Enhanced photocatalytic activity of Pt/WO3 photocatalyst combined with TiO2 nanoparticles by polyelectrolyte-mediated electrostatic adsorption
Abstract
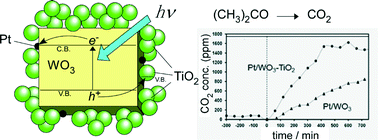
An electrostatic adsorption approach was used to realize a composite structure in which larger metal oxide crystalline particles were surrounded by metal oxide nanoparticles. Poly(diallyldimethylammonium chloride) (PDDA) and poly(sodium 4-styrenesulfonate) were alternately adsorbed onto crystalline tungsten trioxide (WO3) particles (ca. 200 nm) via a layer-by-layer assembly, followed by adsorption of TiO2 particles (ca. 6 nm) on the surfaces covered by PDDA. After calcination to remove the adsorbed polymer layers, Pt particles were dispersed on the composite structure by photodeposition. The resulting Pt/WO3–TiO2 composite photocatalyst showed a higher rate of activity towards the photocatalytic decomposition of gaseous acetone under visible light irradiation (λ > 420 nm) compared with that of Pt/WO3. Pt/WO3–TiO2 also converted acetone to CO2 almost completely, whereas the amount of CO2 produced over Pt/WO3 was much smaller than that expected for the complete oxidation of acetone. The enhanced activity of Pt/WO3–TiO2 was ascribed to hole transfer from the valence band of WO3 to that of TiO2, which likely suppressed electron–hole recombination and enabled the oxidation reaction to take place on the surface of the TiO2 particles.

 Please wait while we load your content...
Please wait while we load your content...