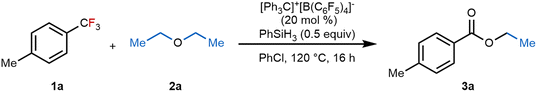Open Access Article
Open Access ArticleConversion of trifluoromethyls into esters along with polyether upcycling via cation-transfer-catalyzed C–O/C–F metathesis
Zhuojun
Li
,
Xiangqian
Shi
,
Dongke
Zhang
and
Qian
Wu
 *
*
State Key Laboratory of Medical Chemical Biology, College of Pharmacy, Academy for Advanced Interdisciplinary Studies, Tianjin Key Laboratory of Molecular Drug Research, Nankai University, 38 Tongyan Road, Jinnan District, Tianjin, 300350, China. E-mail: wuqian@nankai.edu.cn
First published on 1st April 2026
Abstract
Ester and trifluoromethyl groups play crucial roles in chemistry and drug design. However, the implementation of interconversion from a trifluoromethyl into an ester under wide conditions is not an easy task, because the C–F bond of the trifluoromethyl group possesses high bond energy and is extremely difficult to break. Herein, we present for the first time a silylium-ion-initiated method for intermolecular C–F/C–O metathesis, enabling selective activation of inert C(sp3)–F and C(sp3)–O bonds. This approach facilitates the defluoroalkoxylation of trifluoromethyl-substituted substrates and polyfluoroalkanes when combined with ethers or polyethers, thus building a bridge to convert trifluoromethyl groups into ester/ketone groups, along with polyether upcycling. We provide crystallographic evidence that clearly confirms that this reaction involves the formation of silyloxonium ions and Meerwein-type salt intermediates, followed by cationic ligand transformations.
Introduction
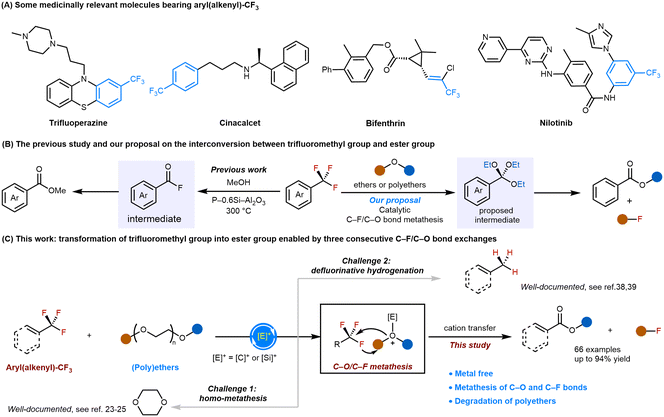
The functionalization of inert bonds represents a fundamental challenge in organic chemistry. Central to this pursuit is the carbon–fluorine bond, which plays a crucial role in synthetic chemistry and drug design (Scheme 1A), while also contributing to persistent fluorinated waste.1–5 Despite its exceptional strength (up to ∼130 kcal mol−1 for C(sp3)–F centers),6 the trifluoromethyl group, which is readily available in commercially abundant compounds, has emerged as a versatile synthetic handle. Recent advances have enabled its use in diverse catalytic transformations, including hydrodefluorination, halogen exchange, and C–C/C–heteroatom bond formation.7–24 Encouraged by these advances, the development of direct and efficient methods to convert trifluoromethyl groups into diverse functionalities through mechanistically novel pathways is scientifically interesting. Such methods would also significantly facilitate access to chemically useful functional groups.Catalytic chemical bond metathesis reactions have garnered significant attention in recent years, as they provide low-energy and redox-neutral pathways for bond activation and formation, enabling the cleavage of strong and inert bonds that are difficult to break through other pathways.25–32 Inspired by recent elegant examples of acidic main-group-catalyzed C–O single bond metathesis33–35 as well as C–F bond cleavage,36–40 we wondered if metathesis involving C–F and C–O bond exchange could proceed smoothly under mild conditions in the presence of acidic main-group catalysts. If this method worked smoothly, it would enable the interconversion between trifluoromethyl groups and ester groups via consecutive C–F/C–O bond exchanges, although a high reaction temperature would decrease its synthetic value in practical applications (Scheme 1B, right). Notably, Dai and coworkers recently achieved this conversion using a heterogeneous catalyst, albeit at a high temperature of 300 °C, underscoring the need for a milder catalytic protocol (Scheme 1B, left).41 Despite recent advancements in main group catalysis, main-group-catalyzed C–F/C–O bond metathesis reactions have never been realized due to the high bond dissociation energies and selectivity challenges. Several challenges for such a strategy are anticipated: (1) the well-documented homo-metathesis of C–O bonds to generate a cyclic ether, and (2) the competitive defluorinative hydrogenation of the C–F bond (Scheme 1C).42,43
Silylium ions are strongly Lewis-acidic silicon-based cations that have shown exceptional ability to activate various inert bonds.44–52 More recently, Oestreich and co-workers discovered that the silylium ion can also efficiently activate the C–O bond in methanol, furnishing the methylation of arenes.53 Inspired by these catalytic patterns, we questioned whether the carbenium ion formed after fluoride abstraction could also activate ether C–O bonds via a reactive Meerwein-type salt54,55 intermediate. If this transformation were achievable, this approach would represent a rare metal-free example of intermolecular bond exchange between two robust single bonds (C–F and C–O), incorporating silicon, the second-most-abundant element in Earth's crust, as a viable catalyst.
