Open Access Article
Open Access ArticleRegulating KBBF-like structures via a charge-assisted hydrogen-bonded framework to enable easily grown ultraviolet nonlinear optical crystals
Mingshu
Zhang
 a,
Shuya
Zhao
a,
Zhen-Cheng
Wu
b,
Yue
Yin
a,
Zheyu
Zhang
a,
Jiafeng
Chen
a,
Sheng-Ping
Guo
a,
Shuya
Zhao
a,
Zhen-Cheng
Wu
b,
Yue
Yin
a,
Zheyu
Zhang
a,
Jiafeng
Chen
a,
Sheng-Ping
Guo
 *b and
Yan
Zhou
*b and
Yan
Zhou
 *a
*a
aSchool of Chemical Science and Technology, Yunnan University, Kunming, Yunnan 650500, P. R. China. E-mail: yanzhou@ynu.edu.cn
bYunnan Key Laboratory of Electromagnetic Materials and Devices, School of Materials and Energy, Yunnan University, Kunming, Yunnan 650500, P. R. China. E-mail: spguo@ynu.edu.cn
First published on 13th April 2026
Abstract
Growing large, high-quality nonlinear optical (NLO) crystals remains a central challenge for advancing ultraviolet (UV) laser technologies. KBe2BO3F2 (KBBF)-type crystals exhibit outstanding NLO properties but suffer from pronounced layer separation that impedes the growth of application-scale single crystals. Here, we introduce a three-dimensional (3D) charge-assisted hydrogen-bonded framework strategy by incorporating sulfanilamide and nitrate anions as hydrogen-bond donors and acceptors into a KBBF-like structure. This approach led to the successful synthesis of a novel ultraviolet (UV) NLO crystal, C6H9N2SO2·NO3 (SAN), which preserves a KBBF-like architecture while being stabilized by an extensive hydrogen-bonding network. SAN demonstrates a stronger second-harmonic generation (SHG) response (2.5 × KH2PO4, KDP) and higher birefringence (0.112 at 546 nm) compared with KBBF. Crucially, the 3D hydrogen-bonded network suppresses layer exfoliation and enables direct growth of centimeter-scale crystals (4.5 × 1 × 0.5 cm3) from aqueous solution by simple evaporation at room temperature. This work pioneers the integration of hydrogen-bonded framework design into the structural modification of NLO crystals. Through rational hydrogen-bond engineering within the KBBF-type framework, the crystal growth behavior has been significantly improved, establishing a generalizable strategy for modifying other layered functional crystalline materials.
Introduction
Nonlinear optical (NLO) crystals are indispensable for frequency conversion beyond intrinsic laser gain bandwidths, enabling coherent radiation in the ultraviolet (UV) and deep-UV regions for high-resolution lithography, photochemistry, and ultrafast science.1–13 Workhorse UV NLO crystals such as LiB3O5 (LBO), β-BaB2O4 (BBO), and KH2PO4 (KDP), together with KBe2BO3F2 (KBBF),14–17 have propelled the development of UV photonics over the past few decades. Among them, KBBF stands out for deep-UV second-harmonic generation (SHG), yet its practical deployment is severely constrained by toxic precursors and the intrinsic tendency toward layered growth, which complicates the growth of thick and crack-free single crystals suitable for device integration.18 In response, considerable research has been devoted to structural modification and compositional engineering aimed at overcoming these inherent limitations of KBBF and developing viable alternatives, leading to a number of important advances: compositional substitution of the toxic [BeO3F] groups and the introduction of interlayer covalent linkages have produced KBBF-family derivatives like Zn2BO3(OH), NaZnCO3(OH), β-Rb2Al2B2O7, and (NH4)3B11PO19F3,19–22 with reduced interlayer spacing while retaining appreciable NLO responses. Beyond covalent interlayer bridging, alternative strategies have been explored to constrain interlayer separation via hydrogen-bonding interactions, as demonstrated in NH4B4O6F, NH4Be2BO3F2, C(NH2)3SO3F and [C2N4H7O][NH2SO3].23–26 Nonetheless, the growth of large single crystals remains challenging for most KBBF-family compounds, with even millimeter-sized crystals often exhibiting pronounced