Second-order nonlinear optical properties of eight-membered centrosymmetric cyclic borasiloxanes†
Abstract
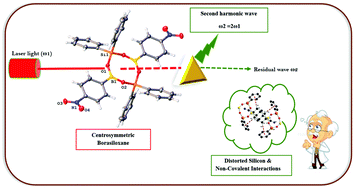
In an efficient 2+2 cyclocondensation reaction, electron-withdrawing arylboronic acids with diphenylsilanediol were converted to eight-membered cyclic borasiloxanes [(RPhBO)(PhSiO)]2 [R = F (1), 2,4-F (2), CF3 (3), CN (4), NO2 (5)]. All these compounds were characterized by elemental analysis, FT-IR, and NMR (1H, 13C, 19F, 29Si and 11B) and structurally confirmed by single-crystal X-ray diffraction studies. Compounds 1–5 crystallized in the centrosymmetric space group, revealing an eight-membered ring (B2O4Si2) configuration with organic substitutions, which occupied axial and equatorial positions. Compounds 1 and 2 feature 2D network hydrogen bonding whereas compound 3 has a solid-state 3D supramolecular architecture. The other two compounds 4 and 5 exhibit only C–H⋯π interactions which lead to a 1D polymeric structure and these intermolecular interactions influence crystal packing. Photophysical properties were studied by UV-visible and fluorescence spectroscopic techniques. In addition, the optical band gaps (Eg) of compounds 1–5 were determined by diffuse reflectance spectra and compound 5 has a lower band gap value than the others, due to the strong electron-withdrawing nitro group in compound 5. The thermal behaviour of compounds 4 and 5 has been investigated using thermogravimetric analysis and differential thermal analysis, both compounds being stable up to 250 °C. The nonlinear optical response of the crystalline powdered borasiloxanes by Q-switched Nd-YAG laser and second harmonic generation (SHG) efficiency of borasiloxanes 4 and 5 were found very similar to those of potassium dihydrogen phosphate. The SHG efficiency of the centrosymmetric borasiloxanes mainly arises from the distorted silicon atom and non-covalent interactions which preclude the dipoles in the antiparallel arrangement in crystal packing. Further, optical and nonlinear properties of the borasiloxanes were investigated by density functional theory calculations.


 Please wait while we load your content...
Please wait while we load your content...