Iodide-mediated Cu catalyst restructuring during CO2 electroreduction†
Abstract
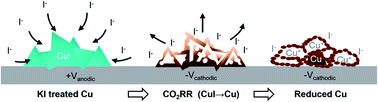
Catalyst restructuring during electrochemical reactions is a critical but poorly understood process that determines the underlying structure–property relationships during catalysis. In the electrocatalytic reduction of CO2 (CO2RR), it is known that Cu, the most favorable catalyst for hydrocarbon generation, is highly susceptible to restructuring in the presence of halides. Iodide ions, in particular, greatly improved the catalyst performance of Cu foils, although a detailed understanding of the morphological evolution induced by iodide remains lacking. It is also unclear if a similar enhancement transfers to catalyst particles. Here, we first demonstrate that iodide pre-treatment improves the selectivity of hexagonally ordered Cu-island arrays towards ethylene and oxygenate products. Then, the morphological changes in these arrays caused by iodide treatment and during CO2RR are visualized using electrochemical transmission electron microscopy. Our observations reveal that the Cu islands evolve into tetrahedral CuI, which then become 3-dimensional chains of copper nanoparticles under CO2RR conditions. Furthermore, CuI and Cu2O particles re-precipitated when the samples are returned to open circuit potential, implying that iodide and Cu+ species are present within these chains. This work provides detailed insight into the role of iodide, and its impact on the prevailing morphologies that exist during CO2RR.


 Please wait while we load your content...
Please wait while we load your content...