lamellar morphology due to incomplete suppression of anisotropic growth. Therefore, a more effective strategy is needed to strengthen interlayer cohesion without sacrificing NLO performance.Hydrogen-bonded organic frameworks (HOFs) provide a promising blueprint.27,28 As an emerging class of crystalline porous materials, HOFs have undergone rapid development in recent years and been extensively applied in cutting-edge fields including gas adsorption/separation, proton conduction, and catalysis.29–31 The self-assembly via intermolecular hydrogen bonding endows these materials with high crystallinity and excellent solution processability.32,33 Moreover, the abundant hydrogen-bonding interactions in HOFs can effectively establish well-defined 3D frameworks. These characteristics provide valuable insights for addressing the inherent crystal growth issues in KBBF-type structures. Specifically, replacing the structural units of KBBF with functional groups that feature rich hydrogen-bonding environments is anticipated to facilitate the formation of 3D architectures. However, the relatively weak nature of hydrogen bonds poses challenges to structural stability. Strengthening intermolecular interactions through charge-assisted hydrogen bonding by integrating paired acids and bases would be a more optimal strategy, as such electrostatically enhanced interactions can significantly improve structural robustness while maintaining framework dimensionality.32,34
Guided by these considerations, we selected sulfanilamide (C6H8N2O2S) and nitric acid (HNO3) for combination based on the following rational design considerations (Fig. 1): (i) the –NH2 and –SO2NH2 groups in sulfanilamide can act as hydrogen bond donors and acceptors, enabling intermolecular hydrogen bonding. Additionally, the nitrate anion serves as an effective hydrogen bond acceptor, further enriching the hydrogen bond network; (ii) as a strong acid, nitric acid can form charge-assisted hydrogen bonds with sulfanilamide, which significantly enhances the structural stability through electrostatically enhanced interactions; (iii) both the benzene ring in sulfanilamide and the nitrate anion are π-conjugated units exhibiting high polarizability anisotropy and hyperpolarizability. Their rational alignment within the framework is conducive to enhancing the SHG response and birefringence. Furthermore, π–π stacking also contributes to the stabilization of the hydrogen-bonded framework. By leveraging these design principles, we successfully constructed a 3D hydrogen-bonded framework structurally analogous to KBBF, leading to the discovery of a novel UV NLO crystal C6H9N2SO2·NO3 (SAN). As intended, centimeter-sized single crystals of SAN were grown at room temperature via simple aqueous solution evaporation, with the largest crystal reaching 4.5 × 1 × 0.5 cm3 in size. Furthermore, SAN exhibits excellent NLO performance, surpassing KBBF in both SHG efficiency (2.5 × KDP) and birefringence (0.112). This work demonstrates the effectiveness of hydrogen-bonded framework engineering in simultaneously optimizing the structural architecture and crystal growth behavior of UV NLO materials, providing a viable and instructive pathway for future design of such materials.
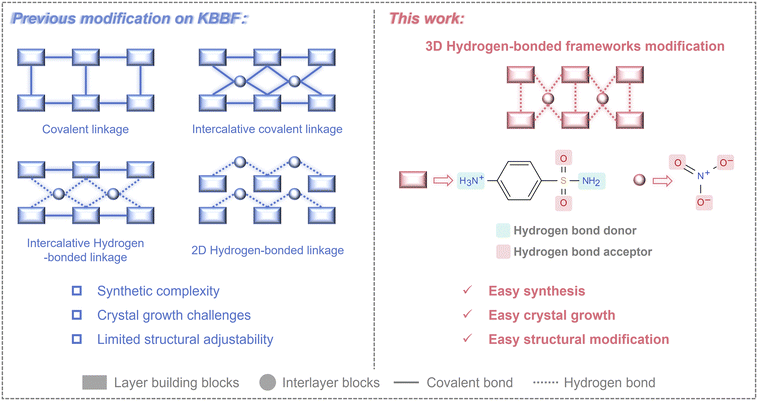 | ||
| Fig. 1 Schematic of modification strategies for KBBF-type crystals in previous studies and this work. | ||
Results and discussion
