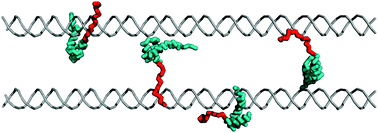
Intrinsically disordered regions, terminal tails, and flexible linkers are abundant in DNA-binding proteins and play a crucial role by increasing the affinity and specificity of DNA binding. Disordered tails often undergo a disorder-to-order transition during interactions with DNA and improve both the kinetics and thermodynamics of specific DNA binding. The DNA search by proteins that interact nonspecifically with DNA can be supported by disordered tails as well. The disordered tail may increase the overall protein–DNA interface and thus increase the affinity of the protein to the DNA and its sliding propensity while slowing linear diffusion. The exact effect of the disordered tails on the sliding rate depends on the degree of positive charge clustering, as has been shown for homeodomains and p53 transcription factors. The disordered tails, which may be viewed as DNA recognizing subdomains, can facilitate intersegment transfer events that occur via a “monkey bar” mechanism in which the domains bridge two different DNA fragments simultaneously. The “monkey bar” mechanism can be facilitated by internal disordered linkers in multidomain proteins that mediate the cross-talks between the constituent domains and especially their brachiation dynamics and thus their overall capability to search DNA efficiently. The residue sequence of the disordered tails has unique characteristics that were evolutionarily selected to achieve the optimized function that is unique to each protein. Perturbation of the electrostatic characteristics of the disordered tails by post-translational modifications, such as acetylation and phosphorylation, may affect protein affinity to DNA and therefore can serve to regulate DNA recognition. Modifying the disordered protein tails or the flexibility of the inter-domain linkers of multidomain proteins may affect the cross-talk between the constituent domains so as to facilitate the search kinetics of non-specific DNA sequences and increase affinity to the specific sequences.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...