Open Access Article
Open Access ArticlePharmacological and phytochemical biodiversity of Corchorus olitorius
Marwa A. M. Abdel-Razek *a,
Miada F. Abdelwahab
*a,
Miada F. Abdelwahab a,
Usama Ramadan Abdelmohsen
a,
Usama Ramadan Abdelmohsen b and
Ashraf N. E. Hamed
b and
Ashraf N. E. Hamed a
a
aDepartment of Pharmacognosy, Faculty of Pharmacy, Minia University, 61519 Minia, Egypt. E-mail: drmarwaali33@gmail.com
bDepartment of Pharmacognosy, Faculty of Pharmacy, Deraya University, Universities Zone, 61111 New Minia City, Egypt
First published on 7th December 2022
Abstract
Corchorus olitorius L. is a nutritious green leafy vegetable that is eaten as a viscous soup in African and Middle Eastern cultures. The purpose of this review is to highlight the nutritional and nutraceutical potential of this plant. The leaves of C. olitorius are rich providers of minerals as calcium and iron in addition to vitamins B1, B2, folic acid C and E. The leaves contain numerous compounds having several biological effects including antidiabetic and antioxidant properties. Besides, the leaves comprise other phytochemicals such as cardiac glycosides, terpenes, flavonoids, fatty acids, hydrocarbons and phenolics. Various extracts of C. olitorius were shown to exhibit antioxidant, anti-inflammatory, hepatoprotective, antihyperlipidemic, immunostimulant, antitumor, antimicrobial, antidiabetic, analgesic, wound-healing properties and cardioprotective activities.
1. Introduction
C. olitorius, belonging to the family Malvaceae, is an annual herbaceous plant with a thin stem. It can be found in all tropical and sub-tropical regions because it is used as a popular leafy soup.1 It is considered as a nutritious vegetable attributed to its high content of vitamins, minerals, and phenolic compounds.2 C. olitorius is a stiff and fibrous annual herb that grows up to 4 m tall, it is also known as bush okra, wild okra, Jew's mallow, tossa jute, long fruited jute, Meloukia, Moroheia, Moroheiya, Mulukhiyah (and other spelling variations), nalta jute, and Tasso.3 It is utilized as a herbal remedy for fevers, chronic cystitis, dysentery and aches, in addition to its cooking purposes. In some parts of Nigeria, the leaf decoction is used for the treatment of iron deficiency as well as folic acid deficiency, in addition to the use of leaves in ascites, dysuria, pectoral pain and female infertility.4–6 Furthermore, typhoid and malarial fevers are treated with the leaves as a herbal medication.7 Leaf twigs are used to treat heart issues, while the leaf infusion is utilized as an appetite enhancer. In Tanzania, the leaves are used to alleviate constipation.5,8,9 Additionally, leaves are used as emollient, diuretic and in treatment of infantile malnutrition in Benin (Fig. 1).92. Methodology
From 2000 to 2022, a systematic research was carried out for the previous literature (66 peer-reviewed articles) in databases such as Pubmed, Google Scholar, El Sevier and Science Direct for reported compounds and biological activities of C. olitorius.3. Pharmacological biodiversity
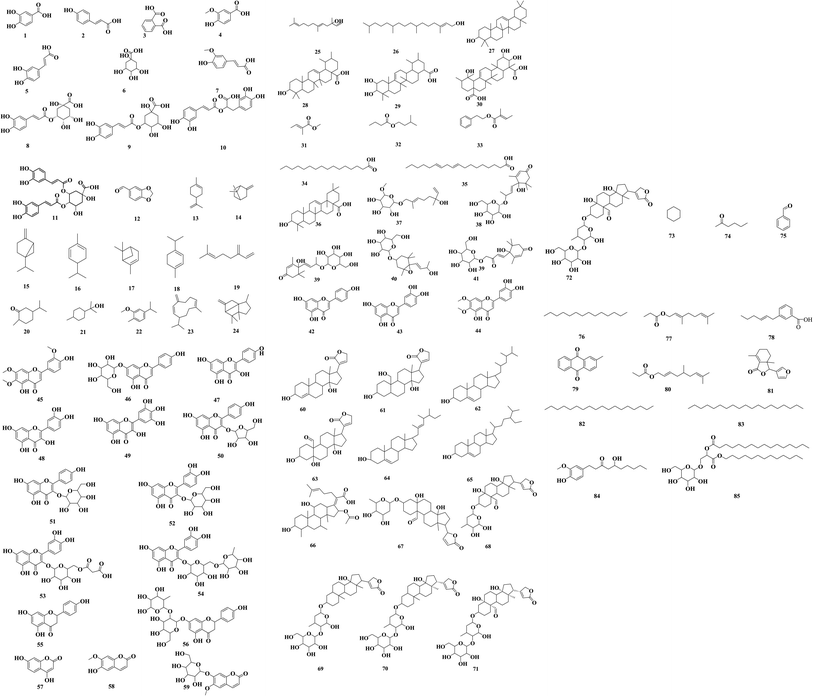
C. olitorius has been shown to have wide spectrum of biological activities, which is certainly related to its phytochemical composition. Table 1 lists some of the main constituents detected in C. olitorius leaves, flowers and seeds covering the literature from 2000 to 2022. Fig. 2 shows the chemical structures of these reported metabolites.| No. | Compound | Molecular weight | Molecular formula | Part used | References |
|---|---|---|---|---|---|
| (1) Phenolic acids | |||||
| 1 | Protocatchuic acid | 154.12 | C7H6O4 | Leaves | 55 |
| 2 | Coumaric acid | 164.15 | C9H8O3 | Leaves | 55 |
| 3 | 1,2-Benzenedicarboxylic acid | 166.14 | C10H10O4 | Flowers | 56 |
| 4 | Vanillic acid | 168.14 | C8H8O4 | Flowers | 57 |
| 5 | Caffeic acid | 180.16 | C9H8O4 | Flowers | 58 |
| 6 | Quinic acid | 192.17 | C7H12O6 | Flowers | 55 |
| 7 | Ferulic acid | 194.18 | C10H10O4 | Flowers | 58 |
| 8 | Chlorogenic acid | 354.31 | C16H18O9 | Flowers | 59 |
| 9 | Isochlorogenic acid | 354.31 | C16H18O9 | Flowers | 59 |
| 10 | Rosmarinic acid | 360.31 | C18H16O8 | Flowers | 55 |
| 11 | 3,4-Di-O-caffeoylquinic acid | 516.45 | C25H24O12 | Flowers | 55 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (2) Terpenes | |||||
| (2i) Monoterpenes | |||||
| 12 | Piperonal | 150.13 | C8H6O3 | Leaves | 55 |
| 13 | Limonene | 136.23 | C9H13 | Leaves | 56 |
| 14 | Camphene | 136.23 | C10H16 | Leaves | 56 |
| 15 | Sabinene | 136.23 | C10H16 | Leaves | 56 |
| 16 | α-Phellandrene | 136.23 | C10H16 | Leaves | 56 |
| 17 | α-Pinene | 136.23 | C10H16 | Leaves | 56 |
| 18 | α-Terpinene | 136.23 | C10H16 | Leaves | 56 |
| 19 | β-Myrcene | 136.23 | C10H16 | Leaves | 56 |
| 20 | Menthone | 154.25 | C10H18O | Leaves | 55 |
| 21 | Dihydroterpineol | 156.26 | C10H20O | Leaves | 60 |
| 22 | Carvacrol methyl ether | 164.24 | C11H16O | Leaves | 55 |
| (2ii) Sesquiterpenes | |||||
| 23 | Germacrene D | 204.35 | C15H24 | Flowers | 56 |
| 24 | β-Cedrene | 204.35 | C15H24 | Flowers | 56 |
| 25 | Nerolidol | 222.27 | C15H26O | Flowers | 56 |
| (2iii) Diterpenes | |||||
| 26 | Phytol | 296.53 | C20H40O | Seeds | 61 |
| (2iv) Triterpenes | |||||
| 27 | β-Amyrin | 426.72 | C30H50O | Seeds | 60 |
| 28 | Ursolic acid | 456.7 | C30H48O3 | Seeds | 8 |
| 29 | Corosolic acid | 472.69 | C30H48O4 | Seeds | 6 |
| 30 | Corrosion | 518.68 | C30H46O7 | Seeds | 6 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (3) Fatty acids | |||||
| 31 | Methyl tiglate | 114.14 | C6H10O2 | Leaves | 56 |
| 32 | Isoamyl butyrate | 158.24 | C9H18O2 | Leaves | 60 |
| 33 | Phenyl ethyl tiglate | 204.26 | C13H16O2 | Leaves | 60 |
| 34 | Palmitic acid | 256.42 | C16H32O2 | Leaves | 60 |
| 35 | Linoleic acid | 280.4 | C18H32O2 | Leaves | 60 |
| 36 | Oleanolic acid | 456.7 | C30H48O3 | Leaves | 61 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (4) Ionones | |||||
| 37 | Betulabuside A | 332.36 | C16H28O7 | Roots | 62 |
| 38 | Corchoiononside C | 386.44 | C19H30O8 | Roots | 6 |
| 39 | 6S,9R-Roseoside | 386.44 | C19H30O8 | Roots | 6 |
| 40 | Corchoiononside A | 388.42 | C19H32O8 | Seeds | 6 |
| 41 | Corchoiononside B | 400.42 | C19H28O9 | Seeds | 6 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (5) Flavonoids | |||||
| (5i) Flavones derivatives | |||||
| 42 | Apigenin | 270.24 | C15H10O5 | Flowers | 55 |
| 43 | Luteolin | 286.24 | C15H10O6 | Flowers | 55 |
| 44 | Cirsiliol | 330.29 | C17H14O7 | Leaves | 55 |
| 45 | Cirsilineol | 344.4 | C18H16O7 | Leaves | 55 |
| 46 | Apigenin 7-O-glucoside | 432.4 | C21H20O10 | Leaves | 55 |
| (5ii) Flavonol derivatives | |||||
| 47 | Kaempferol | 286.24 | C15H10O6 | Flowers | 55 |
| 48 | Quercetin | 302.24 | C15H10O7 | Flowers | 55 |
| 49 | Myricetin | 318.24 | C15H10O8 | Flowers | 55 |
| 50 | Juglanin (kaempferol 3O-α-L-arabinopyranoside) | 418.38 | C20H18O10 | Flowers | 55 |
| 51 | Astragalin | 448.38 | C21H20O11 | Flowers | 59 |
| 52 | Quercetin-3-galactoside | 464.38 | C21H20O12 | Flowers | 55 |
| 53 | Quercetin 3-O-(6′′-O-malonyl)-β-D-glucoside | 550.42 | C24H22O15 | Flowers | 55 |
| 54 | Rutin | 610.52 | C27H30O16 | Flowers | 55 |
| (5iii) Flavanone derivatives | |||||
| 55 | Naringenin | 272.25 | C15H12O5 | Flowers | 55 |
| 56 | Naringin | 580.53 | C27H32O14 | Flowers | 55 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (6) Coumarins | |||||
| 57 | 4,7-Dihydroxycoumarin | 178.14 | C9H6O4 | Leaves | 6 |
| 58 | Scopoletin | 192.17 | C10H8O4 | Leaves | 63 |