The growth of SAN crystals can be readily achieved under laboratory conditions without the need for specialized equipment or elevated temperatures (Fig. 2a). The crystal size demonstrates a dependence on container dimensions, indicating benign growth kinetics. Detailed procedures for crystal synthesis and growth are provided in the SI. To further elucidate the crystallization behavior of SAN, we employed the Bravais–Friedel–Donnay–Harker (BFDH) model to predict its crystal habit. As depicted in Fig. 2b, the simulated crystal morphology of SAN demonstrates a prominent blocky tendency, and there is no significant disparity in the growth rate (D value) of each crystal face along the direction perpendicular to the crystal plane (Fig. 2c). This observation suggests relatively low anisotropy in the crystal growth process. Single-crystal X-ray diffraction (SCXRD) and energy-dispersive X-ray spectroscopy (EDS) analysis results confirm the crystal structure and composition (Fig. S1); powder X-ray diffraction (PXRD) results verify phase purity for optical measurements (Fig. S2). SAN crystals are non-hygroscopic and retain integrity after at least six months under ambient conditions.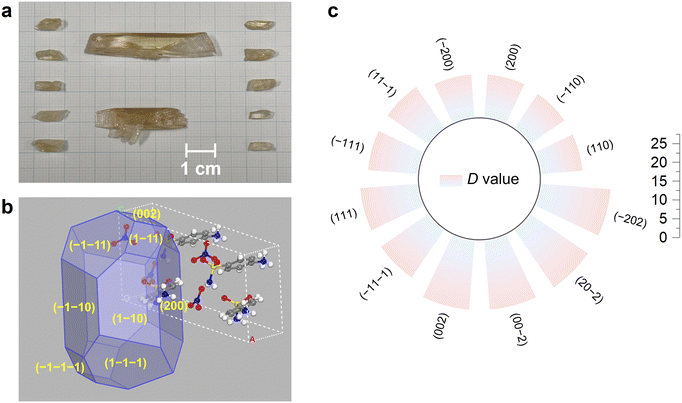 | ||
| Fig. 2 Photograph of as-grown SAN crystals (a); simulated crystal habit of SAN based on the BFDH model (b); simulated growth rates for individual crystal faces of the SAN crystal (c). | ||
SAN crystallizes in the non-centrosymmetric space group Cc (No. 9, detailed crystallographic information is presented in Tables S1 to S3). The asymmetric unit contains one [C6H9N2O2S]+ cation and one [NO3]− anion linked by charge-assisted N–H⋯O hydrogen bonds. A structural comparison between SAN and KBBF is shown in Fig. 3a and b. In KBBF, the honeycomb-like [Be2BO3F] 2D layers are built from [BeO3F] tetrahedra and [BO3] triangles (Fig. 3c), with K+ ions occupying interlayer sites as counterions. The interlayer interactions are dominated by K–F ionic bonds, resulting in a large interlayer spacing and contributing to its pronounced layering tendency. SAN features pseudo-2D layers composed of [C6H9N2O2S]+ moieties with [NO3]− occupying the interlayer regions (Fig. 3d). Notably, whereas the layers of KBBF are rigid due to extensive covalent bonding, the interlayer forces are weak ionic interactions, leading to strongly anisotropic growth. In contrast, the pseudo-2D layers in SAN are formed by [C6H9N2O2S]+ with relatively large intermolecular distances and no direct covalent linkages. These groups are interconnected via N–H⋯O hydrogen bonds between the interlayer [NO3]− anions. Furthermore, the [C6H9N2O2S]+ units across neighboring layers are also bridged by hydrogen bonds (Fig. 3e), giving rise to a highly interconnected zigzag layer topology. This extensive hydrogen-bonding network imparts greater structural homogeneity and enhanced dimensional control in SAN, effectively reducing and stabilizing the interlayer spacing. The maximum and minimum interlayer distances in SAN are 4.88 Å and 3.82 Å, respectively, both significantly smaller than the interlayer spacing in KBBF (6.25 Å).
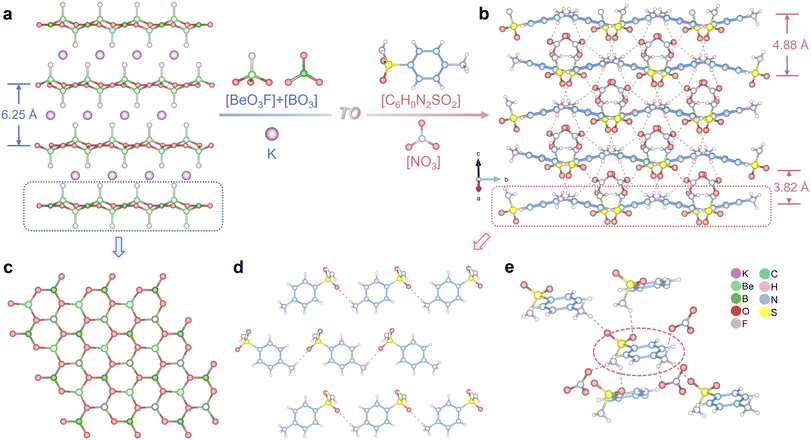 | ||
| Fig. 3 Structural comparison between KBBF and SAN (a and b); 2D/pseudo-2D layers of KBBF (c) and SAN (d); the hydrogen-bonding environment of [C6H9N2O2S]+ (e). | ||
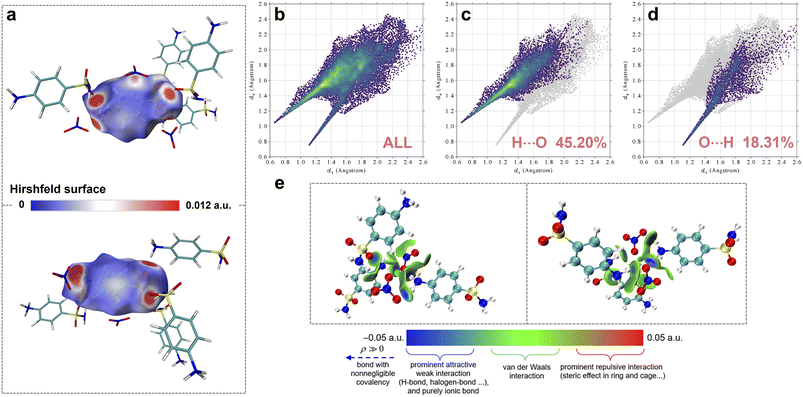
To gain deeper insights into the intermolecular interactions, a Hirshfeld surface analysis was performed on SAN.35–37 As illustrated in Fig. 4a, prominent hydrogen-bonding interactions are observed within the crystal structure. Specifically, the protonated amino group of [C6H9N2O2S]+ engages in strong N–H⋯O hydrogen bonds with both the neighboring –SO2NH2 and [NO3]−. Moreover, weak C–H⋯O contacts exist between aromatic hydrogen atoms and [NO3]−. Fingerprint plots and quantitative analyses of contact contributions derived from the Hirshfeld surface reveal that hydrogen bonds involving oxygen as an acceptor account for 63.5% of all intermolecular contacts in SAN, underscoring the dominant role of O-centered hydrogen bonding in constructing the architecture of SAN (Fig. 4b to d). The independent gradient model based on Hirshfeld partition (IGMH) isosurfaces further map the spatial distribution of these interactions (Fig. 4e),38,39 corroborating the pervasive hydrogen-bonding network responsible for the 3D framework.
