Quaternized poly(styrene-co-vinylbenzyl chloride) anion exchange membranes: role of different ammonium cations on structural, morphological, thermal and physio-chemical properties†
Abstract
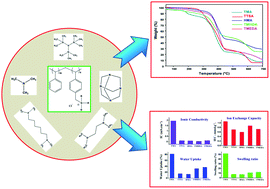
Different ammonium cation containing anion exchange membranes are investigated in this study. Anion exchange polymers have been prepared from styrene and vinylbenzyl chloride monomers by free radical polymerization, followed by a quaternization process. Amines such as trimethylamine (TMA), tris(trimethylsilyl)amine (TTSA), hexamine (HMA), N,N,N′,N′-tetramethyl hexanediamine (TMHDA), and N,N,N′,N′-tetramethyl ethylenediamine (TMEDA) are used in this study. FT-IR and 13C-NMR results confirm the successful quaternization of the co-polymers with different amines. TGA results reveal that the thermal stability of the anion exchange polymers is as high as 165 °C. SEM images show that the membrane morphological features are tuned by the different ammonium cations. The ionic conductivity, ion-exchange capacity and water uptake values of the membranes also change with the different ammonium cations, which reveal the distinctive roles of the different amines used. Among the membranes, the membrane containing the trimethylammonium (TMA+) cation demonstrates the highest ionic conductivity, ion-exchange capacity and water uptake. On the other hand, the hexaminium (HMA+) containing anion exchange membrane displays the lowest ionic conductivity, ion-exchange capacity and water uptake due to the highly entangled structure of the polymer caused by the cross-linking of the polymer units.


 Please wait while we load your content...
Please wait while we load your content...