Copper-catalyzed enantioselective synthesis of bridged bicyclic ketals from 1,1-disubstituted-4-methylene-1,6-hexanediols and related alkenols†
Abstract
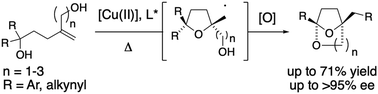
Bridged bicyclic ketals display a range of bioactivities. Their catalytic enantioselective synthesis from acyclic 1,1-disubstituted alkene diols is disclosed. This reaction combines asymmetric catalysis with a distal radical migration. Alkynes and arenes undergo the group transfer.


 Please wait while we load your content...
Please wait while we load your content...