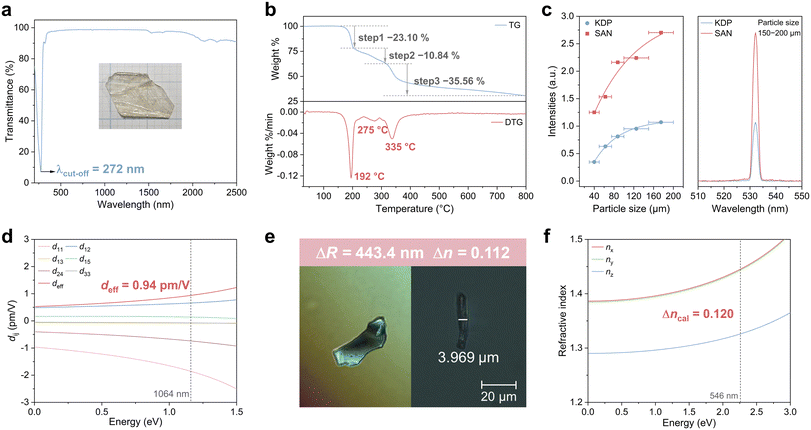
The ultraviolet–visible–near-infrared (UV-Vis-NIR) transmission spectrum of millimeter-scale SAN single crystals reveals that its absorption edge extends into the solar blind region, with a cut-off wavelength at 272 nm (Fig. 5a and S3), corresponding to an optical bandgap of 4.56 eV. Furthermore, the measured crystal maintains a transmittance exceeding 90% across the broad spectral range of 300–2500 nm, demonstrating excellent optical transparency and high crystalline quality. Therefore, SAN emerges as a promising candidate for 355 nm Nd:YAG sum frequency generation as well as other UV applications spanning the 300–400 nm range. The structural modification from KBBF to SAN entails the substitution of functional groups with extended π-conjugated modules, which strengthens the π–π* interactions. Such enhanced electronic delocalization leads to a narrowing of the band gap, consequently inducing a redshift in the UV cut-off edge of SAN compared to that of KBBF. The infrared spectrum of SAN is presented in Fig. S4. The N–H stretching vibration displays distinct red-shift and broadening features, whereas the antisymmetric and symmetric stretching vibrations of S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O exhibit an overall shift toward lower wavenumbers with intense peak characteristics. These observations indicate the presence of relatively strong N–H⋯O(SO2) interactions, which is consistent with the charge-assisted hydrogen bond model. The thermal behavior of SAN was systematically examined through thermogravimetric (TG) and derivative thermogravimetric (DTG) analyses (Fig. 5b). SAN remains thermally stable up to approximately 190 °C, beyond which three successive mass-loss stages occur, culminating in a total weight loss of about 69.5% and leaving a residual mass of roughly 30% at 800 °C. The first decomposition stage, centered at 192 °C and corresponding to a 23.1% mass reduction, is attributed primarily to the denitration or partial decomposition of the [NO3]− moiety. The experimental mass loss closely approximates the theoretical nitrate content (26.4%), suggesting the liberation of nitrogen oxides. The second stage, occurring near 275 °C with a 10.84% mass loss, likely involves further decomposition of nitrogen-containing intermediates and partial cleavage of the sulfonamide linkage. The third decomposition event, centered at 335 °C with a 35.56% weight loss, corresponds to the breakdown of the organic framework, including fragmentation of the carbon skeleton and sulfur oxidation, resulting in the evolution of SOx and hydrocarbon species. Collectively, these results demonstrate that SAN exhibits good thermal stability up to 192 °C, which exceeds that of several recently reported NLO crystals where hydrogen bonding serves as the primary linking interaction, including SbF3·Gly (105 °C), O3SCH2NH3 (187 °C), [CN3H6][C3H3O4] (157 °C), (C5H6ON)+(H2PO4)− (166 °C), and KNO3SO3NH3 (140 °C).40–44
O exhibit an overall shift toward lower wavenumbers with intense peak characteristics. These observations indicate the presence of relatively strong N–H⋯O(SO2) interactions, which is consistent with the charge-assisted hydrogen bond model. The thermal behavior of SAN was systematically examined through thermogravimetric (TG) and derivative thermogravimetric (DTG) analyses (Fig. 5b). SAN remains thermally stable up to approximately 190 °C, beyond which three successive mass-loss stages occur, culminating in a total weight loss of about 69.5% and leaving a residual mass of roughly 30% at 800 °C. The first decomposition stage, centered at 192 °C and corresponding to a 23.1% mass reduction, is attributed primarily to the denitration or partial decomposition of the [NO3]− moiety. The experimental mass loss closely approximates the theoretical nitrate content (26.4%), suggesting the liberation of nitrogen oxides. The second stage, occurring near 275 °C with a 10.84% mass loss, likely involves further decomposition of nitrogen-containing intermediates and partial cleavage of the sulfonamide linkage. The third decomposition event, centered at 335 °C with a 35.56% weight loss, corresponds to the breakdown of the organic framework, including fragmentation of the carbon skeleton and sulfur oxidation, resulting in the evolution of SOx and hydrocarbon species. Collectively, these results demonstrate that SAN exhibits good thermal stability up to 192 °C, which exceeds that of several recently reported NLO crystals where hydrogen bonding serves as the primary linking interaction, including SbF3·Gly (105 °C), O3SCH2NH3 (187 °C), [CN3H6][C3H3O4] (157 °C), (C5H6ON)+(H2PO4)− (166 °C), and KNO3SO3NH3 (140 °C).40–44