| 59 | Scopolin | 354.31 | C16H18O9 | Leaves | 64 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (7) Steroidal derivatives | |||||
| 60 | Canarigenin | 372.49 | C23H32O4 | Seeds | 6 |
| 61 | Cannogenol | 390.49 | C23H32O5 | Seeds | 6 |
| 62 | Campesterol | 400.68 | C28H48O | Seeds | 60 |
| 63 | Strophanthidin | 404.52 | C23H32O6 | Seeds | 65 |
| 64 | Stigmasterol | 412.68 | C29H48O | Seeds | 61 |
| 65 | β-Sitosterol | 414.17 | C29H50O | Seeds | 61 |
| 66 | Fusidic acid | 516.7 | C31H48O6 | Seeds | 63 |
| 67 | Corchoroside A | 534.6 | C29H42O9 | Roots | 66 |
| 68 | Helveticoside (strophanthidin-3-O-β-D-digitoxopyranoside) | 534.61 | C29H42O9 | Seeds | 65 |
| 69 | Coroloside (digitoxigenin-3-O-β-D-glucopyranosyl-(1→4)-O-β-D-boivinopyranoside) | 666.76 | C35H54O12 | Seeds | 65 |
| 70 | Glucoevatromonoside (digitoxigenin-3-O-β-D-glucopyranosyl-(1→4)-O-β-D-digitoxopyranoside) | 666.76 | C35H54O12 | Seeds | 65 |
| 71 | Erysimoside (strophanthidin-3-O-β-D-glucopyranosyl-(1→4)-O-β-D-digitoxopyranoside) | 696.77 | C35H52O14 | Seeds | 65 |
| 72 | Olitoriside (strophanthidin-3-O-β-D-glucopyranosyl-(1→4)-O-β-D-boivinopyranoside) | 696.77 | C35H52O14 | Seeds | 65 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (8) Miscellaneous | |||||
| 73 | Cyclohexane | 84.16 | C6H12 | Leaves | 56 |
| 74 | 2-Hexanone | 100.16 | C6H12O | Leaves | 56 |
| 75 | Benzaldehyde | 106.12 | C7H6O | Leaves | 56 |
| 76 | Tetradecane | 198.39 | C14H30 | Leaves | 61 |
| 77 | Geranyl propionate | 210.31 | C13H22O2 | Leaves | 56 |
| 78 | Hexenyl benzoic acid | 204.29 | C16H18O2 | Leaves | 56 |
| 79 | 2-Methylanthraquinone | 222.24 | C15H10O2 | Leaves | 63 |
| 80 | Geranyl isobutyrate | 224.33 | C14H24O2 | Leaves | 56 |
| 81 | Fraxinellone | 232.28 | C14H16O3 | Leaves | 56 |
| 82 | Octadecane | 254.49 | C18H38 | Leaves | 61 |
| 83 | Nonadecane | 268.52 | C19H40 | Leaves | 61 |
| 84 | Gingerol | 294.39 | C17H26O4 | Leaves | 55 |
| 85 | Monogalactosyldiacylglycerol | 689 | C38H72O10 | Leaves | 8 |
3.1. Antioxidant activity
Potential polyphenols of C. olitorius exhibited antioxidant activity in vitro, and the preliminary results indicated that the studied plant has an interesting free radical scavenging potential, detected by different assays.10C. olitorius infusions and extracts with various polarities were examined for their polyphenolic content and antioxidant activity. The total phenolic content was estimated as gallic acid equivalents (GAE) using the Folin–Ciocalteu technique and spectrometric analysis. The antioxidant activity was investigated through measuring the absorbance of the samples in aqueous emulsion system of linoleic acid and carotene. The ability to scavenge free radicals was also tested against the 2,2-diphenyl-1-picryhydrazyl radical (DPPH). The findings were compared with the synthetic antioxidant, butylated hydroxytoluene (BHT). The overall quantity of phenolic components in the extracts was linked to their antioxidant properties. Samples with elevated phenolic content demonstrated stronger antioxidant activity.11
The presence of secondary metabolites including phenols, glycosides, steroids, terpenoids, and flavonoids was identified in the phytochemical screening of C. olitorius leaf extracts. DPPH radical scavenging assay was used to test the extracts for total phenolic, total flavonoids and antioxidant activity. The results confirmed that the ethanolic extract of C. olitorius leaves had higher antioxidant activity than the petroleum ether and aqueous extracts, due to its higher concentration of phytochemical constituents. The presence of hydroxyl groups in phenolic constituents could explicate the strong scavenging ability.12
When rats are exposed to radiation, their free radical levels rise from subtoxic (24 g mol−1) to toxic (120 g mol−1) levels in their systems. When free radical levels rise above 120 g mol−1, Nf-κB and Nrf-2 are dysregulated, aggravating oxidative stress and cellular changes in rats. The effects of solvent fractions (n-hexane, ethyl acetate and n-butanol) of C. olitorius leaves in radiation-induced Nf-κB and Nrf-2 dysregulation in the cellular system of rats were examined. Irradiation boosted Nf-κB, alkaline phosphatase (ALP), and alanine transaminase (ALT) while decreasing Nrf-2 and antioxidant capacity considerably. The administration of solvent fractions of C. olitorius leaves at 1000 mg kg−1 lowered Nf-κB, ALP, and ALT, while significantly increasing Nrf-2 and antioxidant capacity in rats in the treated groups, indicating that the n-butanol fraction is the most beneficial. As a result, n-butanol fractions could be investigated as an oral treatment for cellular changes in rats.13
The amount of lycopene, a powerful antioxidant, was investigated in fresh, processed, and preserved jute leaves from the cultivated species C. olitorius and C. capsularis. The maximum concentration of lycopene was found in 45 day-old fresh leaves, which subsequently declined as the plant aged. Leaves steeped in vinegar possessed the maximum lycopene for C. olitorius, on the other hand, leaves boiled with salt and water for fifteen minutes had the highest lycopene for C. capsularis. When compared to fresh leaves, all of the processing procedures enhanced the availability of lycopene. After drying and preservation at 20 °C, lycopene was maintained in preserved jute leaves. Cold dried preserved leaves at 4 °C and subsequently air dried leaves at 33–35 °C had the maximum lycopene concentration, whereas the leaves dried in oven at 100 °C had the lowest. The amount of lycopene in the leaves varied depending on the maturity of the plant. When compared to fresh red tomatoes, lycopene levels in 45 day-old plant Corchorus leaves were more than double.14
C. olitorius mucilage was extracted using hot water. Uronic acid made up the majority of the mucilage (34.24%, w/w). At 65 °C, the solubility was 79.48 1.08%, the swelling index was 29.01 2.54%, the water-holding capacity was 28.66 1.48 g g−1, and the oil-binding capacity was 8.423 0.23 g g−1. The mucilage viscosity increased from 4.38 to 154.97 cP, in a concentration-dependent way. Due to the concurrent rise in surface tension and viscosity, the concentration increase resulted in a decrease in emulsion activity and an increase in emulsion stability. The in vitro radical scavenging activity of the mucilage increased with concentration. The study demonstrated the potential of C. olitorius as a new hydrocolloid source with potential applications in a variety of industries, including food, cosmetics, and pharmaceuticals.15
3.2. Anti-inflammatory activity
The inflammatory activity of C. olitorius was evaluated. Consequently, five fractions were generated, including crude phenolic extracts (using 80% aqueous acetone) of the whole plant, leaf, stem, washed leaf (WL) and dried water washing material (WW). On all fractions, linoleic acid autoxidation inhibition was higher than on α-tocopherol. All fractions, with the exception of WL and WW, showed DPPH radical scavenging efficiency. Moreover, all fractions, except WL and WW, demonstrated regulation of the inflammatory responses through suppression of lipopolysaccharide induced protein expression, nitric oxide and prostaglandin E2. The study showed that washing the leaves markedly influenced DPPH radical scavenging and inflammatory responses due to loss of leaf phenolics.16 In a study investigating the therapeutic effect of C. olitorius on rat testicular tissue damage caused by cadmium (Cd), it was found that C. olitorius (given orally at a dose of 250 mg kg−1) reduced the toxic effect of Cd on testicular tissue. The degeneration was still existing in the treated groups, however inflammatory cell numbers, oedema and apoptotic cells were reduced.173.3. Hepatoprotective activity