Here, we report the implementation of the C–F/C–O bond metathesis reaction via the employment of strongly Lewis-acidic silicon-based silylium cations, which were generated in situ via hydride abstraction using excess Si–H, as the catalysts, and ethers or polyethers as the defluoroalkoxylation reagent (Scheme 1C). This C–F/C–O bond metathesis reaction is efficient and scalable, providing a catalytic means of transforming aryl–CF3 or alkenyl–CF3 groups into aryl–COOR or alkenyl–COOR groups.
Results and discussion
With the selectivity challenges of competing C–O homo-metathesis and hydrodefluorination in mind, we began with the reaction between (trifluoromethyl)toluene 1a and diethyl ether 2a in the weakly coordinating solvent chlorobenzene (Table 1). We evaluated the in situ generation of different silylium ions by treating phenylsilane (0.5 equiv.) with [Ph3C]+[B(C6F5)4]− (20 mol%) at room temperature for 15 minutes before adding the substrate. Gratifyingly, the defluoroalkoxylation proceeded efficiently, affording ester product 3a in 95% yield (GC-MS) after 16 hours at 120 °C, and no defluorohydrogenative product was observed (entry 1). This result supports our hypothesis that C–F/C–O bond exchange can occur. The decomposition of a triethoxyalkyl intermediate was proposed to furnish the ester product under strongly acidic conditions. Reducing the [Ph3C]+[B(C6F5)4]− initiator loading to 15 mol% led to a moderate yield (entry 2). Other Lewis acids, including the direct use of [Ph3C]+[B(C6F5)4]−, as well as AlCl3 and B(C6F5)3, proved less effective, delivering lower yields between 16% and 83% (entries 3–5). Additionally, using Et3SiH or MeSiHCl2 as the silicon source gave decreased yields of 33% and 19%, respectively (entries 6 and 7). Alternative solvents, including toluene and dichlorobenzene, were also tested, but did not outperform chlorobenzene (entries 8 and 9). Finally, temperature optimization revealed that a temperature of 120 °C is critical for high efficiency, as lowering the temperature to 100 °C led to a decreased yield (entry 10).| Entry | Deviation from standard conditions | Yieldb (%) |
|---|---|---|
| a Reaction conditions: 1a (0.1 mmol), 2a (0.2 mL), indicated initiator (15–20 mol%), PhCl, 120 °C, 16 h, Ar. b Yields were determined via GC-MS using naphthalene as an internal standard. c Isolated yield. | ||
| 1 | None | 95 (89)c |
| 2 | [Ph3C]+[B(C6F5)4]− 15 mol% instead of 20 mol% | 70 |
| 3 | [Ph3C]+[B(C6F5)4]− 20 mol% without PhSiH3 | 13 |
| 4 | AlCl3 (20 mol%) | 7 |
| 5 | B(C6F5)3 | 83 |
| 6 | Et3SiH (0.5 equiv.), [Ph3C]+[B(C6F5)4]− (20 mol%) | 33 |
| 7 | MeSiHCl2 (0.5 equiv.), [Ph3C]+[B(C6F5)4]− (20 mol%) | 19 |
| 8 | Toluene instead of PhCl | 62 |
| 9 | 1,2 Dichlorobenzene instead of PhCl | 81 |
| 10 | 100 °C instead of 120 °C | 51 |
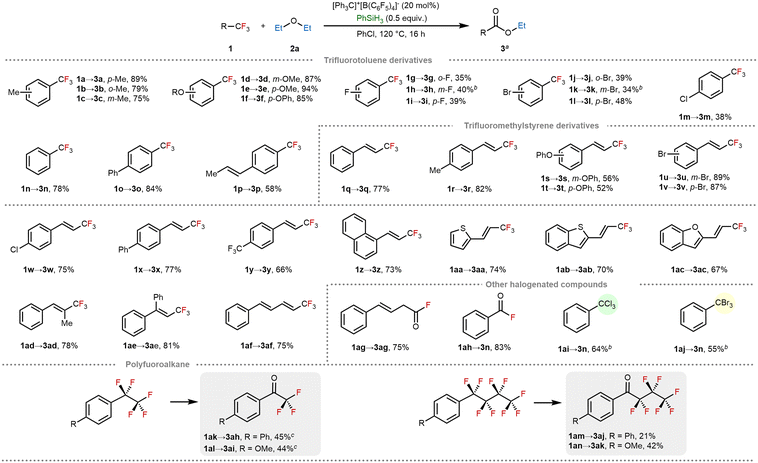
Having established the feasibility of silylium-ion-initiated C–F/C–O metathesis, we next evaluated the substrate scope and limitations of this cation transfer protocol (Scheme 2). Substrates with methyl or alkoxy groups at various positions on the aromatic ring reacted well, delivering the products in good-to-excellent yields (1a–1f), with minimal impact from steric hindrance. However, halogenated analogues showed reduced reactivity, affording only moderate yields (1g–1m). Meta-substituted halogenated substrates were particularly challenging and required an increased amount of silane (3.0 equiv.) to achieve satisfactory conversion (1h, 1k). Conjugated substituents, including phenyl and alkenyl groups, remained intact under the reaction conditions (1o, 1p), highlighting the functional group tolerance of the protocol. Moreover, a range of trifluoromethylstyrene derivatives bearing various substituents, including electron-donating groups (e.g., methyl), halogens (Cl, Br), phenyl, and naphthyl groups, were well tolerated, affording the corresponding ester products in good yields (1q–1x, 1z). Notably, when