Since SAN crystallizes in a non-centrosymmetric space group, its particle-size-dependent SHG response was systematically investigated under 1064 nm laser irradiation. As shown in Fig. 5c, the SHG intensity of SAN increases with increasing particle size, consistent with phase-matching behavior. At particle sizes ranging from 150−200 µm, the SHG intensity of SAN reaches 2.5 times that of KDP of comparable dimensions, outperforming its parent compound KBBF (1.2 × KDP)18 and comparable to several recently reported excellent UV NLO crystals, such as La3Ga3Ge2S3O10 (2 × KDP), Rb3PbBi(P2O7)2 (2.8 × KDP) and Mg(C3O4H2)(H2O)2 (3 × KDP).45–47 We also investigated the NLO properties of SAN using theoretical methods (Fig. 5d). SAN crystallizes in the point group m, with six non-zero SHG coefficients d11, d12, d13, d15, d24 and d33. The calculated SHG coefficients at 1064 nm are −1.87, 0.66, −0.08, 0.14, −0.72 and −0.07 pm V−1, respectively, yielding an effective SHG coefficient deff of 0.94 pm V−1, which is approximately 2.4 times that of KDP (0.39 pm V−1). This result is in excellent agreement with experimental results. To further explore the origin of the NLO properties, we performed a group-resolved analysis to evaluate the contribution of individual structural units. The analysis reveals that the [C6H9N2O2S]+ group dominates the contributions to all SHG coefficients in SAN (Fig. S5). It is well established that the macroscopic NLO performance of a crystal depends not only on the intrinsic properties of its functional groups but also critically on their spatial arrangement within the structure. The hyperpolarizability of the [C6H9N2O2S]+ group was visualized using the unit-sphere representation method,48 revealing that its resultant vector lies coplanar with the benzene ring and is directed toward the sulfonamide moiety (Fig. S6a). Furthermore, the individual hyperpolarizability contributions from the [C6H9N2O2S]+ groups are orthogonally aligned along the c-axis, resulting in a coherent macroscopic polarization (Fig. S6b). This well-ordered spatial arrangement significantly enhances the overall SHG response of SAN.
The birefringence of SAN was experimentally determined using a polarizing microscope with a 546 nm light source (Fig. 5e). The thickness (T) of the measured crystal was 3.969 µm, and the corresponding optical path difference (ΔR) was found to be 443.4 nm. By applying the formula ΔR = Δn × T, the experimental birefringence Δnexp was derived as 0.112, which exceeds that of KBBF (0.077) and several members of the KBBF family,18 such as K15La7(BO3)12 (0.065), Rb3B11P2O23 (0.071) and KLi2CO3F (0.102).49–51 The enhanced birefringence enables materials to achieve an equivalent optical path difference over a reduced physical length, thereby offering significant advantages for the design of integrated optical systems and the development of miniaturized photonic devices. The augmented birefringence of SAN can be ascribed to the substitution of [BeO3F] and [BO3] units with the [C6H9N2O2S] group in SAN. The latter exhibits a markedly higher polarizability anisotropy (58.99) compared to 17.76 for [BeO3F] and 14.97 for [BO3], thereby significantly boosting the overall birefringence response. First-principles calculations were performed to investigate the birefringence of SAN. SAN crystallizes in the monoclinic system and belongs to the biaxial crystal system, with three principal refractive indices designated as nx, ny, and nz. These refractive indices follow the order nx > ny > nz. As a result, the theoretical birefringence at 546 nm, defined as Δncal = nx − nz, is computed to be 0.120, in good agreement with the experimental value (Fig. 5f). Based on the refractive index dispersion, we further calculated the phase-matching cutoff edge of SAN (Fig. S7). The results demonstrate that the phase-matching cutoff edge of SAN is 650 nm, which is consistent with the experimentally observed phase-matching behavior.
