Characterization of the low energy conformations and differential reactivities of d-glucose and d-mannose based oxepines†
Abstract
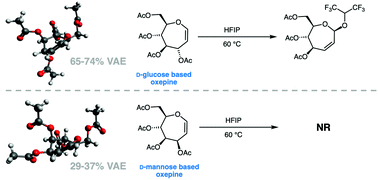
Carbohydrate-based oxepines are valuable intermediates for the synthesis of septanose carbohydrates. Here we report the characterization of the preferred conformations of D-glucose and D-mannose based oxepines 1 and 2 using computational chemistry and NMR spectroscopy. Monte Carlo conformational searches on 1 and 2 were performed, followed by DFT optimization and single-point energy calculations on the low energy conformations of each oxepine. Coupling constants were computed for all unique conformations at a B3LYP/6-31G(d,p)u+1s level of theory and weighted based on a Boltzmann distribution. These values were then compared to the experimental values collected using 3JH,H values collected from 1H NMR spectra. Information from the MC/DFT approach was then used in a least squares method that correlated DFT calculated and observed 3JH,H coupling constants. The conformations of 1 and 2 are largely governed by a combination of the rigidifying enol ether element in combination with the reduction of unfavorable interactions. The vinylogous anomeric effect (VAE) emerged as a consequence, rather than a driver of conformations. Oxepine 1 showed greater reactivity in Ferrier rearrangement reactions relative to oxepine 2, in line with its greater %VAE.
- This article is part of the themed collections: Chemical Biology in OBC and Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...