a para-trifluoromethyl group was introduced on the aromatic ring (1y), the reaction occurred selectively at the allylic position, leaving the benzylic C–F bonds untouched. Heteroaromatic substrates bearing electron-donating sulfur or oxygen atoms, such as thiophenyl, benzothiophenyl, and benzofuranyl derivatives, also underwent smooth transformation under these strongly Lewis-acidic conditions (1aa–1ac). Importantly, no Friedel–Crafts alkylation or heteroarene degradation was observed. Additionally, alkene-substituted trifluoromethylstyrenes with 1- or 2-position substituents reacted efficiently to yield the desired esters (1ad–1ae), and a terminal CF3-containing conjugated diene also delivered the product in 75% yield (1af). The method also proved effective for acyl fluorides: both alkyl and aryl variants underwent smooth C–F/C–O metathesis to provide the ester products in good yields (1ag, 1ah). We further explored the compatibility of other carbon–halogen bonds. Trichlorotoluene and tribromotoluene produced the corresponding esters in moderate yields under the optimized conditions, suggesting partial extension of the reactivity beyond fluorinated substrates (1ai, 1aj).
Given the environmental importance of addressing polyfluoroalkyl substances (PFAS), we explored the applicability of the protocol to polyfluoroalkanes. Benzylic C–F bonds could be selectively activated, producing phenyl polyfluorinated ketones in moderate yields (1ak–1an). However, attempts to functionalize the less-activated C–F bonds in aliphatic polyfluoroalkanes were unsuccessful, even using elevated temperatures (140 °C) and prolonged reaction times (48 h).
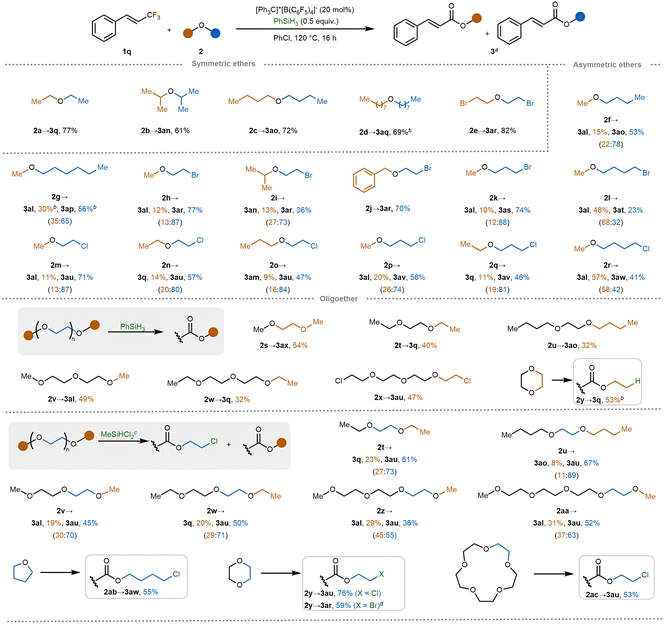
With the feasibility of C–F/C–O metathesis established, we next explored the scope of ethers as alkoxylation partners using trifluoromethylstyrene 1q as the model substrate (Scheme 3). Symmetrical ethers bearing bulkier or longer alkyl chains, such as isopropyl, n-butyl, and n-octyl groups, performed well, delivering the corresponding esters in good yields (2a–2d). Notably, a bromo-substituted ether also proved compatible,56 affording product 2e without evidence of debromination. When asymmetric ethers were used, the product distribution became more complex, raising the question of regioselectivity—specifically, which alkyl fragment acts as the fluoride acceptor and which is incorporated into the ester product. Methylbutyl and methylhexyl ethers gave the desired products in 68% and 86% yield, respectively (2f, 2g), with product ratios favoring incorporation of the longer alkyl group (short:long = 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 78 and 35
78 and 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 65). This suggests that steric and/or electronic factors may bias the direction of alkyl transfer. To further investigate this trend, we examined bromoethyl ethers with various alkyl substituents. In these cases, smaller groups such as methyl, isopropyl, and benzyl were preferentially eliminated, while the bromoethoxy moiety was mainly retained in the product (2h–2j). A similar pattern was observed with methyl bromopropyl ether, with the bromopropyl group being selectively transferred to the product (2k). These results support a mechanistic model in which the smaller, more volatile alkyl group is more readily abstracted as a fluoroalkane, while the bulkier fragment forms the ester. Interestingly, when the bromoalkyl chain was extended to four carbons, the regioselectivity was reversed, with a higher proportion of the methyl-derived ester being observed (Me
