Developing organoboranes as phase transfer catalysts for nucleophilic fluorination using CsF†
Abstract
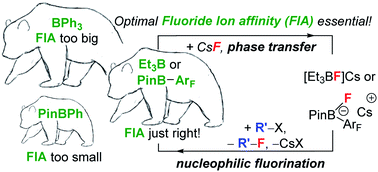
Despite the general high fluorophilicity of boron, organoboranes such as BEt3 and 3,5-(CF3)2C6H3–BPin are shown herein for the first time, to our knowledge, to be effective (solid to solution) phase-transfer catalysts for the fluorination of certain organohalides with CsF. Significant (up to 30% e.e.) chiral induction during nucleophilic fluorination to form β-fluoroamines using oxazaborolidine (pre)catalysts and CsF also can be achieved. Screening different boranes revealed a correlation between calculated fluoride affinity of the borane and nucleophilic fluorination reactivity, with sufficient fluoride affinity required for boranes to react with CsF and form Cs[fluoroborate] salts, but too high a fluoride affinity leading to fluoroborates that are poor at transferring fluoride to an electrophile. Fluoride affinity is only one component controlling reactivity in this context; effective fluorination also is dependent on the ligation of Cs+ which effects both the phase transfer of CsF and the magnitude of the [Cs⋯F-BR3] interaction and thus the B–F bond strength. Effective ligation of Cs+ (e.g. by [2.2.2]-cryptand) facilitates phase transfer of CsF by the borane but also weakens the Cs⋯F–B interaction which in turn strengthens the B–F bond – thus disfavouring fluoride transfer to an electrophile. Combined, these findings indicate that optimal borane mediated fluorination occurs using robust (to the fluorination conditions) boranes with fluoride affinity of ca. 105 kJ mol−1 (relative to Me3Si+) under conditions where a signficant Cs⋯F–B interaction persists.
- This article is part of the themed collection: Most popular 2022 catalysis articles


 Please wait while we load your content...
Please wait while we load your content...