Alcohol intake causes gut microbial dysbiosis, which disturbs the gut–liver axis, resulting in the development and progression of alcoholic liver disease. Do et al. looked into the effects of a water-soluble extract from C. olitorius leaves (WM) on alcohol-induced gut–liver axis disruption in rats. The levels of blood indicators for liver injury were lowered when 50 and 100 mg kg−1 of WM were given. Furthermore, WM therapy reduced the effects of alcohol on hepatic inflammation and lipogenic protein levels. WM ameliorated lipid accumulation in liver and the gut barrier function. In addition, the gut microbial composition has been regulated.18In another study, the ethanolic extract of leaves significantly lowered the levels of alanine aminotransferase (ALT), alkaline phosphatase (ALP) and aspartate aminotransferase (AST) at three dose levels of 50, 100, and 200 mg kg−1. At 50 and 100 mg kg−1, the extract also lowered total cholesterol levels and increased HDL levels in a dose-dependent manner. C. olitorius extract was thought to give hepatoprotection with a probable tendency to elevate total cholesterol levels because it increased cholesterol levels at 200 mg kg−1.19
Interestingly, Iweala and Okedoyin demonstrated that the regular consumption of C. olitorius may exacerbate hepatotoxicity which could be attributed to the presence of some accumulated antinutrients in the plant e.g. oxalate, nitrate and cyanide. Nevertheless, the study showed the importance of processing this vegetable via cooking prior consumption in order to eliminate or reduce this hepatotoxic effect in humans. However, they emphasized the role of this plant in increasing the antioxidant potential of consumers through the significant increase in the reduced glutathione levels compared with the normal and negative groups. In addition, they suggested a hepatoprotective role of this plant due the elevated total plasma protein concentration that maintain protein synthesis efficiency in hepatocytes.20
The hepatoprotective property of C. olitorius ethanolic leaf extract on CCl4-induced liver damage in rats was evaluated. The administration of the extract was oral at doses of 500 mg kg−1, 750 mg kg−1 and 1 g kg−1 daily for 15 days period. The serum enzyme assay showed a marked reduction in the elevated activity of the hepatic enzymes ALT, ALP and AST, compared with the control group. Furthermore, the results revealed lowered serum albumin with no change in bilirubin but increased levels of total protein in all treated groups. Additionally, the white blood cell count and platelets were significantly decreased while the packed cell volume and hemoglobin were not changed.21
3.4. Prevention of obesity
Gut dysbiosis is a metabolic illnesses like obesity and leaky gut, it is caused by a high-fat diet (HFD). In HFD-induced C57BL/6J mice, the impact of a water-soluble extract from C. olitorius leaves (WM) on lipid accumulation in 3T3-L1 adipocytes, hormone levels, gut permeability, gut microbiota, and faecal enzyme activity of the intestinal microflora were investigated. In 3T3-L1 adipocytes, WM treatment dramatically reduced lipid buildup. Mice given 100 mg kg−1 WM had 13.1, 17.4, and 52.4% lower body weights, hepatic fat accumulation and intestinal permeability, respectively, than mice given HFD. WM also had an effect on gut health via reducing metabolic endotoxemia and colon inflammation. It also changed the gut microbiota's composition, increasing the number of bacteria in the gut.22Diet-induced obesity was significantly reduced by C. olitorius leaf mucilage polysaccharide fraction (MPF), which also modulated gut flora and inflammation. During an HFD, the administration of MPF for eight weeks reduced body weight, triglyceride levels, as well as adipocyte size, LDL-cholesterol levels and in the expression of lipid synthesis-related proteins. Furthermore, MPF delivery improved gut health by reducing leaky gut, inflammation and metabolic endotoxemia, in addition to enhancing tight junction protein expression. Findings demonstrated that C. olitorius MPF rich in rhamnogalacturonan-I attenuated HFD-induced obesity in mice through regulating the constitution of gut microbiota.23
3.5. Immune enhancing
Polysaccharides derived from plants have the potential to improve gut health and immune system. Mucilage polysaccharide is abundant in C. olitorius leaves. MPF's prebiotic and intestinal immune-enhancing actions were tested in vitro to investigate its bio-functional effects. MPF enhanced immunological function on Peyer's patches, as evidenced by significant increase in bone marrow cell proliferation activity as well as immunoglobulin A and cytokines production. These findings suggested that MPF could be a potentially helpful prebiotic and intestinal immune-enhancer.243.6. Antitumor activity
Dioxins are mostly ingested and produced a variety of toxicological consequences by attaching to the cytosolic aryl hydrocarbon receptor (AhR) and then transforming it. Certain natural substances have been proven to inhibit AhR transformation in vitro. Among 41 types of extracts from vegetables and fruits, Nishiumi et al. found that the ethanolic extract from C. olitorius had the most powerful suppressive effect on AhR transformation induced by TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin) in a cell-free system utilizing rat hepatic cytosol. The extract also inhibited TCDD-induced AhR transformation in mice hepatoma cells and in intestinal permeability system established with human hepatoma (HepG2) and human colon adenocarcinoma (Caco-2) cells.25The effects of the ethanolic extract of C. olitorius on the proliferation of human hepatocellular carcinoma (HepG2) cells and the underlying mechanisms of action were investigated. HepG2 cells treated for 24 hours with the extract at a concentration more than 12.5 μg mL−1 showed a significant decrease in cell viability, on the other hand, normal FL83B hepatocytes were unaffected. The study indicated that C. olitorius ethanol extract could be efficacious against hepatocellular carcinoma via apoptosis induction involving mitochondria-dependent pathway.26
Darcansoy şeri et al. investigated the cytotoxic and genotoxic effects of C. olitorius leaf extracts (LE) and seed extracts (SE) on multiple myeloma-derived ARH-77 cells. The total phenol content (TPC) and free radical scavenging activity (FRSA) of the extracts were also determined using the Folin–Ciocalteu and DPPH free radical techniques, respectively. The MTT test (4–2048 μg mL−1 range) was used to assess cytotoxicity, and the comet assay was used to assess DNA damage (at IC50 and 1/2 IC50). The LE had much higher total phenol content (78 mg GAE per g extract) with pronounced FRSA (IC50: 23 μg mL−1) while SE total phenol was about 2 mg GAE per g extract and FRSA with IC50 SE: 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 401 μg mL−1. Both LE and SE exhibited cytotoxicity after 48 hours, SE showed higher cytotoxicity on cells compared with LE with IC50 values of 17 μg mL−1 and 151 g mL−1, respectively. In addition, the comet test demonstrated that both extracts caused genotoxic damage when applied at 17 and 8.5 μg mL−1 for SE and at 150 and 75 μg mL−1 for LE.27