65). This suggests that steric and/or electronic factors may bias the direction of alkyl transfer. To further investigate this trend, we examined bromoethyl ethers with various alkyl substituents. In these cases, smaller groups such as methyl, isopropyl, and benzyl were preferentially eliminated, while the bromoethoxy moiety was mainly retained in the product (2h–2j). A similar pattern was observed with methyl bromopropyl ether, with the bromopropyl group being selectively transferred to the product (2k). These results support a mechanistic model in which the smaller, more volatile alkyl group is more readily abstracted as a fluoroalkane, while the bulkier fragment forms the ester. Interestingly, when the bromoalkyl chain was extended to four carbons, the regioselectivity was reversed, with a higher proportion of the methyl-derived ester being observed (Me![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) bromobutyl = 68
bromobutyl = 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32, 2l). This shift may result from the increased steric hindrance of the longer alkyl chain during the defluoroalkoxylation step. Chloroalkyl ethers showed similar regioselectivity under the same conditions (2m–2r). In contrast, diaryl and arylalkyl ethers were unreactive under the optimized conditions.
32, 2l). This shift may result from the increased steric hindrance of the longer alkyl chain during the defluoroalkoxylation step. Chloroalkyl ethers showed similar regioselectivity under the same conditions (2m–2r). In contrast, diaryl and arylalkyl ethers were unreactive under the optimized conditions.
We then turned to glycol, diglycol, and higher glycol ethers, which are common components in pharmaceuticals, cosmetics, and consumer products. Using phenylsilane as the silicon source, we observed a reversal of selectivity relative to dialkyl ethers: the terminal alkyl groups of the glycol ethers were selectively incorporated into the esters (2s–2x), albeit in moderate yields. The use of excess phenylsilane (3.0 equiv.) fulfilled two roles in the reaction system: generating the catalytically active silylium species and supplying protons to promote the ring-opening of cyclic ether intermediates. Under these conditions, transfer of the ethylene linkage occurred efficiently, affording the ester product in 53% yield (2y). This result led us to hypothesize that product distribution might be influenced by the terminating anion. Indeed, when chlorosilanes were used instead of phenylsilane, mixed products derived from both chloroethyl and terminal alkyl fragments were observed. In these cases, the chloroethyl-derived products predominated (2t–2aa). A range of cyclic ethers, including tetrahydrofuran (THF), dioxane, and 15-crown-5, also proved reactive under chlorosilane-mediated conditions, yielding the corresponding chloroalkyl esters in good yields (2ab, 2y and 2ac). These findings highlight the broad substrate compatibility and functional group tolerance of the cation-transfer-enabled C–F/C–O metathesis.
Encouraged by the successful application of our protocol to oligoethers, we next investigated its utility in C–F/C–O metathesis involving polyethers. Specifically, we examined polyethylene glycol (PEG) and polytetrahydrofuran (polyTHF), each with a number-average molecular weight of ∼2000 g mol−1. Under reaction conditions using [Ph3C]+[B(C6F5)4]− (20 mol%) and SiCl4 (3.0 equiv.), both polymers underwent decomposition, accompanied by activation of the C–F bond. This transformation afforded the corresponding chloroethyl ester products 3au and 3aw in reasonable yields (Scheme 4A and 4B). We further extended the protocol to poly(vinylethyl ether) (PEVE), a polymer featuring ethoxy side chains. When treated with MeSiHCl2 (3.0 equiv.) instead of SiCl4, the reaction successfully transferred the ethoxy moiety to the ester product, yielding the desired compound in 54% yield (Scheme 4C). Given the ubiquitous use of polymers in modern materials and products, developing efficient strategies for their chemical recycling is of growing importance. These results demonstrate the potential of our cation-shuffle-enabled C–F/C–O metathesis to not only functionalize fluorinated compounds but also contribute to polymer upcycling by transforming polyethers under mild conditions.
