Operando deconvolution of the degradation mechanisms of iron–nitrogen–carbon catalysts in proton exchange membrane fuel cells†
Abstract
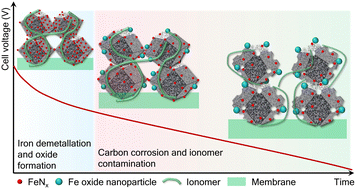
Developing platinum-free catalysts for proton exchange membrane fuel cells (PEMFCs) is crucial to the hydrogen economy. While iron–nitrogen–carbon (Fe–N–C) catalysts are currently the most promising non-Pt alternative for the ORR, their poor stability in PEMFCs are challenging to understand due to the multitude of degradation mechanisms occurring simultaneously. Herein, we deconvolute these mechanisms in PEMFC over 60 hours under high load (1 A cm−2) using advanced electrochemical methods such as the distribution of relaxation times. This allows us to identify when iron demetallation and carbon corrosion occur and unveil an intricate degradation pathway through the operando deterioration of the triple-phase boundary. Firstly, up to 75% of the Fe–N–C active sites become inactive through iron demetallation which initially drives the voltage losses (<10 hours). Then, a five-fold increase in carbon corroded species and four-fold reduction in proton transport kinetics in the catalyst layer lengthen the gas, ionic and electronic pathways to the catalytic sites, reducing the oxygen reduction reaction (ORR) rate by three-fold and becoming the predominant degradation mechanism. These insights are captured via a combination of cyclic voltammetry and the distribution of relaxation times. Altogether, these provide unprecedented insights into this degradation mechanism while proposing operando standards to characterize unstable electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...