401 μg mL−1. Both LE and SE exhibited cytotoxicity after 48 hours, SE showed higher cytotoxicity on cells compared with LE with IC50 values of 17 μg mL−1 and 151 g mL−1, respectively. In addition, the comet test demonstrated that both extracts caused genotoxic damage when applied at 17 and 8.5 μg mL−1 for SE and at 150 and 75 μg mL−1 for LE.27
Acrylamide is a compound formed when starchy foods are cooked at high temperatures. It is carcinogenic, genotoxic and neurotoxic. Short-term acrylamide exposure has also been proven to produce severe hepatic impairment in experimental animals, as well as disruption of antioxidant defense mechanisms due to increased reactive oxygen species generation. As a result, dietary antioxidants may be beneficial in reducing the harmful effects of acrylamide. C. olitorius is an antioxidant with strong organo-protective effects. The aqueous extract of C. olitorius leaves was administered to rats to study if it could protect them against acrylamide-induced liver damage. After three levels of dose, hepatic damage markers such as blood total protein, total bilirubin, ALT, ALP and AST were examined, as well as oxidative stress markers such as MDA, GSH, CAT, and SOD. The extracts significantly lowered high levels of bilirubin, ALT, AST, ALP, and MDA to normal levels. They also restored normal serum protein, GSH, CAT, and SOD levels. Owing to its high radical scavenging ability, C. olitorius leaves extract protects against acrylamide-induced liver damage.28
From the leaves of jute, two anticancer promoters, phytol and monogalactosyldiacylglycerol, were extracted (C. capsularis L.). Immunoblotting analysis was used to look for antitumor-promoting activities. The metabolites concentration in four cultivars of C. capsularis and C. olitorius varied depending on the cultivar. Treatment of the leaves with hot water increased the measurable amount of each active component.8
Taiwo et al. separated two polyphenolic metabolites namely, methyl-1,4,5-tri-O-caffeoyl quinate and trans-3-(4-hydroxy-3-methoxyphenyl) acrylic anhydride from the methanolic extract of C. olitorius. The isolated compounds were evaluated at a range of concentrations up to 1.6 mM. They were found to exhibit mild cytotoxic activity against HeLa cells at ≥800 μM. The compounds revealed high binding affinities to fibroblast growth factor receptor 2 and epidermal growth factor receptor. Therefore, the plant could be considered as a potential source of ‘lead’ drugs with anti-tumour activity.29 C. olitorius extract in ethanol was used to make copper nano complexes. These metal complexes were found to have in vitro antitumor action against hepatocellular carcinoma (HepG-2) cells, with IC50 value of 30.9 μg mL−1, using cis-platin as the standard anticancer drug.30
The methanolic extract of C. olitorius revealed the presence of flavonoids and phenolic compounds as main ingredients, with 699 g GAE per g and 1361.50 g QE per g, respectively. The methanolic extract of the plant demonstrated cytotoxic activity against L929 cell line with IC50 value of 227.84 g mL−1. In addition, the cell viability in MTT experiment for methanol extract against the MCF-7 and A549 cell lines revealed significant activities with IC50 values of 20 g mL−1 and 12.45 g mL−1, respectively. The extract was found to induce early and late apoptosis in the tested lung and breast cancer cell lines through activating caspase-3 and inhibiting Bcl-2 protein, it also caused cell death via DNA damage. Consequently, the results demonstrated that the methanolic extract of C. olitorius had significantly inhibited breast cancer and lung cancer cell lines.31
3.7. Antimicrobial activity
The antibacterial and antifungal properties of C. olitorius leaf extracts collected from various parts of the Turkish Republic of Northern Cyprus were investigated. Seven samples of dried plant material (C. olitorius leaves) were collected from different regions. Methanol, ethanol, chloroform, and hexane (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 [weight/volume]) were used to extract the leaves at room temperature for three days. Following evaporation, each sample was prepared at a concentration of 100 mg mL−1. The disc diffusion method was used to assess antibacterial activity. Only hexane leaf extracts showed antibacterial action against Bacillus subtilis and Staphylococcus aureus, whereas methanol, ethanol, and chloroform extracts showed no antibacterial activity.32
10 [weight/volume]) were used to extract the leaves at room temperature for three days. Following evaporation, each sample was prepared at a concentration of 100 mg mL−1. The disc diffusion method was used to assess antibacterial activity. Only hexane leaf extracts showed antibacterial action against Bacillus subtilis and Staphylococcus aureus, whereas methanol, ethanol, and chloroform extracts showed no antibacterial activity.32
Ilhan et al. investigated the in vitro antimicrobial activity of petroleum ether, methanol, and ethyl acetate–water extracts of C. olitorius leaves, using the agar-well diffusion method. The petroleum ether extract exerted antibacterial activity against Staphylococcus aureus, Escherichia coli and Yersinia enterocolitica with inhibition zones of 19, 20 and 19 mm, respectively. The ethyl acetate–water extract showed considerable efficacy against Botrytis cinerea and Geotrichum candidum with inhibition zones of 12 and 19 mm, respectively.33
The antimicrobial and antioxidant activities in addition to the total phenolic content of the dried leaf extracts of C. olitorius were studied. The extracts were tested for antibacterial activity, using the broth dilution technique, against Bacillus subtilis, Bacillus cereus, Enterococcus faecalis, Listeria monocytogenes, Staphylococcus aureus, Escherichia coli and Salmonella typhimurium. The MIC values of ethanol and methanol extracts ranged from 50 to 6.25%, while water extract had no inhibitory effect. The methanol extract was significantly active against L. monocytogenes, B. subtilis and E. faecalis with MIC value of 6.25%. Ethanol extract had the highest phenolic content (24.61 mg GAE per g). Water, methanol and ethanol extracts suppressed the DPPH radical by 17.50, 86.00 and 87.10%, respectively. These investigations indicated that C. olitorius extracts are valuable sources of antibacterial and antioxidant activities.34
Strophanthidin, a compound present in C. olitorius, was found to be a promising drug candidate in treatment of brucellosis. The study demonstrated that the compound had potential to inhibit Brucella melitensis methionyl-tRNA synthetase using virtual screening and molecular docking. Strophandin bioavailability was as well confirmed through molecular dynamics modelling with the target protein. In vivo and in vitro studies are required in order to assess the efficacy and toxicity of this lead compound.35
C. olitorius leaves were air dried after collection, then extracted with distilled water, methanol, and diethyl ether. The crude extracts were assessed employing the agar well diffusion method against a number of pathogens. The positive control was chloramphenicol. The crude extracts of C. olitorius leaves exhibited antibacterial activity against Staphylococcus aureus (inhibition zone of 14 mm), Streptococcus pneumoniae (inhibition zone of 16 mm), and E. coli (inhibition zone of 11 mm). The positive control showed inhibition zone of 18 mm. The plant extract did not reveal any bioactivity against Candida albicans nor Mycobacterium tuberculosis.36
The antibacterial activity of C. olitorius leaves was assessed against the Gram-negative bacteria E. coli, using the agar well diffusion method. As a result, the aqueous extract exhibited stronger antibacterial activity (16.3 ± 1.7) against the studied bacteria E. coli, compared to the ethanolic extract (8.23 ± 0.7).37
