Ni–M (M = Sn and Sb) intermetallic-based catalytic functional layer as a built-in safeguard for hydrocarbon-fueled solid oxide fuel cells†
Abstract
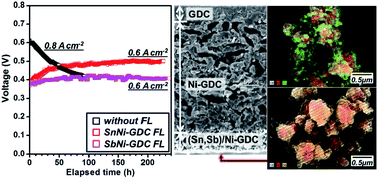
One critical hindrance to solid oxide fuel cells (SOFCs) with a Ni-based anode fueled with hydrocarbons is the coking (carbon deposition) phenomenon; on anodes exposed directly to hydrocarbon fuels, solid carbonaceous materials accumulate during SOFC operation, resulting in severe degradation of the catalytic activity of Ni and structural damage to the anode. This may be overcome by Ni–M (M = Sn, Sb) intermetallic compound-based catalytic functional layers (CFLs) on the anode surface. Sn or Sb incorporated onto Ni surface develops into intermetallic compounds under reducing atmosphere, thereby interrupting the interaction between Ni and the solid carbon produced by hydrocarbon cracking beforehand. Moreover, the hydrophilic properties of Sn or Sb facilitate the formation of hydroxyl groups on their surfaces by reaction with the water vapor generated by the electrochemical oxidation of fuel, and the solid carbon accumulated onto the intermetallic surface is oxidized by the hydroxyls and removed from the anode. With these roles of Sn or Sb in M/Ni–Ce0.8Gd0.2O1.9 CFLs, we show here that the operational stability of SOFCs can be maintained more than 200 h in methane fuel at intermediate temperatures.

 Please wait while we load your content...
Please wait while we load your content...