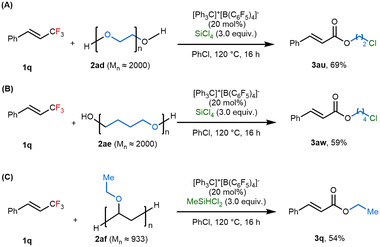 | ||
| Scheme 4 Application of C–O/C–F metathesis in polyethers. (A) The reaction of 1q with (A) PEG (2ad), (B) polyTHF (2ae) or (C) PEVE (2af). | ||
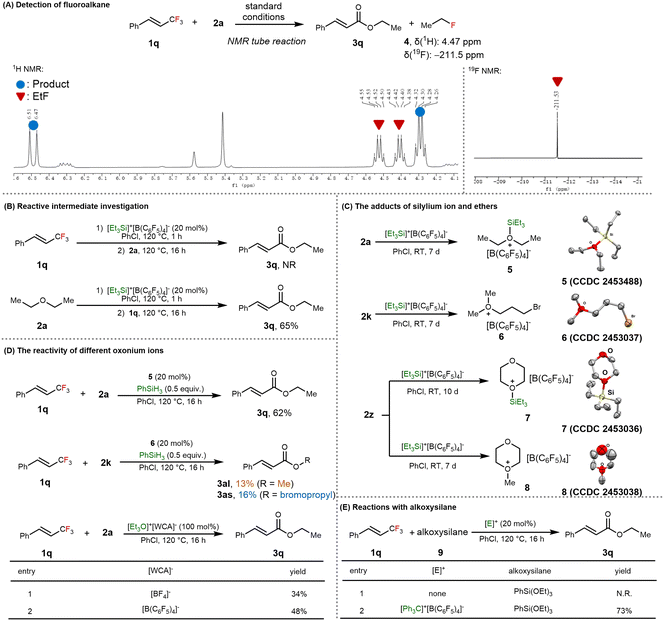
To gain mechanistic insight into the cation-transfer-enabled C–F/C–O metathesis, we began by analyzing the reaction mixture of 1q and 2a using NMR spectroscopy. In the 1H NMR spectrum, a diagnostic doublet of quartets at δ = 4.47 ppm with a 2J(F,H) coupling constant of 47.1 Hz indicated the formation of CH3CH2F, which was further confirmed by the characteristic 19F NMR signal at δ = −211.5 ppm (Scheme 5A).57,58 Quantitative NMR analysis using an internal standard revealed that fluoroethane was formed in an approximately 70% ratio relative to the internal standard, indicating that fluoroalkane generation could be the dominant fate of the fluoride (Fig. S1). These results provided direct evidence of C–F bond cleavage and replacement by an alkoxy group. To clarify the sequence of bond activation, we conducted control experiments to determine whether the silylium ion preferentially engages with the C–F or C–O bond. When the reaction mixture was first treated with 1q at 120 °C or room temperature, followed by the addition of 2a and heating, only a trace amount of the ester product was observed, along with an allylic isomer detected via NMR (Scheme 5B, top, and Fig. S2). However, reversing the order, i.e., adding 1q after pre-mixing the silylium ion and 2a, resulted in a 65% yield of ester 3a (Scheme 5B, bottom). This suggests that the silylium ion first coordinates with the ether oxygen rather than abstracting fluoride directly from the trifluoromethyl group, and the defluoroalkoxylation is likely to proceed via a concerted mechanism.
