Remote carbamate-directed site-selective benzylic C–H oxygenation via synergistic copper/TEMPO catalysis at room temperature†
Abstract
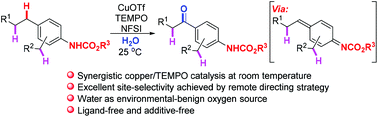
Facilitated by a remote directing strategy, a benzylic C(sp3)–H oxygenation with water at room temperature through a ligand- and additive-free synergistic copper/TEMPO-catalysed radical relay pathway with N-fluorobenzenesulfonimide (NFSI) as a terminal oxidant is disclosed. With the assistance of carbamate directing groups, this oxygenation takes place site-selectively at the para-positions of the directing groups when multiple benzyl sites are available in a substrate. Mechanistic studies reveal the crucial role of catalytic TEMPO as a recyclable SET reductant, as well as the significance of the combined synergistic catalysis and remote directing strategy to the excellent chemoselectivity and site-selectivity. It has also been demonstrated that this novel strategy is promising for future studies on C(sp3)–H functionalisation.


 Please wait while we load your content...
Please wait while we load your content...