3.8. Antidiabetic activity
C. olitorius contains a lot of mucilaginous polysaccharides that has been shown to have anti-diabetic properties in animal studies. Although the presence of phenolic and non-phenolic chemicals is also thought to be responsible for the anti-diabetic properties of this plant, the exact mechanism of action is unknown. C. olitorius leaves powder (at 2 and 4%) was tested in vitro for its capacity to modulate starch digestion and glucose diffusion. Results demonstrated that the powder bonded substantially more glucose than wheat bran (2%) and also decreased glucose diffusion over the dialysis membrane, as evidenced by a significantly higher glucose dialysis retardation index. In addition, the plant powder (4%) showed full suppression of starch digestion and glucose diffusion similar to acarbose in the starch-α-amylase system until 120 minutes. C. olitorius leaves powder (4%) slowed down glucose diffusion by 83.7% and 63.5% at 180 and 240 minutes, respectively. Whereas the plant powder (2%) slowed glucose diffusion by 96%, 65%, and 51% at 120, 180, and 240 minutes, respectively. Furthermore, C. olitorius leaf extract obviously increased glucose absorption by rat hemidiaphragm. These data convincingly showed that the antidiabetic activity of C. olitorius leaves is achieved by delaying starch digestion and physical adsorption of released glucose, reducing glucose diffusion across the intestinal lumen and increasing its uptake in peripheral tissues.38The hypoglycemic and the hypolipidemic impact of the leaves of C. olitorius on alloxan-induced diabetic rats was determined. The diabetic rats were given C. olitorius leaves as food supplement for fourteen days. The post-treated rats were shown to have significant decrease in plasma glucose concentrations, total cholesterol, LDL—cholesterol and triglycerides. These findings suggested that C. olitorius leaf has antihyperglycemic and hypolipidemic effects that could be strongly attributed to the different phyto-constituents and the fiber content. Plant fibers may chelate with intestinal glucose therefore reducing blood glucose levels, in addition they contribute in the reduction of circulating plasma cholesterol because they trap bile in intestine leading to more bile production from cholesterol.39
Mahgoub et al. evaluated the effect of C. olitorius soup (prepared from C. olitorius leaves) on fasting blood glucose, lipid profile, and hepatic oxidative status in STZ-induced diabetes in male albino rats. Treatment with C. olitorius soup (given at a dose of 4.80 g per kg body weight) showed encouraging antihyperglycemic, antihypertriglyceridemic as well as antioxidant properties. The serum levels of glucose, triglycerides and liver peroxidative products were significantly reduced in the treated animals. In the meantime, C. olitorius administration had led to increased glutathione, antioxidant enzymes catalase and superoxide dismutase. Hence, the plant could be considered a valuable candidate in the reversal of diabetes complications.40
C. olitorius leaves were serially extracted with methanol, diethyl ether, and water. 48 rats were separated into 8 groups of 6 for the assessment of the antihyperglycemic and antidiabetic effects, and each group received 500 mg kg−1 of each extract. Distilled water and glibenclamide (10 mg kg−1) were used as controls. The methanolic extract showed the most antihyperglycemic efficacy.41
3.9. Analgesic activity
Methanolic C. olitorius extract was tested for analgesic efficacy against acetic acid-induced writhing in mice. The extract was given at doses of 50, 100, 200, and 400 mg per kg body weight. Writhing was induced by injecting acetic acid intraperitoneally. The reference doses of aspirin were 200 and 400 mg kg−1. The plant extract inhibited acetic acid-induced abdominal contraction in a dose-dependent manner. At the dosage range studied, abdominal contractions were reduced by 20 to 58%. The extract had stronger analgesic effect at 100 mg kg−1 than aspirin at 200 mg kg−1, showing that C. olitorius has significant analgesic characteristics.423.10. Wound healing property
In a rat excision wound model, the wound-healing effect of C. olitorius leaf powder and aqueous extract were tested. When compared to the control, both showed considerable wound healing activity. On the 18th day, a 5% powdered plant ointment or 100 mg mL−1 extract produced 100% wound contraction. C. olitorius was found to have powerful healing activity and it can be used as an alternative wound healing treatment since it effectively decreased microbial burden.43Yokoyama et al. were interested in evaluating the skin barrier protection and the hydration efficacy of C. olitorius leaf extract without the polysaccharides. Their experiment demonstrated that the rats topically treated with the cream supplemented with C. olitorius extract had improved skin hydration and suppressed trans-epidermal water loss. This concluded that the plant may maintain the hydration of the skin and inhibit the disturbance of skin barrier function, therefore, it could be used as a complementary therapy in atopic dermatitis.44
An atopic dermatitis mouse model was used to assess the skin hydration capacity of C. olitorius extract. The extract was combined with a stable base cream and applied to the mice's dorsal skin. Following 14 days of therapy, the skin hydration was improved while the trans-epidermal water loss and atopic dermatitis symptoms were significantly reduced compared with control and untreated groups. The results of this study explicated that C. olitorius leaf extract could be a helpful therapeutic candidate for atopic dermatitis which may be attributed to its suppression of the plasma concentrations of immunoglobulin E and degranulation of mast cells.45
3.11. Cardioprotective activity
The cardioprotective activity of C. olitorius aqueous extract at 50 and 100 mg kg−1 against sodium arsenite-induced cardiotoxicity in rats was investigated. Rats exposed to sodium arsenite (10 mg kg−1) for 10 days had a significant rise in plasma total cholesterol and arsenic levels in heart tissue, as well as a reduction in serum HDL. Through histological examinations, pretreatment with C. olitorius extract reverted all of these parameters in blood and cardiac tissues to normal values, indicating that C. olitorius extract may provide considerable protection against sodium arsenite-induced cardiotoxicity.463.12. Antiviral activity
Antioxidants and polyphenolics derived from the leaves of C. olitorius that can block the activity of the angiotensin converting enzyme 2 (ACE2), which is a key protein in the physiology of Covid-19 and necessary for the entry of the SARS-Cov-2 virus into the host's cells. Compared the co-crystallized inhibitor of the enzyme ACE2, chloroquine, hydroxychloroquine, captopril, and simerprevir antiviral drugs, the results of the docking simulation revealed that méthyl-1,4,5-tri-O-caféoyl quinate had a high affinity, stronger bond and gives the best docking scores. Hence, it could be regarded as a potential inhibitor of essential protease viral enzyme.473.13. Treatment of aphthous ulcers