The electronic structure of SAN was computed to gain a more profound understanding of its structure–property relationship. As depicted in Fig. 6a, SAN is identified as a direct-bandgap semiconductor, with a theoretical bandgap of 3.59 eV. The deviation between the theoretical and experimental values can be ascribed to the intrinsic derivative discontinuity flaw of the GGA-PBE functional. Analyzing the density of states (DOS) of SAN reveals that near the Fermi level, the electronic states are predominantly occupied by C-2p, O-2p and N-2p orbitals (Fig. 6b). This finding suggests that the optical properties of SAN are chiefly governed by the [C6H9N2O2S]+ and [NO3]− groups. We performed a visualization of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) within a unit cell of SAN to elucidate their distributions. As shown in Fig. 6c, the HOMO is primarily localized on the [NO3]− group, whereas the LUMO displays a notable distribution in both [C6H9N2O2S]+ and [NO3]− moieties, indicating intermolecular charge transfer characteristics in SAN. The interaction between [C6H9N2O2S]+ and [NO3]− groups is essentially dominated by hydrogen bonds driven by electrostatic forces, rather than electronic delocalization or sharing, which aligns with our expectations. To further probe the electronic structure, we conducted a 3D electron localization function (ELF) analysis (Fig. 6d). The resulting isosurface maps reveal highly anisotropic electron distribution patterns centered on the [C6H9N2O2S]+ and [NO3]− groups, providing a clear electronic basis for the enhanced SHG response and birefringence observed in SAN relative to KBBF. Moreover, regions of high electron localization are clearly observed at both hydrogen bond donors and acceptors, confirming that the hydrogen bond network in SAN is reinforced through substantial charge polarization, thereby optimizing structural stability and crystal growth habit.
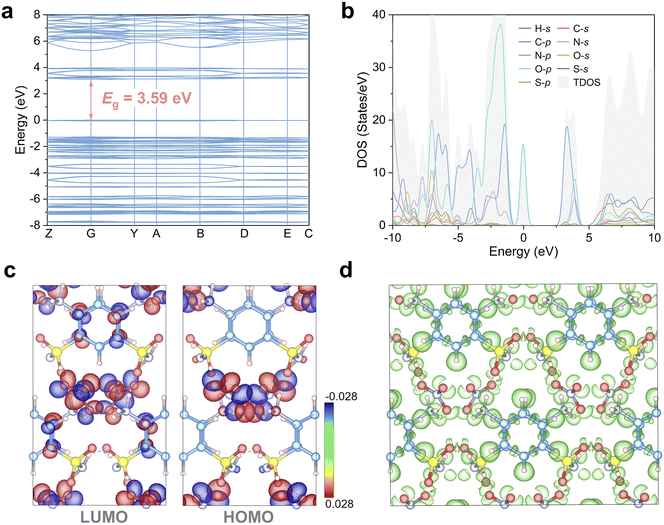 | ||
| Fig. 6 The electronic band structure (a); DOS (b); HOMO–LUMO isosurface (c); and 3D ELF map (d) of SAN. | ||
Conclusions
In summary, we have successfully developed a novel UV NLO crystal, C6H9N2SO2·NO3 (SAN), by incorporating a 3D charge-assisted hydrogen-bonded framework into a KBBF-like structural architecture. This strategy effectively overcomes the layer-separation issue inherent in KBBF and its derivatives, enabling the facile growth of large single crystals (up to 4.5 × 1 × 0.5 cm3) directly from aqueous solution at room temperature. SAN exhibits excellent NLO performance, including a short UV cut-off edge (272 nm), strong SHG (2.5 × KDP) and large birefringence (0.112 at 546 nm). Theoretical calculations corroborate the experimental results and reveal that the optimized alignment of [C6H9N2O2S]+ cations within the framework is responsible for the enhanced optical properties. This work demonstrates the great potential of hydrogen-bonded framework engineering as a general and effective strategy for modifying layered functional crystals. By rationally introducing charge-assisted hydrogen bonds, we have simultaneously improved crystal growth behavior and optical performance, paving the way for the development of next-generation UV NLO materials.Author contributions
Mingshu Zhang: conceptualization, visualization, writing-original draft; Shuya Zhao: data curation; Zhen-Cheng Wu: data curation; Yue Yin: data curation; Zheyu Zhang: data curation; Jiafeng Chen: data curation; Sheng-Ping Guo: writing–review & editing, funding acquisition; Yan Zhou: writing–review & editing, funding acquisition, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc09306f.Acknowledgements
This work was supported by National Natural Science Foundation of China (22401245 and 22371246), Yunnan Fundamental Research Projects (202401AS070120) and the Yunnan University Undergraduate Innovation Training Program Project (20251091).Notes and references
- X. Dong, L. Huang and G. Zou, Acc. Chem. Res., 2025, 58, 150–162 CrossRef CAS PubMed.
