Biomass-derived functional materials as carriers for enzymes: towards sustainable and robust biocatalysts
Abstract
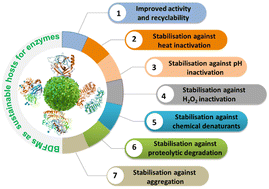
The unique catalytic properties of enzymes have led to the production of useful medicinal intermediates, foods, and biofuels from sustainable sources. However, the instability of soluble/free enzymes under several challenging conditions (e.g., pH, proteolysis, temperature, ionic potential, chemical denaturants) restricts the use of enzyme-based biocatalysts. Encapsulation of enzymes on suitable carriers would mitigate the instability issues faced in robust biocatalysis. An “ideal” carrier material employed for protein immobilization should be nontoxic, scalable, biocompatible, and should not compromise the biological activity and structure of proteins/enzymes. Thus, biodegradable and renewable biomass-derived functional materials (BDFMs) are envisaged as promising carriers for enzymes. BDFMs have in-built chemical functionalities and desirable physicochemical properties that enable their use in enzyme catalysis at the industrial scale. Numerous BDFMs have been used as immobilization matrices to improve the biocatalytic activity and stability of various enzymes. These solid materials are renewable and environmentally friendly compared with synthetic polymers. This review highlights the advancements, challenges and prospects in the emerging field of BDFMs (cellulose, silk protein, chitin, chitosan, lignocellulose, and a combination of biopolymers such as chitin/lignin and chitosan/alginate) for immobilization of enzymes (e.g., α-chymotrypsin, cytochrome c, carbonic anhydrase, glucose oxidase, ribonuclease, cholesterol oxidase, alkaline phosphatase, β-glucosidase, lipase, horseradish peroxidase, catalase, tyrosinase, acetylcholinesterase, amylase, invertase, protease, laccase, β-galactosidase, and several others) for biocatalytic processes. This review also describes the relationship between the structural properties and functionality of several enzymes immobilized in BDFMs, and profiles the impact of pH, temperature, reusability, stability of storage, and the activity of these enzymes. Future perspectives in this promising field, as well as potential difficulties, are discussed. This review will help in refining biocatalysis technologies whereby biomass-derived, environmentally friendly materials are employed as enzyme supports.
- This article is part of the themed collection: Renewables showcase


 Please wait while we load your content...
Please wait while we load your content...