Heterogeneous formulation of the tricopper complex for efficient catalytic conversion of methane into methanol at ambient temperature and pressure†
Abstract
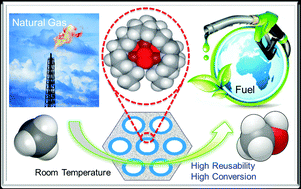
The development of a heterogeneous catalyst capable for efficient selective conversion of methane into methanol with multiple turnovers under ambient conditions is reported here. The catalyst is assembled by immobilizing into mesoporous silica nanoparticles the tricopper complex [CuICuICuI(7-N-Etppz)]1+, where 7-N-Etppz stands for the organic ligand 3,3′-(1,4-diazepane-1,4-diyl)bis[1-(4-ethylpiperazine-1-yl)propan-2-ol]. This tricopper cluster complex has been previously shown to mediate efficient methane oxidation without over-oxidation in homogeneous solution when the catalytic turnover is driven by hydrogen peroxide in acetonitrile. The turnover mechanism of the catalyst is similar between the two formulations. However, the heterogeneous formulation exhibits dramatically higher catalytic efficiencies and turnover numbers, with commensurate improvements in chemical yields, offering the most proficient catalyst for the selective conversion of methane into methanol at room temperature developed to date. To explain the efficient methane oxidation, the over-solubility of nonpolar gases, such as methane, in liquids confined in nanoporous solids is evoked. The much higher solubility of methane within the pores of the mesoporous silica nanoparticles, as compared to the bulk solubility, led to very efficient turnover of the concentrated confined methane. This success underscores the advantages of using nanoparticles to support chemical catalysts for this difficult chemical transformation under these conditions.

 Please wait while we load your content...
Please wait while we load your content...