An effect of Ag(i)-substitution at Cu sites in CuGaS2 on photocatalytic and photoelectrochemical properties for solar hydrogen evolution†
Abstract
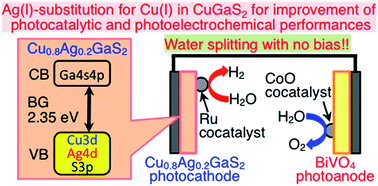
We investigated an effect of Ag(I)-substitution at the Cu sites in CuGaS2 with a chalcopyrite structure on physicochemical, photocatalytic and photoelectrochemical properties. Cu1−xAgxGaS2 (x = 0–0.8) solid solutions possessed 2.34–2.35 eV of band gaps. Ru-loaded Cu1−xAgxGaS2 (x = 0–1.0) powders showed photocatalytic activity for hydrogen evolution from an aqueous solution containing SO32− and S2− as electron donors under simulated sunlight irradiation. The photocatalytic activity of CuGaS2 (x = 0) was improved, when excess Ga2S3 was added in a starting material for the preparation to give a single phase of a chalcopyrite structure. Cu1−xAgxGaS2 (x = 0.1–1.0) showed higher activity than the CuGaS2 (x = 0), when prepared with the optimum excess Ga2S3. Cu1−xAgxGaS2 (x = 0–0.8) electrodes gave cathodic photocurrents under visible light irradiation, indicating that they possessed p-type characters. Cu0.8Ag0.2GaS2 gave the largest cathodic photocurrent and possessed the most positive onset potential of the cathodic photocurrent among the Cu1−xAgxGaS2 (x = 0–0.8) electrodes. Loading a Ru cocatalyst increased the cathodic photocurrent. Solar water splitting proceeded without an external bias using a photoelectrochemical cell consisting of the Ru-loaded Cu0.8Ag0.2GaS2 photocathode and a CoO-loaded BiVO4 photoanode. Thus, the photocatalytic and photoelectrochemical performances of CuGaS2 were improved by Ag(I)-substitution at the Cu sites.
- This article is part of the themed collection: Artificial Photosynthesis

 Please wait while we load your content...
Please wait while we load your content...