To support this hypothesis, we mixed stoichiometric amounts of Et3Si+[B(C6F5)4]− and 2a in chlorobenzene. The resulting silyloxonium ion 5 was successfully isolated and characterized (Scheme 5C). When the asymmetric ether 2k was used instead, we obtained a crystal structure consistent with Meerwein-type salt 6 bearing two methyl groups and one bromopropyl moiety. To further explore the coordination of silylium and ether in the absence of C–F bonds, we treated triethylene glycol dimethyl ether (TEGDME) with the silylium reagent. This led to the formation of cyclic oxonium ions 7 and 8 with a silyl or methyl substituent, respectively. These structures provide the first crystallographic evidence for the Lewis-acid-catalyzed intramolecular C–O/C–O metathesis of aliphatic ethers, aligning with prior reports by Morandi and Greb. Single crystals of intermediates 5–8 were obtained via vapor diffusion from hexane at room temperature.59 To verify the roles of oxonium ions as mechanistic intermediates, we directly applied silyloxonium ion 5 and Meerwein-type salt 6 in catalytic reactions between 1q and their corresponding ethers (2a and 2k). The reaction involving 5 gave the ester product in yields comparable to those under standard conditions, while 6 produced a mixture of ester products in diminished yield (Scheme 5D). Furthermore, when the reaction was conducted using stoichiometric Meerwein-type salts Et3O+BF4− and Et3O+[B(C6F5)4]− in the absence of silane, both the ester product and fluoroethane were detected, confirming that fluorine transfer from carbon to carbon is operative (Scheme 5D, bottom, and Fig. S3). To the best of our knowledge, this mode of reactivity is unprecedented and offers a new mechanistic paradigm for C–F functionalization. Moreover, these results suggest that both oxonium species are catalytically relevant, but the silyloxonium ion is likely the more reactive and effective intermediate for alkyl group transfer. The reaction of 1a with an alkoxysilane (9) proceeded smoothly in the presence of a catalytic amount of trityl cation, further confirming the participation of silyloxonium species in the reaction mechanism (Scheme 5E). Finally, when (triethoxymethyl)benzene was subjected to the standard reaction conditions, the corresponding ester product was obtained in 89% yield, supporting its role as an intermediate in the catalytic cycle (for details, see the SI).60
Based on our experimental findings, we propose that the formation of oxonium intermediates and subsequent ligand exchange processes are keys to the success of the cation-transfer-enabled C–F/C–O metathesis. The proposed catalytic cycle is depicted in Scheme 6. The reaction is initiated by hydride abstraction from a hydrosilane by a trityl salt, generating highly electrophilic silylium ion 10. This species coordinates with an ether to form silyloxonium ion 11, which serves as a central intermediate in the catalytic cycle. In the catalytic cycle at the right side, the fluoride on 1 is abstracted by a cationic alkyl group on 11, accompanied by the formation of fluoroalkane 4. Simultaneously, RCF2+ was transferred to the oxygen atom, resulting in the formation of the new silyloxonium species 12. The silicon cation then jumped to another ether molecule to give the formation of alkoxylation intermediate 16, completing the C–F/C–O metathesis and regenerating the catalytic intermediate, i.e., the silyloxonium ion. Alternatively, the silyl group may act as the fluoride acceptor, forming Meerwein-type intermediate 14 containing three alkyl substituents. This species could participate in alkyl cation transfer, producing the new Meerwein-type salt 15 attached to the substrate moiety and simultaneously facilitating alkoxylation of the C–F bond to continue the catalytic cycle. Notably, the key compound 16 produced by both of these mechanistic routes could undergo a sequential C–O/C–F bond exchange followed by elimination to furnish the final ester product 3.
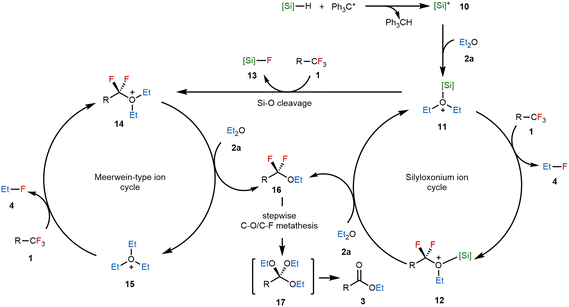 | ||
| Scheme 6 Proposed mechanism of cation-transfer-enabled C–F/C–O metathesis. The counter anion has been omitted for clarity. | ||
Conclusions
In this study, we have developed a silylium-ion-initiated method for the C–F/C–O metathesis of a trifluoromethyl group and an ether, which provides a versatile and efficient approach to activate and transform otherwise inert C–F and C–O bonds. The ability to selectively exchange these bonds under mild conditions offers significant potential for resource utilization and environmental sustainability, particularly in the context of polyfluoroalkyl substances and biomass-derived polyethers. Mechanistic investigations confirmed the involvement of key intermediates, such as silyloxonium ions and Meerwein-type salts, which play crucial roles in the transformation. These findings open new avenues for further exploration of cation shuffle catalysis in organic synthesis, offering a promising strategy for the selective activation and transformation of stable chemical bonds in both synthetic and environmental contexts.Author contributions
Z. L., D. Z. and X. S. performed the experiments. Q. W. conceived the concept, directed the project and wrote the paper.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2453488 (5), 2453037 (6), 2453036 (7) and 2453038 (8) contain the supplementary crystallographic data for this paper.59a–dThe data that support the findings of this study are available in the supplementary information (SI) of this article. Supplementary information: experimental procedures, single-crystal X-ray data, NMR and HRMS spectra. See DOI: https://doi.org/10.1039/d5sc10208a.
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (no. 22371133, 22171142, 22188101), the Fundamental Research Funds for the Central Universities, Nankai University (no. 63243135, 63241203, 63253258), College of Pharmacy, Academy for Advanced Interdisciplinary Studies and Nankai University.Notes and references
- D. O'Hagan, Chem. Soc. Rev., 2008, 37, 308–319 RSC.
- S. Murai, Topics in Organometallic Chemistry: Activation of Unreactive Bonds and Organic Synthesis, Springer-Verlag, Berlin, 1999 Search PubMed.
