Towards a robust description of intrinsic protein disorder using nuclear magnetic resonance spectroscopy†
Abstract
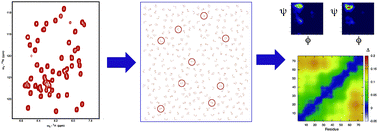
In order to understand the conformational behaviour of Intrinsically Disordered
- This article is part of the themed collection: Intrinsically Disordered Proteins

 Please wait while we load your content...
Please wait while we load your content...