chem., 2022, 964, 122305 CrossRef CAS.
- A. Modarresi Chahardehi, H. Arsad and V. Lim, Plants, 2020, 9, 1345 CrossRef.
- J. C. Achenbach, C. Leggiadro, S. A. Sperker, C. Woodland and L. D. Ellis, Toxics, 2020, 8, 126 CrossRef CAS PubMed.
- K. C. Brannen, J. M. Panzica-Kelly, T. L. Danberry and K. A. Augustine-Rauch, Birth Defects Res., Part B, 2010, 89, 66–77 CrossRef CAS PubMed.
- A. V. Sadybekov and V. Katritch, Nature, 2023, 616, 673–685 CrossRef CAS.
- S. Alqahtani, Expert Opin. Drug Metab. Toxicol., 2017, 13, 1147–1158 CrossRef CAS.
- M. Wadman, Science, 2023, 379, 127–128 CrossRef CAS PubMed.
- S. K. Niazi, Science, 2022, 377, 162–163 CrossRef.
- C. B. Kimmel, W. W. Ballard, S. R. Kimmel, B. Ullmann and T. F. Schilling, Dev. Dyn., 1995, 203, 253–310 CrossRef CAS PubMed.
- S. Xu, F. Chen, H. Zhang, Z. Huang, J. Li, D. Wu and X. Chen, Toxicol. Mech. Methods, 2023, 33, 104–112 CrossRef CAS PubMed.
- OECD, Fish Embryo Acute Toxicity (FET) Test, 2013, Test No. 236.
Footnote |
| † Electronic supplementary information (ESI) available: Materials and methods, experimental section, compound characterization data, spectrofluorimetric and voltammetric data on the molecular receptor application, and details on biological assays. See DOI: https://doi.org/10.1039/d3dt03810f |
| This journal is © The Royal Society of Chemistry 2024 |