- S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Chem. Soc. Rev., 2008, 37, 320–330 RSC.
- M. Inoue, Y. Sumii and N. Shibata, ACS Omega, 2020, 5, 10633–10640 CrossRef CAS PubMed.
- A. Leeson, T. Thompson, H. F. Stroo, R. H. Anderson, J. Speicher, M. A. Mills, J. Willey, C. Coyle, R. Ghosh, C. Lebrón and C. Patton, Environ. Toxicol. Chem., 2021, 40, 24–36 CrossRef CAS PubMed.
- D. M. Lemal, J. Org. Chem., 2004, 69, 1–11 CrossRef CAS PubMed.
- H. Amii and K. Uneyama, Chem. Rev., 2009, 109, 2119–2183 CrossRef CAS PubMed.
- T. Ahrens, J. Kohlmann, M. Ahrens and T. Braun, Chem. Rev., 2015, 115, 931–972 CrossRef CAS PubMed.
- J.-D. Hamel and J.-F. Paquin, Chem. Commun., 2018, 54, 10224–10239 RSC.
- F. Jaroschik, Chem. Eur. J., 2018, 24, 14572–14582 CrossRef CAS PubMed.
- Y. Wang and A. Liu, Chem. Soc. Rev., 2020, 49, 4906–4925 RSC.
- L. Zhou, Molecules, 2021, 26, 7051 CrossRef CAS PubMed.
- G. Yan, K. Qiu and M. Guo, Org. Chem. Front., 2021, 8, 3915–3942 RSC.
- H.-J. Ai, X. Ma, Q. Song and X.-F. Wu, Sci. China. Chem., 2021, 64, 1630–1659 CrossRef CAS.
- X. An, Z. Feng, L. Huang, Y. Yang and Z. Liu, Chin. J. Org. Chem., 2021, 41, 4554–4564 CrossRef CAS.
- T. T. Simur, T. Ye, Y.-J. Yu, F.-L. Zhang and Y.-F. Wang, Chin. Chem. Lett., 2022, 33, 1193–1198 CrossRef CAS.
- G. Yan, Chem. Eur. J., 2022, 28, e202200231 CrossRef CAS PubMed.
- Y. Nishimoto, N. Sugiharaa and M. Yasuda, Synthesis, 2022, 54, 2765–2777 CrossRef CAS.
- S. Yoshida, Chem. Rec., 2023, 23, e202200308 CrossRef CAS PubMed.
- L. V. Hooker and J. S. Bandar, Angew. Chem., Int. Ed., 2023, 62, e202308880 CrossRef CAS PubMed.
- D. Ge, J.-W. Chen, Y.-L. Chen, M. Ma, Z.-L. Shen and X.-Q. Chu, Org. Chem. Front., 2023, 10, 3909 RSC.
- S. Roy and T. Besset, JACS Au, 2025, 5, 466–485 CrossRef CAS PubMed.
- C. Cheng, Y.-F. Huang, L.-J. Wang, M.-J. Luo and Q. Xiao, Adv. Synth. Catal., 2025, 367, e2025888 Search PubMed.
- L. Zeng, Y. Zhang, M. Hu, X.-H. Ouyang and J.-H. Li, Chem. Soc. Rev., 2026, 55, 955–1007 RSC.
- B. N. Bhawal and B. Morandi, Angew. Chem., Int. Ed., 2019, 58, 10074–10103 CrossRef CAS.
- M. Liu, N. Yan, H. Tian, B. Li and D. Zhao, Angew. Chem., Int. Ed., 2024, 63, e202319187 CrossRef CAS PubMed.
- R. Li, B. Li, H. Zhang, C.-W. Ju, Y. Qin, X.-S. Xue and D. Zhao, Nat. Chem., 2021, 13, 1006–1016 CrossRef CAS PubMed.
- C. Liu, D. Chen, Y. Fu, F. Wang, J. Luo and S. Huang, Org. Lett., 2020, 22, 5701–5705 CrossRef CAS PubMed.
- Y. Ma, L. Zhang, Y. Luo, M. Nishiura and Z. Hou, J. Am. Chem. Soc., 2017, 139, 12434–12437 CrossRef CAS PubMed.
- Z. Lian, B. N. Bhawal, P. Yu and B. Morandi, Science, 2017, 356, 1059–1063 CrossRef CAS PubMed.
- S. Okumura, F. Sun, N. Ishida and M. Murakami, J. Am. Chem. Soc., 2017, 139, 12414–12417 CrossRef CAS PubMed.
- N. A. Stephenson, J. Zhu, S. H. Gellman and S. S. Stahl, J. Am. Chem. Soc., 2009, 131, 10003–10008 CrossRef CAS PubMed.
- K. Leszczyńska, A. Mix, R. J. F. Berger, B. Rummel, B. Neumann, H.-G. Stammler and P. Jutzi, Angew. Chem., Int. Ed., 2011, 50, 6843–6846 CrossRef.
