Understanding the electrochemical reaction mechanisms of precious metals Au and Ru as cathode catalysts in Li–CO2 batteries†
Abstract
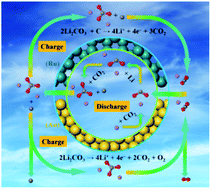
The electrochemical reaction mechanisms of Au and Ru as cathode catalysts in Li–CO2 batteries are firstly studied by first-principles density functional theory (DFT) calculations. During the discharge process, the free energy changes of different intermediates during the nucleation processes of lithium oxalate (Li2C2O4) and lithium carbonate (Li2CO3) are systematically investigated, showing that on both Au and Ru surfaces, Li2C2O4 nucleations are kinetically favorable for the discharge intermediate product. However, Li2CO3 nucleations are thermodynamically favorable for the final discharge product, and Li2C2O4 cannot be stabilized as the final product anymore and has to decompose into Li2CO3 as the final discharge product. During the charging process, the Li2CO3 decomposition mechanism is also studied on both Au and Ru surfaces, showing that the discharge products of Li2CO3 and carbon can be much more reversibly decomposed on the Ru catalyst than on the Au surface. The electrochemical free energy diagrams are also introduced to identify the overpotentials compared with the experimental results. A series of electrochemical reaction pathways proposed here can effectively evaluate the catalytic activity of the catalysts toward the discharging and charging processes, thereby providing more insights into the design and optimization cathode catalysts for Li–CO2 batteries.


 Please wait while we load your content...
Please wait while we load your content...