A bucco-adhesive fast-dissolving film that contains C. olitorius seed extract was created in order to treat recurrent minor aphthous ulceration. With special focus on wound healing molecular targets like TGF-, TNF-, and IL-1. An excision wound model was used to evaluate the in vivo wound healing capacity of C. olitorius L. seed extract. Interestingly, the wound healing data showed that C. olitorius seed extract increased wound closure rates, boosted TGF- levels, and dramatically downregulated TNF- and IL-1, when compared to the Mebo-treated group. Additionally, by applying in silico analysis, the study revealed the potential of C. olitorius seed extract in wound repair and identified the most likely mechanisms of action.484. Natural products from C. olitorius
Eleven phenolic acids were reported from C. olitorius that showed diverse biological activities. Caffeic acid and chlorogenic acid phenolic phytochemicals may be responsible for the antidiabetic and antihypertensive properties of C. olitorius.49 Additionally, 3,4-dicaffeoylquinic acid had a powerful antioxidant activity besides neuroprotective action.50 Terpenoids are widely distributed in C. olitorius, with 18 terpenoidal compounds have been reported. Phytol diterpenoidal compounds showed antimicrobial, antioxidant and anticancer activities.51 Ionones are a class of compounds contains corchoiononside A, B and C which were isolated from C. olitorius showing antinociception (strong analgesia)52 in addition to antiallergic activity.53 Flavonoids showed diverse biological activities and 14 flavonoidal derivatives were reported including quercetin, kaempferol, and luteolin.54 They know for their antioxidant and anti-inflammatory health benefits as well as the support of the cardiovascular and nervous systems.54 C. olitorius also had several steroidal compounds including β-sitosterol and cardiac glycosides. Helveticoside for example showed strong cardiotonic activity.53 Finally, there are uncategorized or miscellaneous group including 12 compounds. Monogalactosyldiacylglycerol had anticancer activity against different cell lines.265. Phytochemical biodiversity
Several studies have identified many compounds from different parts of C. olitorius as shown in Table 1. The chemical structures of the reported compounds are illustrated in Fig. 2.6. Conclusion
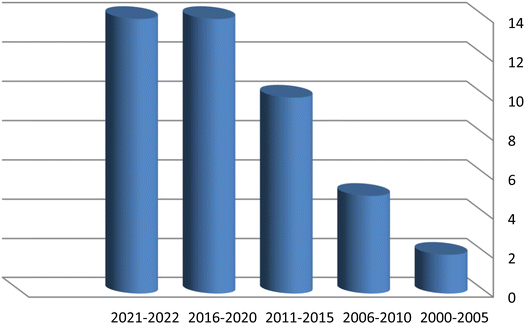
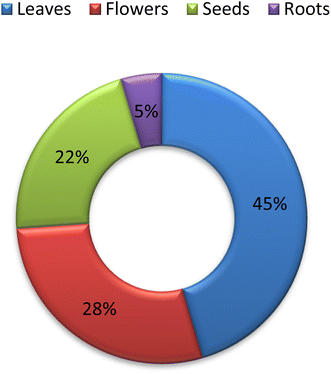
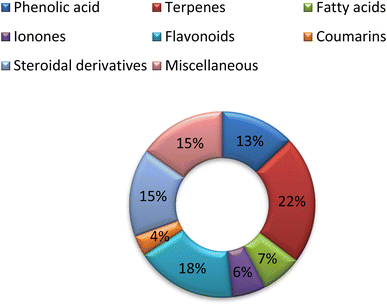
Despite the fact that the leaves of C. olitorius are utilized in traditional medicine and have been known to have a variety of pharmacological effects, their use is restricted to a few cultures and widely employed in eating habits. Significant progress has been detected in publication rates of C. olitorius over the past 22 years (Fig. 3). Researchers have been investigating C. olitorius leaves, flowers as well as seeds and roots (Fig. 4). Investigation of C. olitorius has been shown to have a wide spectrum of interesting bioactivities including antioxidant, hepatoprotective, anti-inflammatory, immunostimulant antitumor, antimicrobial, antidiabetic, analgesic and cardioprotective activities, in addition to the wound healing properties and its role in prevention of obesity and its antiviral activity against Covid 19. This biological diversity is certainly attributed to its diverse phytochemical composition. Flavonoids and terpenes are the most detected phytochemical classes in C. olitorius, followed by phenolic acids, cardiac glycosides and fatty acids (Fig. 5). C. olitorius is regarded as a valuable source for developing novel natural drug candidates for clinical application.Although C. olitorius is used worldwide in complementary medicine, the knowledge about its phytopharmacological biodiversity is still being scanty. Detailed investigations are required to fully understand the relation between the active constituents and the proved biological activities. Analysis of the reported data indicated that the research was particularly limited to the total extracts or fractions, whereas the purified compounds have not received much interest. Additional investigations should be comprehensively performed in order to identify the secondary metabolites and their actual contribution to the medicinal activities of C. olitorius. Moreover, the mechanism of action and the probable synergistic interactions of the different phytoconstituents should be considered as well. This will control the safe and beneficial application of Corchorus olitorius in phytotherapy. From a therapeutic viewpoint, the anticancer and the antimicrobial potentials of C. olitorius represent indispensable future research themes involving the study of the pure compounds, their structure–activity relationship and the mechanisms of action C. olitorius is an interesting candidate for the development of alternative drugs beside its effective utilization as a functional food ingredient due to its prominent nutritional value.
Conflicts of interest
No conflicts to declare.References
- A. Kundu, D. Sarkar, N. A. Mandal, M. K. Sinha and B. S. Mahapatra, A secondary phloic (bast) fibre-shy (bfs) mutant of dark jute (Corchorus olitorius L.) develops lignified fibre cells but is defective in cambial activity, Plant Growth Regul., 2012, 67, 45–55 CrossRef CAS.
- A. Giro and A. Ferrante, Yield and quality of Corchorus olitorius baby leaf grown in a floating system, J. Hortic. Sci. Biotechnol., 2016, 91, 603–610 CrossRef CAS.
- M. M. Islam, Biochemistry, medicinal and food values of jute (Corchorus capsularis L. and C. olitorius L.) leaf: a review, Int. J. Res. Appl. Sci. Eng. Technol., 2013, 2, 135–144 Search PubMed.
- H. M. Al-Yousef, M. Amina and S. R. Ahamad, Comparative study on the chemical composition of Corchorus olitoriusl leaf and stem dry oils, Biomed. Res., 2017, 28, 4581–4587 CAS.
- J. Fasinmirin and A. Olufayo, Yield and water use efficiency of jute mallow Corchorus olitorius under varying soil water management strategies, J. Med. Plants Res., 2009, 3, 186–191 Search PubMed.
- M. Khan, S. Bano, K. Javed and M. A. Mueed, A comprehensive review on the chemistry and pharmacology of Corchorus species—a source of cardiac glycosides, triterpenoids, ionones, flavonoids, coumarins, steroids and some other compounds, J. Sci. Ind. Res., 2006, 65, 283–298 CAS.
- H. O. Adebo, L. E. Ahoton, F. Quenum, H. Adoukonou-Sagbadja, D. O. Bello and C. Chrysostome, Ethnobotanical knowledge of jute (Corchorus olitorius L.) in Benin, Journal of Medicinal Plants, 2018, 26, 1–11 CrossRef.
- T. Furumoto, R. Wang, K. Okazaki, F. Afm, M. I. Ali, A. Kondo and H. Fukui, Antitumor promoters in leaves of jute (Corchorus capsularis and Corchorus olitorius), Food Sci. Technol. Res., 2002, 8, 239–243 CrossRef CAS.
- T. I. E. Fagbohun, 2. physicochemical properties and in vitro anti-bacterial activity of Corchorus olitorius linn. seed oil by 1 TA Ibrahim and 2 ED Fagbohun, Life Sciences Leaflets, 2011, 15, 499–505 Search PubMed.
- T. N. Mouas, Z. Kabouche, N. Benabid and M. Bendal, Investigations on Bioactive Compounds and In Vitro Biological Potent of Corchorus olitorius. L from Algerian Cultivar, Proceedings, 2021, 65, DOI:10.3390/BDEE2021-09439.
- N. Öztürk and F. Savaroğlu, in Survival and Sustainability, Springer, 2010, pp. 535–543 Search PubMed.
- N. M. Hussien, S. Labib, R. El-Massry and H. Hefnawy, Phytochemical studies and antioxidant activity of leaves extracts of Corchorus olitorius L.(molokhia), Zagazig Journal of Agricultural Research, 2017, 44, 2231–2239 CrossRef.
- O. K. Bello, H. O. B. Oloyede, M. O. Salawu and M. T. Yakubu, Amelioration of radiation-induced cellular alterations in rats administered with solvent fractions of methanol leaf extracts of Adansonia digitata and Corchorus olitorius, Biokemistri, 2021, 33, 187–202 Search PubMed.
