QM/MM MD simulations reveal an asynchronous PCET mechanism for nitrite reduction by copper nitrite reductase†
Abstract
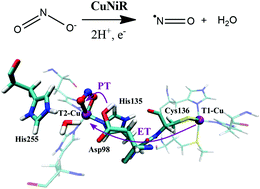
Nitrite reductases are enzymes that aid in the denitrification process by catalyzing the reduction of nitrite to nitric oxide gas. Since this reaction is the first committed step that involves gas formation, it is regarded to be a vital step for denitrification. However, the mechanism of copper-containing nitrite reductase is still under debate due to the discrepancy between the theoretical and experimental data, especially in terms of the roles of secondary shell residues Asp98 and His255 and the electron transfer mechanism between the two copper sites. Herein, we revisited the nitrite reduction mechanism of A. faecalis copper nitrite reductase using QM(B3LYP)/MM-based metadynamics. It is found that the intramolecular electron transfer from T1-Cu to T2-Cu occurs via an asynchronous proton-coupled electron transfer (PCET) mechanism, with electron transfer (ET) preceding proton transfer (PT). In particular, we found that the ET process is driven by the conformation conversion of Asp98 from the gatekeeper to the proximal one, which is much more energy-demanding than the PCET itself. These results highlight that the inclusion of an electron donor is vital to investigate electron-transfer related processes such as PCET.
- This article is part of the themed collection: Quantum Theory: The Challenge of Transition Metal Complexes


 Please wait while we load your content...
Please wait while we load your content...