Two-dimensional metal–organic frameworks as efficient electrocatalysts for bifunctional oxygen evolution/reduction reactions†
Abstract
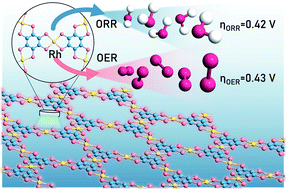
With the continuous development of sustainable and renewable energy, the electrocatalytic water cycle and rechargeable metal–air batteries are attracting increasing attention. As two main reactions, oxygen evolution/reduction reactions (OER/ORR) require highly efficient bifunctional electrocatalysts. Herein, by first-principles calculations, we have systematically investigated the OER and ORR performance for a new class of two-dimensional (2D) metal–organic frameworks (MOFs) M3(C6S6)2, with M including Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ir, and Pt. It is demonstrated that Rh3(C6S6)2 possesses promising bifunctional OER/ORR activity, with calculated overpotentials of 0.43 and 0.42 V for the OER and ORR, respectively. Moreover, the volcano plots and contour maps regarding activity vs. intermediate adsorption strength are established to describe the activity trends of the OER and ORR for M3(C6S6)2. A combination of the d-band center and crystal orbital Hamilton populations (COHPs) is employed to correlate the activity and electronic structure of the catalyst. The catalytic origin of OER and ORR activities can be ascribed to the simple d-electron number of the central metal of M3(C6S6)2. Our findings not only provide cost-effective bifunctional oxygen electrocatalysts but also offer important insights into advancing clean energy technology.


 Please wait while we load your content...
Please wait while we load your content...