- A. Nasreen, Z. Ahmed, M. Ali and T. Shimu, Jute leaves: A potential sources of lycopene, Int. J. Veg. Sci., 2022, 28, 180–188 CrossRef.
- S. Oh and D.-Y. Kim, Characterization, Antioxidant Activities, and Functional Properties of Mucilage Extracted from Corchorus olitorius L, Polymers, 2022, 14, 2488 CrossRef CAS PubMed.
- Y.-Y. Yan, Y.-W. Wang, S.-L. Chen, S.-R. Zhuang and C.-K. Wang, Anti-inflammatory effects of phenolic crude extracts from five fractions of Corchorus Olitorius L, Food Chem., 2013, 138, 1008–1014 CrossRef CAS PubMed.
- K. Komili, G. Söyler, P. Toros, İ. Çalış and A. Kükner, Effects of Corchorus Olitorius and Protocatechuic Acid on Cadmium-Induced Rat Testicular Tissue Degeneration, Cyprus Journal Of Medical Sciences, 2022, 7, 87–94 CrossRef.
- M. H. Do, H. H. L. Lee, Y. Kim, H.-B. Lee, E. Lee, J. H. Park and H.-Y. Park, Corchorus olitorius L. ameliorates alcoholic liver disease by regulating gut-liver axis, J. Funct. Foods, 2021, 85, 104648 CrossRef CAS.
- K. Omeje, H. Omeje, A. Odiba, O. Anunobi and C. Ukegbu, Liver enzymes and lipid activities in response to Corchorus olitorius leaf extract, Int. J. Curr. Res. Biosci. Plant Biol., 2016, 3, 45–49 CrossRef CAS.
- E. Iweala and A. Okedoyin, Effect of Consumption of corchus olitorus L., in carbon tetrachloride-induced Liver Damage in male Wistar rats, 2014.
- O. Ujah, S. Ipav, C. Ayaebene and I. Ujah, Phytochemistry and hepatoprotective effect of ethanolic leaf extract of Corchorus olitorius on carbon tetrachloride induced toxicity, Eur. J. Med. Plants, 2014, 4, 882–892 CrossRef.
- H.-B. Lee, M.-J. Oh, M. H. Do, Y.-S. Kim and H.-Y. Park, Molokhia leaf extract prevents gut inflammation and obesity, J. Ethnopharmacol., 2020, 257, 112866 CrossRef CAS PubMed.
- H.-B. Lee, M.-J. Oh, M. H. Do, Y. Kim, I. Choi, Y.-S. Kim and H.-Y. Park, Dietary rhamnogalacturonan-I rich extracts of molokhia ameliorate high fat diet-induced obesity and gut dysbiosis, J. Nutr. Biochem., 2022, 108954 CrossRef CAS PubMed.
- H.-B. Lee, S.-U. Son, J.-E. Lee, S.-H. Lee, C.-H. Kang, Y.-S. Kim, K.-S. Shin and H.-Y. Park, Characterization, prebiotic and immune-enhancing activities of rhamnogalacturonan-I-rich polysaccharide fraction from molokhia leaves, Int. J. Biol. Macromol., 2021, 175, 443–450 CrossRef CAS PubMed.
- S. Nishiumi, Y. Yabushita, I. Fukuda, R. Mukai, K.-i. Yoshida and H. Ashida, Molokhia (Corchorus olitorius L.) extract suppresses transformation of the aryl hydrocarbon receptor induced by dioxins, Food Chem. Toxicol., 2006, 44, 250–260 CrossRef CAS PubMed.
- C.-J. Li, S.-Y. Huang, M.-Y. Wu, Y.-C. Chen, S.-F. Tsang, J.-H. Chyuan and H.-Y. Hsu, Induction of apoptosis by ethanolic extract of Corchorus olitorius leaf in human hepatocellular carcinoma (HepG2) cells via a mitochondria-dependent pathway, Molecules, 2012, 17, 9348–9360 CrossRef CAS PubMed.
- Ö. Darcansoy İşeri, E. Yurtcu, F. I. Sahin and M. Haberal, Corchorus olitorius (jute) extract induced cytotoxicity and genotoxicity on human multiple myeloma cells (ARH-77), Pharm. Biol., 2013, 51, 766–770 CrossRef PubMed.
- A. I. A. Al-Khalifah and F. Ahmed, Corchorus olitorius L. Leaf Extract Protects Rats from Acrylamide-Induced Hepatic Injury, Current Research in Nutrition and Food Science Journal, 2021, 9, 833–840 Search PubMed.
- B. J. Taiwo, G. O. Taiwo, O. O. Olubiyi and A. A. Fatokun, Polyphenolic compounds with anti-tumour potential from Corchorus olitorius (L.) Tiliaceae, a Nigerian leaf vegetable, Bioorg. Med. Chem. Lett., 2016, 26, 3404–3410 CrossRef CAS PubMed.
- S. Hosny, G. A. Gouda and S. M. Abu-El-Wafa, Novel Nano Copper Complexes of a New Schiff Base: Green Synthesis, a New Series of Solid Cr (II), Co (II), Cu (II), Pd (II) and Cd (II) Chelates, Characterization, DFT, DNA, Antitumor and Molecular Docking Studies, Appl. Organomet. Chem., 2022, 36, e6627 CrossRef CAS.
- A. M. Alshabi, S. A. Alkahtani, I. A. Shaikh, M. A. Orabi, B. A. Abdel-Wahab, I. A. Walbi, M. S. Habeeb, M. M. Khateeb, J. H. Hoskeri and A. K. Shettar, Phytochemicals from Corchorus olitorius methanolic extract induce apoptotic cell death via activation of caspase-3, anti-Bcl-2 activity, and DNA degradation in breast and lung cancer cell lines, J. King Saud Univ., Sci., 2022, 34, 102238 CrossRef.
- M. S. Özdenefe, A. Muhammed, K. Süer, E. Güler, H. Aysun and M. Takcı, Determination of antimicrobial activity of Corchorus olitorius leaf extracts, Cyprus Journal Of Medical Sciences, 2018, 3, 159–164 CrossRef.
- S. Ilhan, F. Savaroğlu and F. Çolak, Antibacterial and Antifungal Activity of Corchorus olitorius L.(Molokhia) Extracts, Int. J. Nat. Eng. Sci., 2007, 1, 59–61 CAS.
- İ. l. Yücelşengün and A. Gargi, Comparative study of total phenolic contents, antioxidant and antimicrobial activities of different extracts of Corchorus olitorius L. growing in North Cyprus, Biyolojik Çeşitlilik ve Koruma, 2020, 13, 298–304 Search PubMed.
- A. B. Rowaiye, A. J. Ogugua, G. Ibeanu, D. Bur, M. T. Asala, O. B. Ogbeide, E. O. Abraham and H. B. Usman, Identifying potential natural inhibitors of Brucella melitensis Methionyl-tRNA synthetase through an in-silico approach, PLoS Neglected Trop. Dis., 2022, 16, e0009799 CrossRef CAS PubMed.
- R. Nakaziba, S. B. Amanya, C. D. Sesaazi, F. Byarugaba, J. Ogwal-Okeng and P. E. Alele, Antimicrobial Bioactivity and GC-MS Analysis of Different Extracts of Corchorus olitorius L. Leaves, Sci. World J., 2022, 2022, 9 Search PubMed.
- M. I. Khan, J. Akhtar and M. Ahmad, Pharmacognostic Studies and Antibacterial Activity of Corchorus olitorius L. Leaf, Pharmacogn. Res., 2022, 14, 474–482 CrossRef.
- F. Ahmed, In vitro hypoglycemic effects of molokhia leaves (Corchorus olitorius L.), Pharmacogn. Mag., 2021, 17, 246 CrossRef.
- A. Nduka, A. Oluwatoyin, A. Temitope and O. Odutola, Hypoglycemic and hypolipidemic effects of Corchorus olitorius leaves as a food supplement on rats with alloxan-induced diabet, Ukr. Biochem. J., 2020, 92, 63–69 CrossRef CAS.
- S. Mahgoub, F. El-Sharkawi and O. Badary, Effect of Molokhia Extract on Experimentally Induced Diabetes Mellitus, LAP LAMBERT Academic Publishing, 2016 Search PubMed.