- N. Ansmann, T. Thorwart and L. Greb, Angew. Chem., Int. Ed., 2022, 61, e202210132 CrossRef CAS.
- F. S. Tschernuth, L. Bichlmaier and S. Inoue, ChemCatChem, 2023, 15, e202300281 CrossRef CAS.
- N. Sugihara, Y. Nishimoto, Y. Osakada, M. Fujitsuka, M. Abe and M. Yasuda, Angew. Chem., Int. Ed., 2024, 63, e202401117 CrossRef CAS PubMed.
- B. Trang, Y. Li, X.-S. Xue, M. Ateia, K. N. Houk and W. R. Dichtel, Science, 2022, 377, 839–845 CrossRef CAS PubMed.
- K. Lye and R. D. Young, Chem. Sci., 2024, 15, 2712–2724 RSC.
- Y. Zhu, J. Jia, X. Song, C. Gong and Y. Xia, Chem. Sci., 2024, 15, 13800–13806 RSC.
- J. Koo, W. Kim, B. H. Jhun, S. Park, D. Song, Y. You and H. G. Lee, J. Am. Chem. Soc., 2024, 146, 22874–22880 CrossRef CAS PubMed.
- M. Cai, X. Zhang, H. Zhu, X. Wang, Y. Chen and L. Dai, J. Mater. Sci., 2024, 59, 1896–1913 CrossRef CAS.
- C. Douvris and O. V. Ozerov, Science, 2008, 321, 1188–1190 CrossRef CAS PubMed.
- V. J. Scott, R. Çelenligil-Çetin and O. V. Ozerov, J. Am. Chem. Soc., 2005, 127, 2852–2853 CrossRef CAS PubMed.
- H. F. T. Klare, L. Albers, L. Süsse, S. Keess, T. Müller and M. Oestreich, Chem. Rev., 2021, 121, 5889–5985 CrossRef CAS PubMed.
- J. C. L. Walker, H. F. T. Klare and M. Oestreich, Nat. Rev. Chem., 2020, 4, 54–62 CrossRef CAS.
- T. Stahl, H. F. T. Klare and M. Oestreich, ACS Catal., 2013, 3, 1578–1587 CrossRef CAS.
- T. He, H. F. T. Klare and M. Oestreich, Nature, 2023, 623, 538–543 CrossRef CAS PubMed.
- Q. Wu, E. Irran, R. Müller, M. Kaupp, H. F. T. Klare and M. Oestreich, Science, 2019, 365, 168–172 CrossRef CAS PubMed.
- S. Popov, B. Shao, A. L. Bagdasarian, T. R. Benton, L. Zou, Z. Yang, K. N. Houk and H. M. Nelson, Science, 2018, 361, 381–387 CrossRef CAS PubMed.
- B. Shao, A. L. Bagdasarian, S. Popov and H. M. Nelson, Science, 2017, 355, 1403–1407 CrossRef CAS PubMed.
- J. Dosso, J. Tasseroul, F. Fasano, D. Marinelli and N. Biot, Angew. Chem., Int. Ed., 2017, 56, 4483–4487 CrossRef CAS PubMed.
- O. Allemann, S. Duttwyler, P. Romanato, K. K. Baldridge and J. S. Siegel, Science, 2011, 332, 574–577 CrossRef CAS PubMed.
- T. He, H. F. T. Klare and M. Oestreich, J. Am. Chem. Soc., 2023, 145, 3795–3801 CrossRef CAS PubMed.
- V. G. Granik, B. M. Pyatin and R. G. Glushkov, Russ. Chem. Rev., 1971, 40, 747–759 CrossRef.
- H. Meerwein, G. Hinz, P. Hofmann, E. Kroning, E. Pfeil and J. Prakt, Chem, 1937, 147, 257–285 CAS.
- C. Douvris, C. M. Nagaraja, C.-H. Chen, B. M. Foxman and O. V. Ozerov, J. Am. Chem. Soc., 2010, 132, 4946–4953 CrossRef CAS PubMed.
- M. Talavera, G. Meißner, S. G. Rachor and T. Braun, Chem. Commun., 2020, 56, 4452–4455 RSC.
- J. W. Emsley, L. Phillips and V. Wray, Prog. in NMR Spectros., 1975, 10, 105–122 Search PubMed.
- (a) CCDC 2453488: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nc1sw; (b) CCDC 2453037: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nbl7v; (c) CCDC 2453036: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nbl6t; (d) CCDC 2453038: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nbl8w.
- Ketal intermediates were observed in the reaction of polyfluoroalkyl substances (1ak–1an) by the GC-MS detection, suggesting a sequential C–O/C–F metathesis in the reaction process..
| This journal is © The Royal Society of Chemistry 2026 |