- G. Zou and K. M. Ok, Chem. Sci., 2020, 11, 5404–5409 RSC.
- C. Wu, G. Yang, M. G. Humphrey and C. Zhang, Coord. Chem. Rev., 2018, 375, 459–488 CrossRef CAS.
- W. Zhou and S.-P. Guo, Acc. Chem. Res., 2024, 57, 648–660 CrossRef CAS PubMed.
- M. Mutailipu, J. Han, Z. Li, F. Li, J. Li, F. Zhang, X. Long, Z. Yang and S. Pan, Nat. Photonics, 2023, 17, 694–701 CrossRef CAS.
- W. Zeng, J. Zhang, Y. Tian, H. Zeng, Z. Lin, L. Zhou, K. M. Ok and G. Zou, Angew. Chem., Int. Ed., 2025, 65, e20861 CrossRef PubMed.
- C. C. Jin, Y. Li, J. S. Kim, J.-H. Lim, H. Huang, C.-A. Chen, J. Lee, Y. Heo, B. Zhang, J. I. Jang and K. M. Ok, Angew. Chem., Int. Ed., 2025, 64, e202512618 CrossRef CAS PubMed.
- Y. Kang, C. Yang, J. Gou, Y. Zhu, Q. Zhu, W. Xu and Q. Wu, Angew. Chem., Int. Ed., 2024, 63, e202402086 CrossRef CAS PubMed.
- X. Zhang, X. Jiang, H. Gao, K. Duanmu, C. Wu, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2025, e202513438 CAS.
- H. Tian, C. Lin, Y. Zhou, X. Zhao, H. Fan, T. Yan, N. Ye and M. Luo, Angew. Chem., Int. Ed., 2023, 62, e202304858 CrossRef CAS PubMed.
- Y. Zhu, J. Gou, C. Yang, Q. Zhu, Y. Xiong and Q. Wu, Angew. Chem., Int. Ed., 2025, 64, e202509290 CrossRef CAS PubMed.
- C. Wu, X. Jiang, Z. Wang, H. Sha, Z. Lin, Z. Huang, X. Long, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2021, 60, 14806–14810 CrossRef CAS PubMed.
- Y. Yan, X. Wen, P. Tang, J. Lu, J. Chen, J. Wang, Z. Hu, N. Ye and G. Peng, Angew. Chem., Int. Ed., 2025, 64, e202516953 CrossRef CAS PubMed.
- C. Chen, Y. Wu, A. Jiang, B. Wu, G. You, R. Li and S. Lin, J. Opt. Soc. Am. B, 1989, 6, 616–621 CrossRef CAS.
- C. Chen, B. Wu, A. Jiang and G. You, Sci. China, Ser. B, 1985, 28, 235–243 Search PubMed.
- J. J. De Yoreo, A. K. Burnham and P. K. Whitman, Int. Mater. Rev., 2002, 47, 113–152 CrossRef CAS.
- D. Cyranoski, Nature, 2009, 457, 953–955 CrossRef CAS PubMed.
- C. T. Chen, G. L. Wang, X. Y. Wang and Z. Y. Xu, Appl. Phys. B, 2009, 97, 9–25 CrossRef CAS.
- X. Wang, F. Zhang, L. Gao, Z. Yang and S. Pan, Adv. Sci., 2019, 6, 1901679 CrossRef CAS PubMed.
- G. Peng, C. Lin and N. Ye, J. Am. Chem. Soc., 2020, 142, 20542–20546 CrossRef CAS PubMed.
- T. T. Tran, N. Z. Koocher, J. M. Rondinelli and P. S. Halasyamani, Angew. Chem., Int. Ed., 2017, 56, 2969–2973 CrossRef CAS PubMed.
- B. Cheng, Z. Li, Y. Chu, A. Tudi, M. Mutailipu, F. Zhang, Z. Yang and S. Pan, Natl. Sci. Rev., 2022, 9, nwac110 CrossRef CAS PubMed.
- G. Shi, Y. Wang, F. Zhang, B. Zhang, Z. Yang, X. Hou, S. Pan and K. R. Poeppelmeier, J. Am. Chem. Soc., 2017, 139, 10645–10648 CrossRef CAS PubMed.
- G. Peng, N. Ye, Z. Lin, L. Kang, S. Pan, M. Zhang, C. Lin, X. Long, M. Luo, Y. Chen, Y.-H. Tang, F. Xu and T. Yan, Angew. Chem., Int. Ed., 2018, 57, 8968–8972 CrossRef CAS PubMed.