- R. Nakaziba, A. Lubega, J. Ogwal-Okeng and P. E. Alele, Phytochemical Analysis, Acute Toxicity, as well as Antihyperglycemic and Antidiabetic Activities of Corchorus olitorius L. Leaf Extracts, Sci. World J., 2022, 2022 DOI:10.1155/2022/1376817.
- S. Parvin, M. Marzan, S. Rahman, A. K. Das, S. Haque and M. Rahmatullah, Preliminary phytochemical screening, antihyperglycemic, analgesic and toxicity studies on methanolic extract of aerial parts of Corchorus olitorius L, J. Appl. Pharm. Sci., 2015, 5, 68–71 CrossRef CAS.
- V. Barku, A. Boye and N. Quansah, Antioxidant and wound healing studies on the extracts of Corchorus olitorius leaf, Univ. Cape Coast Inst. Repository, 2013, 123456789/5638 Search PubMed.
- S. Yokoyama, K. Hiramoto, T. Fujikawa, H. Kondo, N. Konishi, S. Sudo, M. Iwashima and K. Ooi, Skin hydrating effects of Corchorus olitorius extract in a mouse model of atopic dermatitis, J. Cosmet., Dermatol. Sci. Appl., 2014, 4, 42341 Search PubMed.
- S. Yokoyama, K. Hiramoto, T. Fujikawa, H. Kondo, N. Konishi, S. Sudo, M. Iwashima and K. Ooi, Topical application of Corchorus olitorius leaf extract ameliorates atopic dermatitis in NC/Nga mice, Dermatol. Aspect., 2014, 2 DOI:10.7243/2053-530-2-3.
- A. K. Das, R. Sahu, T. K. Dua, S. Bag, M. Gangopadhyay, M. K. Sinha and S. Dewanjee, Arsenic-induced myocardial injury: protective role of Corchorus olitorius leaves, Food Chem. Toxicol., 2010, 48, 1210–1217 CrossRef CAS PubMed.
- F. Mesli, M. Ghalem, I. Daoud and S. Ghalem, Potential inhibitors of angiotensin converting enzyme 2 receptor of COVID-19 by Corchorus olitorius Linn using docking, molecular dynamics, conceptual DFT investigation and pharmacophore mapping, J. Biomol. Struct. Dyn., 2021, 1–13 Search PubMed.
- N. H. Shady, A. H. Altemani, F. H. Altemani, S. A. Maher, M. A. Elrehany, E. A. Saber, A. M. Badawi, F. M. A. El-Mordy, N. M. Mohamed and M. A. Abourehab, The Potential of Corchorus olitorius Seeds Buccal Films for Treatment of Recurrent Minor Aphthous Ulcerations in Human Volunteers, Molecules, 2022, 27, 7020 CrossRef CAS PubMed.
- G. Oboh, A. O. Ademiluyi, A. J. Akinyemi, T. Henle, J. A. Saliu and U. Schwarzenbolz, Inhibitory effect of polyphenol-rich extracts of jute leaf (Corchorus olitorius) on key enzyme linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting) in vitro, J. Funct. Foods, 2012, 4, 450–458 CrossRef CAS.
- R. Wagdy, R. M. Abdelkader, A. H. El-Khatib, M. W. Linscheid, N. Hamdi and H. Handoussa, Neuromodulatory Activity of Dietary Phenolics Derived from Corchorus olitorius L, J. Food Sci., 2019, 84, 1012–1022 CrossRef CAS PubMed.
- M. H. Shariare, H. B. Noor, J. H. Khan, J. Uddin, S. R. Ahamad, M. A. Altamimi, F. K. Alanazi and M. Kazi, Liposomal drug delivery of Corchorus olitorius leaf extract containing phytol using design of experiment (DoE): In-vitro anticancer and in-vivo anti-inflammatory studies, Colloids Surf., B, 2021, 199, 111543 CrossRef CAS PubMed.
- Z. A. Zakaria, M. Neelendran, S. Pubalan, M. Sulaiman and C. Fatimah, Effects of opioid and non-opioid antagonists, pH and enzymes on Corchorus olitorius antinociception in mice, Adv. Tradit. Med., 2006, 6, 186–195 Search PubMed.
- M. T. Islam, R. M. de Freitas, I. Sultana, J. A. Hossain, A. Mahmood and A. K. Kundu, Revision: Chemical and biological activities of Corchorus olitorius L, Am. J. PharmTech Res., 2013, 3, 337–348 Search PubMed.
- K. Dhayal and D. Kumar, Qualitative and Quantitative Evaluation of Flavonoids from Corchorus olitorius L, J. Drug Deliv. Therapeut., 2019, 9, 348–352 CrossRef.
- A. R. B. Yakoub, O. Abdehedi, M. Jridi, W. Elfalleh, M. Nasri and A. Ferchichi, Flavonoids, phenols, antioxidant, and antimicrobial activities in various extracts from Tossa jute leave (Corchorus olitorus L.), Ind. Crops Prod., 2018, 118, 206–213 CrossRef.
- D. Driss, M. Kaoubaa, R. B. Mansour, F. Kallel, B. Eddine Abdelmalek and S. E. Chaabouni, Antioxidant, antimutagenic and cytotoxic properties of essential oil from Corchorus olitorius L. flowers and leaf, Free Radic. Antioxidants, 2016, 6, 34–43 CrossRef CAS.
- A. O. Ademiluyi, G. Oboh, F. P. Aragbaiye, S. I. Oyeleye and O. B. Ogunsuyi, Antioxidant properties and in vitro α-amylase and α-glucosidase inhibitory properties of phenolics constituents from different varieties of Corchorus spp, Journal of Taibah University Medical Sciences, 2015, 10, 278–287 CrossRef.
- M. Ali, K. Ahmed, H. Hossain, B. Roy, B. Rokeya, M. Rahman, I. Jahan and M. Rahman, Total antioxidant capacity and profiling of polyphenolic compounds in jute leaves by hplc-dad, Food Res., 2021, 5, 343–348 Search PubMed.
- H. Handoussa, R. Hanafi, I. Eddiasty, M. El-Gendy, A. El Khatib, M. Linscheid, L. Mahran and N. Ayoub, Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from Corchorus olitorius and Vitis vinifera, J. Funct. Foods, 2013, 5, 1204–1216 CrossRef CAS.
- A. Z. Hassan, M. K. Mekhael, A. G. Hanna, A. Simon, G. Tóth and H. Duddeck, Phytochemical investigation of Corchorus olitorius and Corchorus capsularis (Family Tiliaceae) that grow in Egypt, Egypt. Pharm. J., 2019, 18, 123 Search PubMed.
- C. Y. Ragasa, J. L. A. Vivar, M. C. S. Tan and C. C. Shen, Chemical constituents of Corchorus olitorius L, International Journal of Pharmacognosy and Phytochemical Research, 2016, 8, 2085 Search PubMed.
- D. Ramadevi and S. Ganapaty, Phytochemical Examination of Corchorus aestuans (Tiliaceae) Capsule, Int. J. Pharmacogn., 2012, 4, 54–58 Search PubMed.
- D. Ramadevi and S. Ganapathy, Fusidic acid from Corchorus aestuans L, J. Global Trends Pharm. Sci., 2012, 3, 550–557 Search PubMed.
- H. T. Hasan and E. J. Kadhim, Phytochemical investigation of leaves and seeds of Corchorus olitorius L. Cultivated in Iraq, Asian J. Pharm. Clin. Res., 2018, 11, 408–417 CrossRef CAS.
- M. Ogawa, K. Hayasi, S. Tomimori, N. Konishi and O. Nakayama, Contents of strophanthidin glycosides and digitoxigenin glycosides in “moroheiya” (Corchorus olitorius L.) and its products, J. Jpn. Soc. Food Sci. Technol., 2002, 49, 282–287 CrossRef CAS.
- D. Ramadevi, Phytochemical examination of Corchorus capsularis leaves, Int. J. Curr. Pharmaceut. Res., 2014, 4, 42–46 Search PubMed.
| This journal is © The Royal Society of Chemistry 2022 |