- M. Luo, C. Lin, D. Lin and N. Ye, Angew. Chem., Int. Ed., 2020, 59, 15978–15981 CrossRef CAS PubMed.
- X. Wen, Y. Yan, J. Lu, X. Shi, P. Tang, J. Chen, G. Yang, G. Peng, H. Yu, H. Zhang, Z. Hu, J. Wang and N. Ye, Angew. Chem., Int. Ed., 2025, 64, e202424153 CrossRef CAS PubMed.
- H. Ali, B. A. Alwan, A. Abdulrahman, D. Yue and A. Hayat, Mater. Today, 2025, 88, 783–813 CrossRef CAS.
- R.-B. Lin and B. Chen, Chem, 2022, 8, 2114–2135 CAS.
- A. Ebadi Amooghin, H. Sanaeepur, M. Ghomi, R. Luque, H. Garcia and B. Chen, Coord. Chem. Rev., 2024, 505, 215660 CrossRef CAS.
- Y. Sun, J. Wei, Z. Fu, M. Zhang, S. Zhao, G. Xu, C. Li, J. Zhang and T. Zhou, Adv. Mater., 2023, 35, 2208625 CrossRef CAS PubMed.
- H. Yuan, J. Xiao, A.-A. Zhang, Z.-B. Fang and T.-F. Liu, EnergyChem, 2025, 7, 100151 CrossRef CAS.
- X. Song, Y. Wang, C. Wang, D. Wang, G. Zhuang, K. O. Kirlikovali, P. Li and O. K. Farha, J. Am. Chem. Soc., 2022, 144, 10663–10687 CrossRef CAS PubMed.
- B. Wang, R.-B. Lin, Z. Zhang, S. Xiang and B. Chen, J. Am. Chem. Soc., 2020, 142, 14399–14416 CrossRef CAS PubMed.
- X.-Y. Chen, L.-H. Cao, X.-T. Bai and X.-J. Cao, Chem.–Eur. J., 2024, 30, e202303580 CrossRef CAS PubMed.
- M. A. Spackman and D. Jayatilaka, CrystEngComm, 2009, 11, 19–32 RSC.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- T. Lu, J. Chem. Phys., 2024, 161, 082503 CrossRef CAS PubMed.
- T. Lu and Q. Chen, J. Comput. Chem., 2022, 43, 539–555 CrossRef CAS PubMed.
- T. Lu, Angew. Chem., Int. Ed., 2025, 64, e202504895 CrossRef CAS PubMed.
- Z. Bai, J. Lee, C.-L. Hu, G. Zou and K. M. Ok, Chem. Sci., 2024, 15, 6572–6576 RSC.
- Z. Wang, X. Jiang, X. Wang, K. Duanmu, L. Bai, C. Wu, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, J. Am. Chem. Soc., 2026, 148, 135–140 CrossRef CAS PubMed.
- Z. Chen, C. Liu, C. Li, J. Lu, Z. Yang, S. Pan and M. Mutailipu, Adv. Opt. Mater., 2025, 13, 2500631 CrossRef CAS.
- J. Lu, X. Liu, M. Zhao, X.-B. Deng, K.-X. Shi, Q.-R. Wu, L. Chen and L.-M. Wu, J. Am. Chem. Soc., 2021, 143, 3647–3654 CrossRef CAS PubMed.
- H. Tian, C. Lin, X. Zhao, S. Fang, H. Li, C. Wang, N. Ye and M. Luo, Mater. Today Phys., 2022, 28, 100849 CrossRef CAS.
- H. Yan, Y. Matsushita, K. Yamaura and Y. Tsujimoto, Angew. Chem., Int. Ed., 2021, 60, 26561 CrossRef CAS PubMed.
- X. Lu, Z. Chen, X. Shi, Q. Jing and M.-H. Lee, Angew. Chem., Int. Ed., 2020, 59, 17648 CrossRef CAS PubMed.
- L. Wu, C. Lin, H. Tian, Y. Zhou, H. Fan, S. Yang, N. Ye and M. Luo, Angew. Chem., Int. Ed., 2024, 63, e202315647 CrossRef CAS PubMed.
- A. Tuer, S. Krouglov, R. Cisek, D. Tokarz and V. Barzda, J. Comput. Chem., 2011, 32, 1128–1134 CrossRef CAS PubMed.
- F. Bu, X. Liu, H. Yu, R. Liu, Z. Hu, J. Wang, Y. Wu and H. Wu, Inorg. Chem., 2024, 63, 24059–24064 CrossRef CAS PubMed.
- H. Liu, H. Wu, Z. Hu, J. Wang, Y. Wu, P. S. Halasyamani and H. Yu, ACS Mater. Lett., 2023, 5, 155–161 CrossRef CAS.
- Q. Wang, W. Song, Y. Lan, L. Cao, L. Huang, D. Gao, J. Bi and G. Zou, Inorg. Chem. Front., 2022, 9, 3590–3597 RSC.
| This journal is © The Royal Society of Chemistry 2026 |